Advanced Synthesis of Stable Sulfonic Acid Salts for Antifolate Drug Manufacturing
Introduction to Novel Antifolate Intermediate Technology
The development of robust synthetic routes for antifolate agents, particularly pyrrolo[2,3-d]pyrimidines, remains a critical challenge in oncology drug manufacturing. Patent CN1475477A introduces a transformative methodology that addresses the inherent instability of key aldehyde precursors. Traditionally, the synthesis of these vital building blocks involved handling highly reactive and unstable alpha-halogen aldehydes, which complicated purification and storage. This patent discloses a series of novel sulfonic acid metal cation salts of formula IV, which serve as stable, isolable intermediates. By converting the transient aldehyde species into crystalline bisulfite adducts, the process significantly enhances the feasibility of large-scale production. This technological leap allows for the decoupling of synthetic steps, enabling manufacturers to produce and stockpile high-purity intermediates before proceeding to the final cyclization stages.
For R&D directors and process chemists, the implications of this invention are profound. It shifts the paradigm from telescoped, high-risk reactions to a modular approach where intermediate quality can be rigorously controlled. The ability to isolate the intermediate as a stable salt means that impurities generated during the initial coupling can be effectively removed via crystallization, a unit operation that is far more scalable and cost-effective than chromatography. This report analyzes the mechanistic advantages and commercial viability of this route, positioning it as a superior choice for reliable pharmaceutical intermediate supplier networks aiming to optimize their antifolate portfolios.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Prior art methods for synthesizing the requisite aldehydes, such as those disclosed by Taylor and Harrington or via Larock palladium-catalyzed coupling, suffer from significant drawbacks that hinder commercial scalability. As illustrated in the background of the patent, conventional routes often yield complex mixtures of desired products and unwanted by-products that are extremely difficult to separate. The target aldehydes, typically of general formula XIV or III, possess inherent chemical instability, making them prone to decomposition during isolation attempts. This instability forces manufacturers to use these intermediates immediately in situ, preventing any opportunity for quality control or storage. Furthermore, the difficulty in obtaining pure aldehyde forms leads to variability in downstream reactions, ultimately affecting the purity and yield of the final antifolate API. These factors collectively increase the cost of goods and introduce substantial supply chain risks.
The Novel Approach
The novel process described in the patent overcomes these hurdles by introducing a selective production method for general formula IV compounds. Instead of struggling to isolate the unstable free aldehyde, the process converts the reaction mixture directly into a sulfonic acid metal cation salt. This transformation is achieved by reacting the crude aldehyde mixture with a metal bisulfite agent, such as sodium bisulfite, in a suitable solvent system. The resulting salt precipitates as a crystalline solid, allowing for easy filtration and washing to remove soluble impurities and catalyst residues. 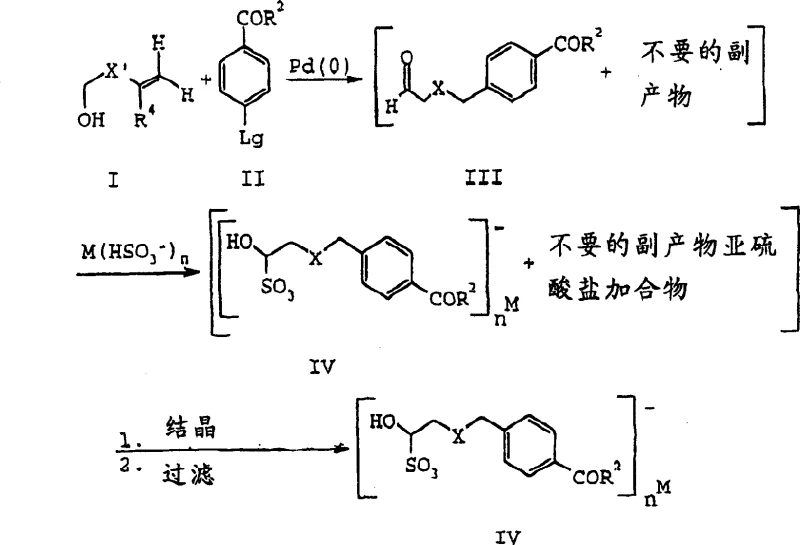 This approach not only simplifies the purification workflow but also stabilizes the molecule for long-term storage. The ability to handle the intermediate as a stable solid facilitates cost reduction in pharmaceutical intermediates manufacturing by minimizing waste and maximizing throughput. The process is versatile, accommodating various substituents and protecting groups, making it applicable to a wide range of antifolate analogues.
This approach not only simplifies the purification workflow but also stabilizes the molecule for long-term storage. The ability to handle the intermediate as a stable solid facilitates cost reduction in pharmaceutical intermediates manufacturing by minimizing waste and maximizing throughput. The process is versatile, accommodating various substituents and protecting groups, making it applicable to a wide range of antifolate analogues.
Mechanistic Insights into Palladium-Catalyzed Coupling and Bisulfite Addition
The core of this innovative synthesis lies in the efficient palladium(0)-catalyzed coupling of an allylic alcohol (Formula I) with an aryl halide (Formula II). The reaction proceeds under mild thermodynamic conditions, typically utilizing a base such as lithium acetate and a phase-transfer catalyst like tetrabutylammonium bromide. The palladium catalyst, often introduced as palladium(II) chloride, facilitates the formation of the carbon-carbon bond, generating the aldehyde intermediate (Formula III) in situ. Crucially, the reaction conditions are optimized to minimize side reactions; for instance, when the leaving group is iodide, the reaction can proceed at room temperature, whereas bromides may require heating to approximately 60-65°C. This flexibility allows for precise control over the reaction kinetics, ensuring high conversion rates while preserving the integrity of sensitive functional groups.
Following the coupling, the mechanism shifts to nucleophilic addition. The aldehyde carbonyl carbon is attacked by the bisulfite anion (HSO3-), forming a stable hydroxy-sulfonate adduct. This step is thermodynamically favorable and drives the equilibrium towards the salt formation. The choice of metal cation (M) influences the solubility and crystallization properties of the final product. For example, sodium and potassium salts exhibit different precipitation behaviors, allowing process engineers to tune the isolation step for maximum recovery. 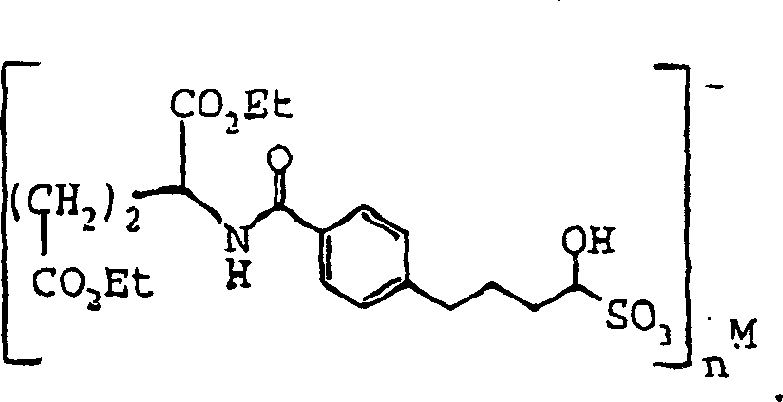 This mechanistic understanding is vital for impurity control. By optimizing the stoichiometry of the bisulfite reagent (typically 0.95-1.0 equivalents) and the solvent composition (e.g., ethyl acetate and ethanol mixtures), manufacturers can selectively crystallize the desired isomer while leaving impurities in the mother liquor. This level of control is essential for meeting the stringent purity specifications required for high-purity pharmaceutical intermediates.
This mechanistic understanding is vital for impurity control. By optimizing the stoichiometry of the bisulfite reagent (typically 0.95-1.0 equivalents) and the solvent composition (e.g., ethyl acetate and ethanol mixtures), manufacturers can selectively crystallize the desired isomer while leaving impurities in the mother liquor. This level of control is essential for meeting the stringent purity specifications required for high-purity pharmaceutical intermediates.
How to Synthesize Stable Sulfonic Acid Salts Efficiently
Implementing this synthesis route requires careful attention to reaction parameters and workup procedures to ensure optimal yield and purity. The process begins with the preparation of the coupling mixture, followed by the critical bisulfite addition and crystallization steps. Detailed operational guidelines are essential for reproducibility, especially when scaling from laboratory to pilot plant. The following section outlines the standardized protocol derived from the patent examples, highlighting key checkpoints for quality assurance. For a comprehensive breakdown of the specific reagents, temperatures, and timing required for each stage, please refer to the detailed synthesis guide below.
- Perform palladium-catalyzed coupling of an allylic alcohol and an aryl halide in the presence of a thermodynamic base and phase-transfer catalyst.
- Extract the resulting aldehyde mixture into an organic solvent and react with a metal bisulfite agent to form the sulfonic acid salt.
- Induce crystallization by adjusting solvent volumes and filter the pure sulfonic acid metal cation salt product.
Commercial Advantages for Procurement and Supply Chain Teams
From a procurement and supply chain perspective, the adoption of this sulfonic acid salt technology offers tangible benefits that extend beyond mere chemical elegance. The primary advantage lies in the stabilization of the supply chain for critical antifolate intermediates. By converting unstable liquids into stable solids, the inventory management of these materials becomes significantly more predictable and secure. This stability reduces the risk of material degradation during transport and storage, ensuring that the raw materials arriving at the production site meet the required specifications. Furthermore, the simplified purification process eliminates the need for expensive and time-consuming chromatographic separations, which are often bottlenecks in multi-kilogram production runs.
- Cost Reduction in Manufacturing: The elimination of complex purification steps directly translates to lower operational costs. Traditional methods often require multiple distillation or chromatography steps to remove palladium residues and side products, which consume significant amounts of solvents and stationary phases. In contrast, the crystallization-based isolation of the sulfonic acid salt is a low-cost unit operation. Additionally, the ability to recover and recycle solvents like ethyl acetate and ethanol further enhances the economic efficiency of the process. The reduced consumption of high-purity reagents and the minimization of waste disposal costs contribute to a substantially lower cost of goods sold, making this route highly attractive for cost-sensitive generic drug production.
- Enhanced Supply Chain Reliability: The robustness of this synthetic route ensures a consistent supply of high-quality intermediates. Since the intermediate can be stored for extended periods without degradation, manufacturers can build up safety stock to buffer against fluctuations in demand or raw material availability. This decoupling of synthesis steps means that a disruption in the final cyclization step does not necessarily halt the production of the precursor, providing greater flexibility in production scheduling. Moreover, the use of commercially available starting materials, such as aryl halides and allylic alcohols, reduces dependency on specialized custom synthesis vendors, thereby mitigating supply chain risks associated with single-source suppliers.
- Scalability and Environmental Compliance: The process is inherently scalable, having been demonstrated to work effectively with standard industrial equipment. The use of aqueous workups and common organic solvents aligns well with existing waste treatment infrastructure in chemical plants. The crystallization step generates a solid waste stream that is easier to handle and dispose of compared to the complex liquid waste streams generated by chromatographic purifications. This environmental compatibility simplifies regulatory compliance and reduces the environmental footprint of the manufacturing process. The ability to scale from 100 kgs to 100 MT annual commercial production without significant process re-engineering makes this technology a viable solution for meeting global market demand.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this sulfonic acid salt synthesis technology. These answers are derived directly from the experimental data and claims presented in the patent documentation, providing a reliable basis for decision-making. Understanding these nuances is crucial for technical teams evaluating the feasibility of adopting this route for their specific product pipelines. The responses cover aspects ranging from reaction conditions to the handling of chiral centers, ensuring a comprehensive overview of the process capabilities.
Q: Why are sulfonic acid metal cation salts preferred over free aldehydes for antifolate synthesis?
A: Free aldehydes of formula III are inherently unstable and difficult to purify. Converting them into sulfonic acid metal cation salts (Formula IV) creates stable, crystalline solids that can be stored, purified, and handled in large quantities without degradation.
Q: What catalysts are essential for the novel coupling reaction described in the patent?
A: The process relies on a Palladium(0) source, such as palladium(II) chloride, typically used in conjunction with a phase-transfer catalyst like tetrabutylammonium bromide and a thermodynamic base like lithium acetate to drive the coupling efficiency.
Q: How does this method improve supply chain reliability for antifolate APIs?
A: By generating a stable, isolable intermediate, manufacturers can decouple the synthesis steps. This allows for bulk production and quality verification of the intermediate before committing to the final cyclization, reducing the risk of batch failure in late-stage synthesis.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Antifolate Intermediate Supplier
At NINGBO INNO PHARMCHEM, we recognize the strategic importance of stable and scalable intermediate technologies in the competitive landscape of oncology drug development. Our team of expert process chemists has extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that your project transitions smoothly from benchtop to full-scale manufacturing. We are committed to delivering high-purity pharmaceutical intermediates that meet the rigorous standards of the global market. Our state-of-the-art facilities are equipped with rigorous QC labs and advanced analytical instrumentation to guarantee stringent purity specifications for every batch we produce, giving you confidence in the consistency and quality of your supply chain.
We invite you to collaborate with us to leverage this advanced synthesis technology for your antifolate projects. Our technical procurement team is ready to provide a Customized Cost-Saving Analysis tailored to your specific volume requirements and timeline. By partnering with us, you gain access to our deep expertise in palladium-catalyzed couplings and crystallization engineering. Please contact us today to request specific COA data and route feasibility assessments, and let us help you optimize your manufacturing strategy for maximum efficiency and profitability.
