Advanced Synthesis of 3,5-Asymmetric BODIPY Dyes for High-Performance Imaging Applications
Advanced Synthesis of 3,5-Asymmetric BODIPY Dyes for High-Performance Imaging Applications
The landscape of functional fluorescent dyes is undergoing a significant transformation, driven by the increasing demand for near-infrared (NIR) probes that offer superior tissue penetration and minimal background interference in biological systems. A pivotal development in this sector is documented in patent CN111793371A, which discloses a novel class of 3,5-position asymmetrically modified BODIPY (Boron-dipyrromethene) near-infrared fluorescent dyes. Unlike traditional symmetric analogues, these asymmetric structures provide tunable photophysical properties essential for advanced applications such as intracellular fluorescence imaging and optoelectronic materials. The core innovation lies in a streamlined synthetic strategy that bypasses the cumbersome multi-step protocols typically required to introduce asymmetry at the pyrrole ring positions. By leveraging a direct Knoevenagel condensation, this methodology enables the efficient construction of extended conjugated systems that red-shift absorption and emission wavelengths into the desirable NIR window. For research directors and procurement specialists seeking a reliable pharma intermediate supplier, understanding the nuances of this synthetic route is critical for securing high-quality materials for next-generation diagnostic and imaging tools.
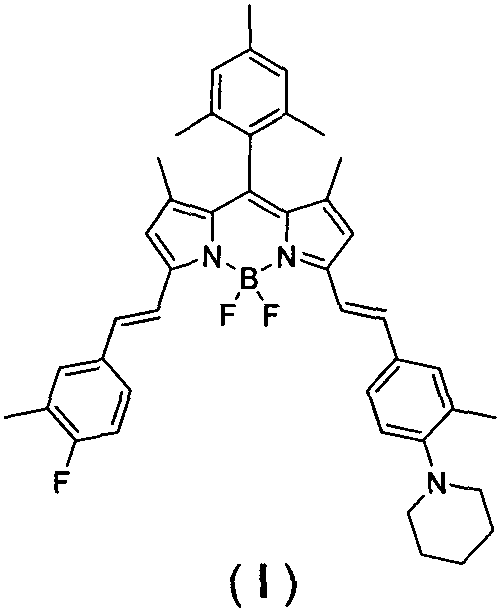
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of asymmetric BODIPY derivatives has been fraught with significant technical hurdles that impact both cost and scalability. Conventional routes often require the sequential construction of the dipyrromethene core using different aldehydes or pyrroles, necessitating rigorous protection and deprotection strategies to prevent statistical mixtures of symmetric byproducts. This lack of regioselectivity frequently results in complex reaction crude mixes that are difficult to separate, leading to substantial losses in overall yield and purity. Furthermore, traditional methods for extending conjugation to achieve NIR emission often involve harsh conditions or expensive transition metal catalysts, which introduce heavy metal impurities that are unacceptable for biological applications. The cumulative effect of these inefficiencies is a supply chain characterized by long lead times, high production costs, and inconsistent batch-to-batch quality, posing a severe bottleneck for the commercial scale-up of complex fluorescent dyes.
The Novel Approach
The methodology outlined in the patent data represents a paradigm shift by utilizing a post-functionalization strategy on a pre-formed symmetric BODIPY core. Instead of building asymmetry from scratch, the process employs a Knoevenagel condensation between a methyl-substituted BODIPY precursor and an aromatic aldehyde, specifically 3-methyl-p-fluorobenzaldehyde. This approach is revolutionary because it achieves the desired asymmetric modification in a single operational step, effectively collapsing a multi-stage synthesis into a concise transformation. The use of piperidine serves a dual purpose, acting simultaneously as a base catalyst and a reactant component, which simplifies the reagent profile and reduces the overall material inventory required. This simplification not only accelerates the reaction timeline but also inherently improves the impurity profile by eliminating intermediate isolation steps where degradation often occurs. For stakeholders focused on cost reduction in fluorescent dye manufacturing, this direct route offers a compelling value proposition by minimizing unit operations and maximizing process throughput.
Mechanistic Insights into Piperidine-Catalyzed Knoevenagel Condensation
The chemical elegance of this synthesis relies on the activation of the methyl groups at the 3 and 5 positions of the BODIPY core, which possess sufficient acidity to undergo condensation with aldehydes under mild basic conditions. In this specific protocol, piperidine facilitates the formation of an enamine intermediate or acts as a base to generate a carbanion at the methyl position, which then nucleophilically attacks the carbonyl carbon of the aldehyde. The presence of p-toluenesulfonic acid (PTSA) creates a buffered acidic environment that aids in the dehydration step, driving the equilibrium towards the formation of the styryl double bond. This dual-catalyst system ensures high conversion rates while maintaining the integrity of the sensitive BODIPY fluorophore, which can be prone to demetallation or decomposition under strongly acidic or basic extremes. The result is the formation of a rigid, planar conjugated system that extends the pi-electron cloud, directly correlating to the observed red-shift in absorption and emission spectra.
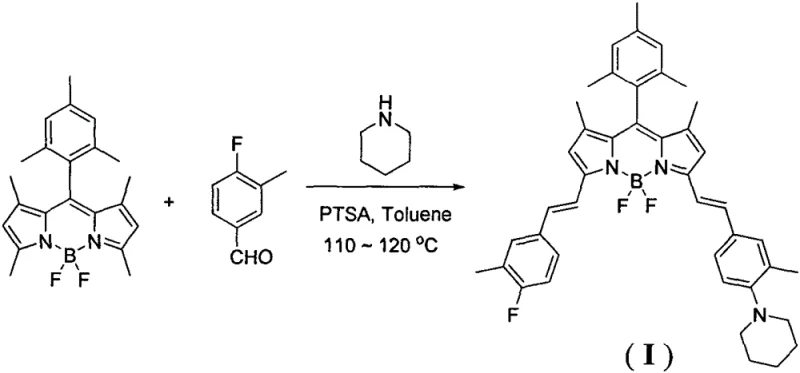
Controlling the stoichiometry and reaction parameters is paramount for ensuring high purity and minimizing the formation of bis-condensed byproducts or unreacted starting materials. The patent specifies a molar ratio of the BODIPY derivative to the aldehyde of approximately 1:1.8 to 1:2.0, which provides a slight excess of the aldehyde to drive the reaction to completion without overwhelming the system with difficult-to-remove impurities. The use of a Dean-Stark apparatus during the reflux in toluene is a critical engineering control that continuously removes water generated during the condensation, shifting the thermodynamic equilibrium towards the product side according to Le Chatelier's principle. This meticulous attention to water removal is essential for achieving high conversion in condensation reactions and prevents the hydrolysis of the imine-like intermediates. For R&D teams, understanding these mechanistic details allows for precise troubleshooting and optimization when scaling the process from gram to kilogram quantities, ensuring that the high-purity OLED material or imaging agent specifications are consistently met.
How to Synthesize 3,5-Asymmetric BODIPY Dye Efficiently
The practical execution of this synthesis is designed for robustness and ease of handling, making it highly suitable for industrial adoption. The process begins by charging a reaction vessel with the BODIPY core, the specific aldehyde, and the acid catalyst, followed by the addition of the solvent system. The reaction is then heated to reflux, allowing the thermal energy to overcome the activation barrier for the condensation. Following the reaction period, standard workup procedures involving extraction and drying are employed to isolate the crude product. While the general workflow is straightforward, precise control over temperature and reaction time is necessary to balance conversion with product stability. For detailed operational parameters and safety considerations, please refer to the standardized synthesis guide below.
- Charge a round-bottom flask equipped with a Dean-Stark trap with the BODIPY core, 3-methyl-p-fluorobenzaldehyde, and p-toluenesulfonic acid.
- Dissolve the mixture in toluene and piperidine, then heat to reflux at 110-120°C for 4 to 7 hours to drive the condensation.
- Cool the reaction, extract with dichloromethane, wash with water, dry over sodium sulfate, and purify via silica gel column chromatography.
Commercial Advantages for Procurement and Supply Chain Teams
From a supply chain perspective, the adoption of this one-step condensation methodology offers profound advantages in terms of operational efficiency and risk mitigation. By eliminating the need for multiple synthetic steps and intermediate purifications, the manufacturing footprint is significantly reduced, leading to lower capital expenditure on equipment and reduced labor costs per kilogram of output. The reliance on commodity chemicals such as toluene, piperidine, and substituted benzaldehydes ensures that the raw material supply chain is resilient and less susceptible to geopolitical disruptions or shortages of exotic reagents. This stability is crucial for procurement managers who need to guarantee continuous supply for long-term clinical or commercial projects. Furthermore, the simplified process flow reduces the generation of hazardous waste streams, aligning with increasingly stringent environmental regulations and lowering the costs associated with waste disposal and compliance auditing.
- Cost Reduction in Manufacturing: The most significant economic driver of this technology is the drastic reduction in process complexity. By consolidating the synthesis into a single reactor train, manufacturers eliminate the yield losses inherent in multi-step sequences where material is lost during each isolation and purification stage. The removal of expensive transition metal catalysts, which often require specialized scavenging resins to meet residual metal limits for pharmaceutical grades, further drives down the bill of materials. Additionally, the shorter cycle time per batch increases the asset utilization rate of the production facility, allowing for higher throughput without the need for additional reactor capacity. These factors combine to create a substantially lower cost of goods sold (COGS), providing a competitive edge in the pricing of high-purity electronic chemical intermediates.
- Enhanced Supply Chain Reliability: The robustness of the reaction conditions contributes directly to supply security. The use of standard reflux temperatures and common organic solvents means that the process can be easily transferred between different manufacturing sites without requiring specialized high-pressure or cryogenic equipment. This flexibility allows for a diversified manufacturing network, reducing the risk of supply interruption due to site-specific issues. Moreover, the high selectivity of the reaction minimizes the formation of hard-to-separate impurities, which often cause batch failures and delays in quality control release. A more predictable and consistent manufacturing process translates to more reliable delivery schedules for downstream customers relying on these dyes for critical imaging applications.
- Scalability and Environmental Compliance: Scaling this chemistry from laboratory to commercial production is facilitated by the homogeneous nature of the reaction mixture and the absence of exothermic hazards that typically plague large-scale condensations. The ability to use a Dean-Stark trap for water removal is a scalable unit operation that functions effectively from liters to thousands of liters. From an environmental standpoint, the atom economy of the condensation is favorable, and the avoidance of heavy metals simplifies the effluent treatment process. This alignment with green chemistry principles not only reduces environmental liability but also enhances the brand reputation of the supply chain partners as sustainable manufacturers of specialty chemicals.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the production and application of these asymmetric BODIPY derivatives. The answers are derived directly from the patented technical data to ensure accuracy and relevance for potential partners. Understanding these details is essential for evaluating the feasibility of integrating this material into your specific product pipeline.
Q: What is the primary advantage of this asymmetric BODIPY synthesis method?
A: The primary advantage is the ability to achieve 3,5-position asymmetric modification in a single synthetic step via Knoevenagel condensation, significantly reducing process complexity compared to traditional multi-step approaches.
Q: What are the optimal reaction conditions for this transformation?
A: The reaction utilizes toluene as a solvent with piperidine acting as both a catalyst and reactant, alongside p-toluenesulfonic acid. The mixture is heated to 110-120°C under reflux conditions for 4 to 7 hours.
Q: What are the photophysical properties of the resulting dye?
A: The synthesized dye exhibits a maximum absorption wavelength red-shifted to 639nm and a fluorescence emission wavelength reaching 678nm, making it highly suitable for near-infrared biological imaging.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable BODIPY Dye Supplier
The development of advanced fluorescent probes like the 3,5-asymmetric BODIPY dye described in patent CN111793371A represents a significant opportunity for innovation in the life sciences and electronic materials sectors. At NINGBO INNO PHARMCHEM, we recognize the critical importance of translating such promising academic and patent technologies into robust commercial realities. As a dedicated CDMO partner, we possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that your transition from benchtop discovery to market launch is seamless. Our facilities are equipped with state-of-the-art rigorous QC labs capable of verifying stringent purity specifications, including residual solvent analysis and trace metal testing, which are vital for materials intended for biological imaging or optoelectronic devices.
We invite you to collaborate with us to leverage this efficient synthetic route for your specific needs. Our technical team is ready to conduct a Customized Cost-Saving Analysis tailored to your volume requirements, identifying further opportunities for process optimization. We encourage you to contact our technical procurement team today to request specific COA data and route feasibility assessments. By partnering with us, you gain access to a supply chain that prioritizes quality, consistency, and technical excellence, empowering you to bring next-generation imaging and sensing solutions to market faster and more efficiently.
