Revolutionizing Bio-Fluorescent Probes: Scalable Synthesis of Carboxyl-BODIPY Dyes
The landscape of biochemical analysis is constantly evolving, driven by the demand for probes that offer superior sensitivity, stability, and ease of use. A pivotal advancement in this domain is documented in Chinese patent CN102061103A, which introduces a novel class of boron fluoride complexed dipyrromethene (BODIPY) fluorescent dyes. Unlike conventional fluorophores that often suffer from poor photostability or complex synthesis, these new derivatives are engineered with specific carboxylic acid functionalities that enhance their solubility and reactivity in biological systems. The core innovation lies in a streamlined synthetic strategy that bypasses the limitations of traditional aldehyde-based condensations, utilizing acid anhydrides to construct the dipyrromethene scaffold in a single pot. This technological leap not only improves the photophysical properties—such as achieving fluorescence quantum yields between 0.6 and 0.8—but also addresses critical manufacturing bottlenecks related to yield and scalability.
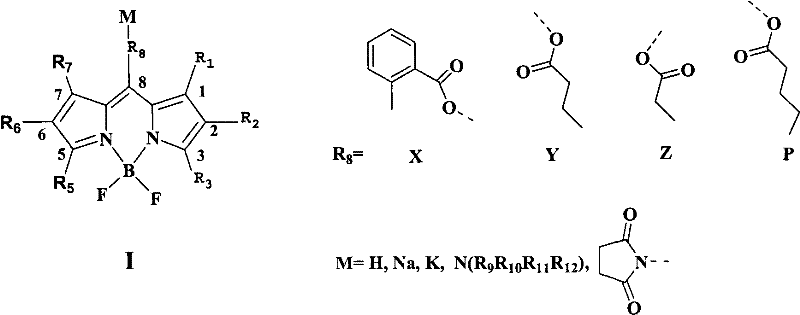
For research directors and procurement specialists seeking a reliable fluorescent dye supplier, understanding the underlying chemistry is paramount. The dyes described in this patent possess a general Formula I structure where the 8-position is substituted with various alkyl chains terminating in carboxylic acid groups (structures X, Y, Z, or P). This specific architectural choice is not arbitrary; it is designed to prevent the self-aggregation phenomena often observed in hydrophobic dyes at low concentrations (1×10⁻⁵ M). By ensuring the dye remains monomeric and fully dispersed in aqueous media, the technology guarantees consistent signal intensity and reliable quantification in high-throughput screening environments. Furthermore, the ability to tune the R groups allows for precise modulation of lipophilicity, facilitating optimal cell membrane penetration while maintaining intracellular solubility.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of carboxyl-functionalized BODIPY dyes has been plagued by inefficiencies that drive up costs and limit availability. Traditional routes often rely on the condensation of substituted pyrroles with specific aldehydes or acid chlorides, a process that can require up to six distinct synthetic steps. Literature precedents, such as those cited in the patent background, report overall yields as low as 12%, which is economically unsustainable for large-scale production. Moreover, the starting materials for these conventional methods, particularly specialized aldehydes with protected carboxyl groups, are often expensive and difficult to source in bulk. The multi-step nature of these syntheses also introduces multiple purification stages, increasing solvent consumption and waste generation, which contradicts modern green chemistry principles. Additionally, the harsh conditions sometimes required for acyl chloride reactions can lead to side reactions and impurity profiles that are challenging to remove, ultimately compromising the purity required for sensitive biological applications.
The Novel Approach
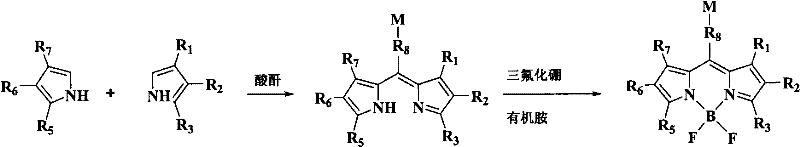
The methodology disclosed in patent CN102061103A represents a paradigm shift by employing organic dibasic acid anhydrides—such as succinic, glutaric, or phthalic anhydride—as the central bridging unit. This approach leverages the inherent reactivity of the anhydride ring, which opens during the condensation with two molecules of substituted pyrrole to simultaneously form the dipyrromethene backbone and introduce the free carboxylic acid functionality. This "kill two birds with one stone" strategy drastically reduces the step count and eliminates the need for pre-functionalized aldehyde precursors. The reaction is conducted under mild conditions (15-120°C) in common organic solvents like acetonitrile or dichloromethane, achieving yields of up to 25%, which is a significant improvement over prior art. This streamlined process not only enhances cost reduction in biochemical reagent manufacturing but also simplifies the supply chain by utilizing commodity chemicals as key starting materials.
Mechanistic Insights into Acid Anhydride-Mediated Cyclization
The success of this synthetic route hinges on the precise control of the acid-catalyzed condensation between the pyrrole and the anhydride. Mechanistically, the reaction initiates with the protonation of the anhydride or the activation of the pyrrole ring, facilitating a nucleophilic attack at the carbonyl carbon. The subsequent ring-opening of the anhydride generates a transient acyl-pyrrole intermediate, which rapidly undergoes a second condensation with another pyrrole molecule to close the dipyrromethene system. A critical aspect of this mechanism is the in situ elimination of water to form the methine bridge, a step that must be carefully managed to prevent the polymerization of the pyrrole starting material. The patent data indicates that maintaining the pyrrole concentration between 0.1% and 20% (optimally 4-8%) is essential to suppress the formation of viscous polymeric byproducts, which are a common failure mode in pyrrole chemistry. This concentration control ensures that the intermolecular condensation outcompetes uncontrolled polymerization, leading to a cleaner reaction profile.
Following the formation of the dipyrromethene intermediate, the introduction of the boron difluoride core is achieved through chelation with boron trifluoride (BF3) in the presence of a base like triethylamine. This step rigidifies the molecular structure, locking the dipyrromethene into a planar conformation that is responsible for the dye's high fluorescence quantum yield and narrow emission bandwidth. The rigidity minimizes non-radiative decay pathways, such as bond rotation, thereby maximizing photon emission. Furthermore, the presence of the carboxyl group at the meso-position (8-position) exerts an electronic influence that stabilizes the excited state, contributing to the observed photostability in both organic and aqueous environments. For R&D teams focused on high-purity fluorescent probes, understanding that the final purification involves standard silica gel chromatography and recrystallization confirms that the process is compatible with existing GMP infrastructure, ensuring that impurity levels can be tightly controlled to meet stringent analytical specifications.
How to Synthesize Carboxyl-BODIPY Dyes Efficiently
The synthesis protocol outlined in the patent provides a robust framework for producing these advanced fluorophores with high reproducibility. The process begins with the preparation of an absolutely anhydrous reaction environment, typically using solvents like tetrahydrofuran or acetonitrile that have been freshly distilled to remove trace moisture which could hydrolyze the anhydride prematurely. The stoichiometry is critical, with a molar ratio of pyrrole to anhydride ranging from 0.5:1 to 10:1, though a 2:1 ratio is often optimal to drive the double condensation to completion. Once the dipyrromethene intermediate is formed, indicated by a color change to light brown or red, the reaction mixture is cooled, and the boron trifluoride etherate is added dropwise under nitrogen protection. The subsequent workup involves extraction, drying, and column chromatography, yielding the final brick-red or orange-red powder. For detailed operational parameters, safety precautions, and specific stoichiometric calculations tailored to your facility's equipment, please refer to the standardized guide below.
- Condense two molecules of substituted pyrrole with one molecule of organic dibasic acid anhydride (e.g., succinic, glutaric, or phthalic anhydride) in an anhydrous organic solvent at 15-120°C to form the dipyrromethene intermediate.
- React the resulting dipyrromethene intermediate with a boron trifluoride compound (such as BF3-etherate) in the presence of an organic amine neutralizer like triethylamine at -10°C to 100°C to form the final fluorophore.
- Purify the crude product via silica gel column chromatography and recrystallization, optionally activating the carboxyl group with NHS/DCC for bioconjugation applications.
Commercial Advantages for Procurement and Supply Chain Teams
From a strategic sourcing perspective, the adoption of this anhydride-based synthesis route offers compelling advantages that extend beyond mere technical performance. For procurement managers evaluating cost reduction in fine chemical manufacturing, the substitution of expensive, custom-synthesized aldehydes with commodity acid anhydrides represents a direct material cost saving. Acid anhydrides like succinic and phthalic anhydride are produced on a massive industrial scale for the polymer and resin industries, ensuring a stable and competitive pricing structure that is immune to the volatility often seen in niche pharmaceutical intermediates. Furthermore, the reduction in synthetic steps from six down to essentially two major transformations significantly lowers the conversion costs associated with labor, energy, and reactor occupancy time. This efficiency gain allows suppliers to offer more competitive pricing tiers for bulk quantities, making high-performance bio-probes accessible for large-scale diagnostic kit production.
- Cost Reduction in Manufacturing: The elimination of multi-step protection and deprotection sequences, which are characteristic of traditional aldehyde routes, removes the need for expensive reagents and extensive purification between steps. By consolidating the synthesis into a one-pot condensation followed by a single chelation step, the overall process mass intensity (PMI) is drastically reduced. This means less solvent is consumed per kilogram of product, directly translating to lower waste disposal costs and a smaller environmental footprint. The higher yield of up to 25% compared to the 12% benchmark further amplifies these savings, effectively doubling the output from the same amount of raw material input without requiring additional capital investment in plant capacity.
- Enhanced Supply Chain Reliability: The reliance on widely available starting materials mitigates the risk of supply disruptions that can occur when depending on single-source specialty chemical vendors. Since the key precursors (pyrroles and anhydrides) are standard catalog items with multiple global suppliers, the supply chain becomes more resilient to geopolitical or logistical shocks. This redundancy ensures consistent lead times and enables manufacturers to maintain safety stock levels more effectively. For supply chain heads concerned with reducing lead time for high-purity fluorescent dyes, the simplified workflow means faster batch turnover, allowing for more responsive fulfillment of urgent orders and better alignment with just-in-time inventory models.
- Scalability and Environmental Compliance: The reaction conditions described are mild, operating at temperatures between 15°C and 120°C, which are easily manageable in standard stainless steel reactors without the need for specialized cryogenic or high-pressure equipment. This thermal flexibility facilitates seamless scale-up from laboratory grams to commercial metric tons, supporting the commercial scale-up of complex fluorescent intermediates. Additionally, the process avoids the use of highly toxic reagents often associated with acyl chloride chemistry, such as thionyl chloride, thereby reducing hazardous waste generation and simplifying regulatory compliance. The ability to recycle solvents like dichloromethane and acetonitrile further enhances the sustainability profile of the manufacturing process, aligning with corporate ESG goals.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation and application of these novel BODIPY dyes. These insights are derived directly from the experimental data and embodiments provided in the patent documentation, ensuring accuracy and relevance for potential partners.
Q: What are the primary advantages of the anhydride-based synthesis route over traditional aldehyde methods?
A: The anhydride-based route described in patent CN102061103A significantly simplifies the synthetic pathway by utilizing readily available acid anhydrides instead of complex aldehydes or acyl chlorides. This approach reduces the number of reaction steps, avoids harsh conditions associated with traditional methods, and achieves yields up to 25%, compared to as low as 12% in some literature precedents, thereby lowering overall production costs.
Q: How does the structural design of these dyes improve their performance in biological assays?
A: These dyes feature a carboxylic acid functional group at the 8-position, which prevents self-aggregation at low concentrations and ensures excellent solubility in aqueous media. They exhibit high fluorescence quantum yields (0.6-0.8), narrow emission bandwidths, and exceptional photostability, making them superior to pyrene or coumarin dyes for sensitive detection in complex biological matrices.
Q: Can these dyes be directly conjugated to proteins for labeling applications?
A: Yes, the carboxyl group on the dye scaffold can be easily activated using standard coupling agents like N-hydroxysuccinimide (NHS) and carbodiimides (DCC/DSC) to form reactive esters. These activated derivatives allow for efficient covalent labeling of amino groups on proteins such as BSA, enabling robust fluorescence immunoassays and cellular imaging without significant loss of protein activity.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable BODIPY Dye Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical role that high-quality fluorescent probes play in advancing life science research and diagnostic development. Our team of expert chemists has extensively analyzed the synthetic pathways described in patent CN102061103A and possesses the technical capability to replicate and optimize these processes for industrial production. We bring extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that your transition from R&D to market is seamless. Our state-of-the-art facilities are equipped with rigorous QC labs capable of verifying stringent purity specifications, including HPLC, NMR, and MS analysis, to guarantee that every batch meets the exacting standards required for biological applications. Whether you require custom derivatives with specific R-group modifications or bulk supply of the standard carboxyl-BODIPY scaffolds, our flexible manufacturing platform is designed to accommodate your unique needs.
We invite you to collaborate with us to leverage this cutting-edge technology for your next-generation bio-assays. By partnering with NINGBO INNO PHARMCHEM, you gain access to a Customized Cost-Saving Analysis that identifies specific opportunities to optimize your supply chain and reduce overall material costs. We encourage you to contact our technical procurement team today to request specific COA data for our available dye variants and to discuss route feasibility assessments for your proprietary conjugation projects. Let us help you illuminate your research with superior fluorescence technology.
