Revolutionizing Quinoxaline-3-one Production: A High-Selectivity Base-Catalyzed Route for Commercial Scale-up
The landscape of heterocyclic chemistry is constantly evolving, driven by the relentless demand for higher purity and specific regioisomers in drug discovery. Patent CN111018795A introduces a transformative methodology for the synthesis of quinoxaline-3-one derivatives, addressing a long-standing challenge in regioselective cyclization. This innovation utilizes a simple yet highly effective organic base catalysis system to convert 5-substituted o-phenylenediamines and alpha-ketoesters into the desired quinoxaline-3-one scaffold with exceptional precision. For R&D directors and process chemists, this represents a paradigm shift away from harsh, non-selective conditions toward a milder, more controlled synthetic environment. The ability to dictate the formation of the 3-one isomer over the thermodynamically favored 2-one isomer opens new avenues for generating diverse chemical libraries without the burden of complex separation processes. This technical breakthrough not only enhances the structural diversity available to medicinal chemists but also lays a robust foundation for industrial scalability.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of quinoxaline derivatives has been plagued by poor regioselectivity, particularly when asymmetric substrates are employed. Traditional condensation methods often rely on acidic conditions or high-energy inputs that fail to distinguish between the two amine groups on the phenylenediamine ring. For instance, earlier approaches utilizing microwave-assisted organic synthesis (MAOS) in methanol and acetic acid systems at 160°C resulted in a nearly statistical distribution of isomers, typically yielding a 1:1 ratio of regioisomers.
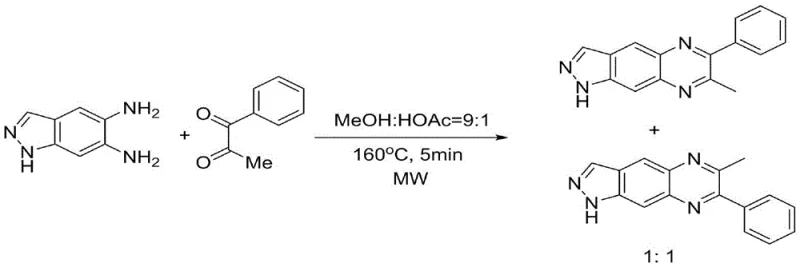
Furthermore, alternative strategies involving in-situ iodination and oxidation processes introduce unnecessary complexity and environmental hazards. These multi-step sequences require stoichiometric amounts of iodine and oxidants, generating significant waste streams and complicating the purification profile. The reliance on such aggressive reagents not only increases the cost of goods sold but also poses significant safety risks during scale-up. Additionally, methods employing alpha-imino esters mediated by aniline auxiliaries suffer from prolonged reaction times exceeding 24 hours and only moderate selectivity, rendering them inefficient for large-scale manufacturing. The cumulative effect of these limitations is a bottleneck in the supply chain, where low yields and difficult separations drive up costs and extend lead times for critical pharmaceutical intermediates.
The Novel Approach
In stark contrast, the methodology disclosed in CN111018795A leverages the subtle electronic differences between carbonyl groups through base catalysis to achieve unprecedented control. By reacting 5-substituted o-phenylenediamine with an ortho-dicarbonyl compound in the presence of an organic base, the process selectively generates the quinoxaline-3-one isomer as the major product. This approach eliminates the need for transition metals, strong acids, or auxiliary reagents, streamlining the reaction workflow significantly. The reaction operates under mild thermal conditions, typically between 20°C and 70°C, which drastically reduces energy consumption compared to traditional reflux or microwave protocols.
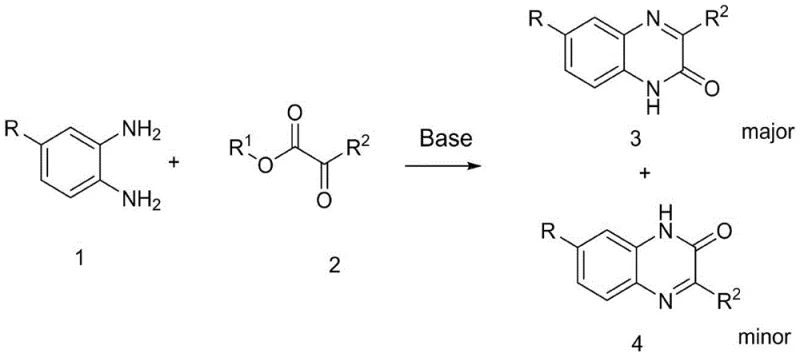
The simplicity of this novel route is its greatest asset; it utilizes readily available solvents like methanol or ethanol and common organic bases such as triethylamine or DIPEA. The absence of heavy metals means that the stringent purification steps required to meet ICH Q3D guidelines for elemental impurities are rendered unnecessary. This results in a cleaner crude product profile, facilitating easier isolation of the target compound. Moreover, the high regioselectivity, with ratios reaching up to 11:1 in favor of the 3-one isomer, ensures that the final API intermediate meets rigorous purity specifications with minimal material loss. This efficiency translates directly into improved process economics and a more reliable supply chain for downstream users.
Mechanistic Insights into Base-Catalyzed Regioselective Cyclization
The success of this synthesis lies in the nuanced interaction between the organic base and the electrophilic centers of the alpha-ketoester. Under alkaline conditions, the base likely facilitates the deprotonation of the more nucleophilic amine or activates the carbonyl species in a manner that favors attack at the ester-adjacent ketone, leading to the 3-one cyclization pathway. This stands in opposition to acid-catalyzed mechanisms where protonation patterns often favor the formation of the 2-one isomer. The structural integrity of the final product is confirmed by the distinct substitution patterns allowed on the benzene ring, where R groups can range from alkoxy and hydroxyl to halogens and carboxylates without compromising the reaction outcome.
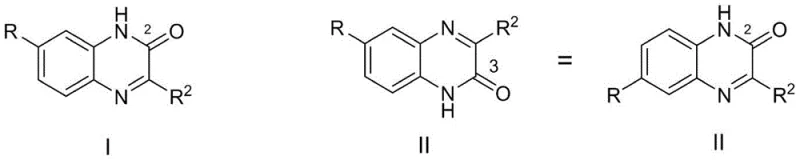
Understanding the impurity profile is critical for regulatory compliance, and this base-catalyzed route offers superior control over byproduct formation. The primary impurity is the regioisomeric quinoxaline-2-one, which is formed in minor quantities due to the kinetic preference established by the base. Unlike traditional methods that generate a complex soup of oligomers and decomposition products, this process yields a binary mixture that is easily separated via standard column chromatography or recrystallization. The ability to tune the selectivity by adjusting the molar ratio of the base, with optimal results observed at 3:1 to 5:1 equivalents, provides process chemists with a valuable handle for optimization. This level of mechanistic understanding ensures that the process is robust and reproducible, key factors for technology transfer from the laboratory to the pilot plant.
How to Synthesize Quinoxaline-3-one Efficiently
The operational simplicity of this protocol makes it highly attractive for both laboratory discovery and commercial production. The general procedure involves dissolving the substituted o-phenylenediamine in a polar protic solvent, followed by the addition of the organic base under an inert atmosphere. Once the mixture is equilibrated, the alpha-ketoester is introduced slowly to manage the exotherm, and the reaction is allowed to proceed at ambient or slightly elevated temperatures. Detailed standardized synthesis steps including specific stoichiometry, workup procedures, and purification parameters are outlined in the guide below.
- Dissolve 5-substituted o-phenylenediamine in a suitable solvent such as methanol or ethanol under stirring.
- Add an organic base catalyst like triethylamine or DIPEA, maintaining a molar ratio of 3: 1 to 5:1 relative to the diamine.
- Introduce the alpha-ketoester substrate slowly and stir at 20-70°C for 1-40 hours, followed by standard extraction and purification.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the adoption of this base-catalyzed synthesis offers tangible strategic benefits that extend beyond mere chemical elegance. The elimination of expensive and hazardous reagents such as iodine, heavy metal catalysts, or specialized microwave equipment significantly lowers the barrier to entry for production. This simplification of the bill of materials reduces dependency on volatile raw material markets and mitigates the risk of supply disruptions caused by the scarcity of niche reagents. Furthermore, the mild reaction conditions imply lower energy costs and reduced wear and tear on reactor vessels, contributing to a lower total cost of ownership for the manufacturing facility.
- Cost Reduction in Manufacturing: The most significant economic driver here is the removal of transition metal catalysts and the associated downstream processing costs. In traditional pharma manufacturing, removing trace metals to ppb levels requires specialized scavengers and extensive filtration, which adds substantial time and expense. By utilizing simple organic bases like triethylamine, this process bypasses those costly unit operations entirely. Additionally, the high regioselectivity minimizes the loss of valuable starting materials to isomeric byproducts, effectively increasing the overall yield of the usable intermediate. This efficiency gain directly impacts the margin, allowing for more competitive pricing in the global market for pharmaceutical intermediates.
- Enhanced Supply Chain Reliability: The raw materials required for this synthesis, including substituted phenylenediamines and alpha-ketoesters, are commodity chemicals with well-established global supply chains. Unlike proprietary catalysts or exotic ligands that may have single-source suppliers, these inputs are readily available from multiple vendors, ensuring continuity of supply. The robustness of the reaction conditions, which tolerate a range of temperatures and solvents, further enhances reliability by reducing the sensitivity to minor fluctuations in process parameters. This resilience is crucial for maintaining consistent delivery schedules to downstream API manufacturers, preventing costly production delays.
- Scalability and Environmental Compliance: From an environmental, health, and safety (EHS) perspective, this process is markedly superior to oxidative or microwave-assisted alternatives. The absence of heavy metals and halogenated oxidants simplifies waste treatment and disposal, aligning with increasingly stringent environmental regulations. The reaction can be safely scaled from gram to kilogram scales without the need for specialized pressure vessels or high-energy input devices. This ease of scale-up accelerates the timeline from clinical trial material to commercial launch, enabling faster time-to-market for new drug candidates. The greener profile of the synthesis also supports corporate sustainability goals, a key metric for modern supply chain management.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this synthesis route. These answers are derived directly from the experimental data and advantageous effects described in the patent documentation, providing clarity on the practical application of this technology.
Q: What is the primary advantage of this base-catalyzed method over traditional microwave synthesis?
A: Unlike traditional microwave methods which often yield a 1:1 mixture of regioisomers, this base-catalyzed approach achieves a regioselectivity ratio of up to 11:1 favoring the quinoxaline-3-one isomer, significantly simplifying downstream purification.
Q: Does this process require expensive transition metal catalysts?
A: No, the process relies solely on inexpensive organic bases such as triethylamine or DIPEA, eliminating the need for costly transition metals and the associated heavy metal removal steps required for pharmaceutical compliance.
Q: What represents the optimal reaction temperature for this synthesis?
A: The patent data indicates that the reaction proceeds efficiently under mild conditions ranging from 20°C to 70°C, allowing for significant energy savings compared to high-temperature reflux or microwave protocols.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Quinoxaline-3-one Supplier
At NINGBO INNO PHARMCHEM, we recognize that the successful commercialization of complex heterocycles requires more than just a patent; it demands deep process expertise and a commitment to quality. Our team of expert chemists has extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the transition from lab bench to reactor is seamless. We adhere to stringent purity specifications and operate rigorous QC labs to guarantee that every batch of quinoxaline-3-one intermediate meets the exacting standards required by the global pharmaceutical industry. Our infrastructure is designed to handle the specific nuances of base-catalyzed reactions, optimizing yield and consistency for our partners.
We invite you to leverage our technical capabilities to accelerate your drug development programs. By partnering with us, you gain access to a Customized Cost-Saving Analysis tailored to your specific volume requirements and purity needs. We encourage potential clients to contact our technical procurement team to request specific COA data and route feasibility assessments. Let us demonstrate how our advanced synthesis capabilities can enhance your supply chain efficiency and reduce your overall manufacturing costs.
