Scalable Synthesis of 4-Deoxy Daunorubicin Intermediates via Optimized Demethylation
Scalable Synthesis of 4-Deoxy Daunorubicin Intermediates via Optimized Demethylation
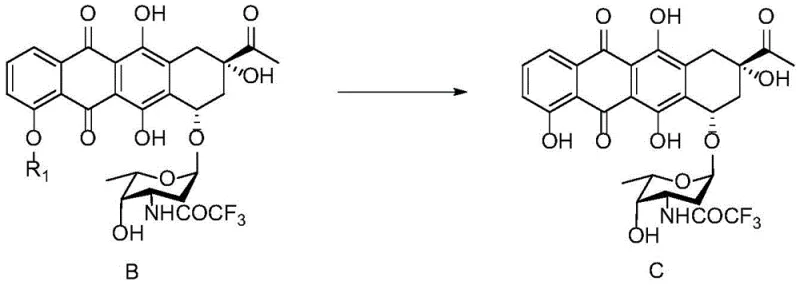
The pharmaceutical industry constantly seeks robust synthetic routes for complex anthracycline antibiotics, particularly for life-saving oncology treatments like 4-deoxy daunorubicin. A significant technological breakthrough in this domain is detailed in patent CN112010913A, which discloses a novel preparation method for key intermediates leading to 4-deoxy daunorubicin. This patent addresses long-standing challenges in the demethylation of protected anthracycline precursors, specifically the conversion of Compound B to Compound C. By leveraging a specialized catalytic system involving magnesium chloride, potassium iodide, and tetrabutylammonium iodide under strictly anhydrous conditions, the invention offers a pathway that drastically reduces salt consumption and suppresses the formation of critical impurities. For global procurement teams and R&D directors, this represents a pivotal shift towards more sustainable and high-yielding manufacturing processes for high-value anti-cancer agents.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Traditional synthetic routes for converting methoxy-substituted anthracyclines to their hydroxy counterparts often rely on harsh Lewis acids such as boron tribromide (BBr3) or aluminum chloride (AlCl3). These conventional reagents, while effective in small-scale laboratory settings, present severe limitations when translated to industrial production. The primary issue lies in the difficulty of controlling side reactions; harsh acidic conditions frequently lead to the degradation of the sensitive tetracyclic core and the sugar moiety. Furthermore, these methods typically generate substantial amounts of inorganic waste and require complex, hazardous workup procedures to quench excess reagents. In the context of 4-deoxy daunorubicin synthesis, traditional demethylation often results in the uncontrolled formation of structural analogs and degradation products, identified in the patent as Impurity I and Impurity II. These impurities not only lower the overall yield but also impose a heavy burden on downstream purification, increasing both production costs and environmental impact.
The Novel Approach
The methodology described in patent CN112010913A introduces a refined catalytic system that overcomes these historical bottlenecks. Instead of relying on aggressive, stoichiometric Lewis acids, the process utilizes a synergistic combination of anhydrous magnesium chloride (MgCl2), potassium iodide (KI), and tetrabutylammonium iodide (TBAI). This specific reagent cocktail facilitates a milder yet highly efficient demethylation of the methoxy group at the C-4 position of the anthracycline ring system. The innovation is not merely in the reagents themselves but in their precise molar ratios and the implementation of a fed-batch addition strategy. By carefully controlling the introduction of the catalyst mixture, the process maintains optimal reaction kinetics, preventing localized hotspots that could degrade the product. This approach ensures that the conversion of Compound B to Compound C proceeds with high selectivity, preserving the integrity of the adjacent functional groups and the glycosidic bond.
Mechanistic Insights into MgCl2-KI-TBAI Catalyzed Demethylation
The core of this technological advancement lies in the unique mechanistic role of the magnesium-halide complex. In this system, MgCl2 acts as a Lewis acid to coordinate with the oxygen atom of the methoxy group, thereby weakening the carbon-oxygen bond and making the methyl group more susceptible to nucleophilic attack. The presence of iodide ions (from KI and TBAI) serves as a potent nucleophile that displaces the methyl group, effectively cleaving the ether linkage to reveal the phenolic hydroxyl group. The tetrabutylammonium cation plays a crucial phase-transfer and solubilizing role, ensuring that the inorganic salts remain sufficiently dispersed in the organic solvent (typically tetrahydrofuran) to interact effectively with the substrate. This homogeneous-like environment enhances the reaction rate without requiring the extreme conditions associated with traditional methods. Furthermore, the patent highlights the critical importance of water control; the reaction is conducted in the presence of activated molecular sieves (3Å or 4Å) to maintain water content below 1%. This stringent dryness is essential because water can hydrolyze the reactive intermediates or deactivate the Lewis acid catalyst, leading to incomplete conversion and the proliferation of Impurity I and Impurity II.
Understanding the impurity profile is vital for R&D directors aiming for regulatory compliance. Impurity I and Impurity II are structurally related byproducts that likely arise from partial demethylation or rearrangement side reactions exacerbated by moisture or thermal stress. The patented process inhibits the increase of these impurities through two main mechanisms: first, by optimizing the molar ratio of MgCl2 to KI and TBAI (preferably around 1:1.7:1.6), the system ensures complete conversion without excess reagent that could promote side reactions. Second, the controlled temperature range of 50-60°C provides sufficient energy for the desired transformation while staying below the threshold where thermal degradation becomes significant. This precise balance allows for the production of Compound C with a purity profile that is far superior to batch reactions performed at higher temperatures or with less controlled reagent addition, directly translating to easier downstream processing and higher final API quality.
How to Synthesize Compound C Efficiently
The synthesis of Compound C from Compound B is a critical step in the manufacturing of 4-deoxy daunorubicin, requiring precise attention to reagent quality and process parameters. The patented method outlines a robust protocol that begins with the preparation of an anhydrous reaction environment using tetrahydrofuran and activated molecular sieves. The substrate, Compound B, is introduced along with the drying agent, and the system is purged with nitrogen to exclude atmospheric moisture. The catalyst mixture, comprising anhydrous MgCl2, KI, and TBAI, is then added in multiple batches rather than a single charge. This fed-batch technique is crucial for managing the reaction exotherm and maintaining consistent reagent concentration throughout the process. The reaction is typically maintained at 50-60°C for a duration of 11 to 15 hours, allowing for complete conversion as monitored by HPLC. Following the reaction, standard workup procedures involving filtration to remove salts and molecular sieves, followed by solvent exchange and crystallization, yield the high-purity Compound C ready for subsequent triflation and reduction steps.
- Prepare the reaction vessel with anhydrous THF, Compound B, and activated molecular sieves under nitrogen atmosphere.
- Add the catalyst mixture of anhydrous MgCl2, KI, and TBAI in multiple batches to control exotherm and reaction rate.
- Maintain reaction temperature between 50-60°C for 11-15 hours, monitoring conversion via HPLC to minimize impurity formation.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the adoption of this novel synthesis route offers tangible strategic benefits beyond mere technical elegance. The primary advantage lies in the significant optimization of raw material usage and waste generation. By reducing the overall amount of salt required in the reaction system compared to conventional excess reagent protocols, the process lowers the direct material cost per kilogram of product. Moreover, the suppression of Impurity I and Impurity II means that less material is lost during purification stages such as chromatography or recrystallization, effectively boosting the overall process yield. This efficiency gain is compounded by the simplified workup procedure; the use of soluble organic salts and the avoidance of aggressive aqueous quenches reduce the volume of wastewater generated, aligning with increasingly stringent environmental regulations and lowering disposal costs.
- Cost Reduction in Manufacturing: The elimination of expensive and hazardous traditional Lewis acids like BBr3 in favor of commodity chemicals like MgCl2 and KI results in a drastic reduction in raw material expenditure. Additionally, the improved selectivity minimizes the loss of high-value intermediates to side reactions, ensuring that a greater proportion of the starting material ends up as saleable product. The reduced need for extensive purification to remove difficult-to-separate impurities further decreases the operational costs associated with solvent usage and energy consumption during isolation.
- Enhanced Supply Chain Reliability: The reagents utilized in this process—magnesium chloride, potassium iodide, and tetrabutylammonium iodide—are widely available commodity chemicals with stable global supply chains. Unlike specialized or regulated reagents that may face sourcing bottlenecks, these materials can be procured reliably from multiple vendors, ensuring continuity of supply. The robustness of the reaction conditions also means that the process is less sensitive to minor variations in raw material quality, reducing the risk of batch failures and ensuring consistent delivery schedules to downstream API manufacturers.
- Scalability and Environmental Compliance: The fed-batch addition strategy and moderate temperature requirements make this process inherently scalable from pilot plant to commercial production scales without the need for specialized high-pressure or cryogenic equipment. The ability to control impurities at the source reduces the environmental footprint of the manufacturing process by minimizing the generation of hazardous waste streams. This alignment with green chemistry principles not only simplifies regulatory compliance but also enhances the corporate sustainability profile of the manufacturing entity, a key factor for modern pharmaceutical partnerships.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this demethylation technology. These insights are derived directly from the experimental data and embodiments provided in the patent documentation, offering a clear picture of the process capabilities and limitations. Understanding these details is essential for technical teams evaluating the feasibility of integrating this route into existing manufacturing lines or for procurement specialists assessing the long-term viability of the supply source.
Q: How does the new method control Impurity I and II compared to traditional methods?
A: The patented process utilizes a specific combination of MgCl2, KI, and TBAI with strict moisture control (molecular sieves), which significantly suppresses the formation of side-products like Impurity I and II that typically arise from hydrolysis or over-reaction in conventional Lewis acid demethylations.
Q: What are the critical reaction parameters for scaling up this demethylation?
A: Critical parameters include maintaining water content below 1% using molecular sieves, controlling the temperature strictly between 50-60°C, and employing a fed-batch addition strategy for the salt catalysts to prevent local overheating and ensure uniform reaction kinetics.
Q: Is this process suitable for multi-kilogram production?
A: Yes, the method is explicitly designed for scale-up. It reduces the total salt usage compared to prior art and mitigates yield loss during amplification, making it highly viable for commercial manufacturing of high-purity pharmaceutical intermediates.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 4-Deoxy Daunorubicin Intermediate Supplier
The advanced synthesis route for 4-deoxy daunorubicin intermediates described in patent CN112010913A exemplifies the kind of process innovation that drives value in the pharmaceutical supply chain. At NINGBO INNO PHARMCHEM, we specialize in translating such complex laboratory methodologies into robust, commercial-scale manufacturing operations. Our team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the theoretical benefits of this low-impurity, high-yield process are fully realized in large-volume batches. We maintain stringent purity specifications and operate rigorous QC labs equipped with state-of-the-art analytical instrumentation to guarantee that every batch of Compound C and subsequent intermediates meets the exacting standards required for oncology drug development.
We invite pharmaceutical companies and contract research organizations to collaborate with us to leverage this superior synthetic technology. By partnering with our technical procurement team, you can access a Customized Cost-Saving Analysis tailored to your specific volume requirements and quality targets. We encourage potential partners to contact us directly to request specific COA data for our anthracycline intermediates and to discuss detailed route feasibility assessments. Let us help you secure a reliable, cost-effective, and high-quality supply of 4-deoxy daunorubicin precursors for your critical drug development programs.
