Scalable Synthesis of 4-Deoxy Daunorubicin Intermediates via Optimized Demethylation Technology
Scalable Synthesis of 4-Deoxy Daunorubicin Intermediates via Optimized Demethylation Technology
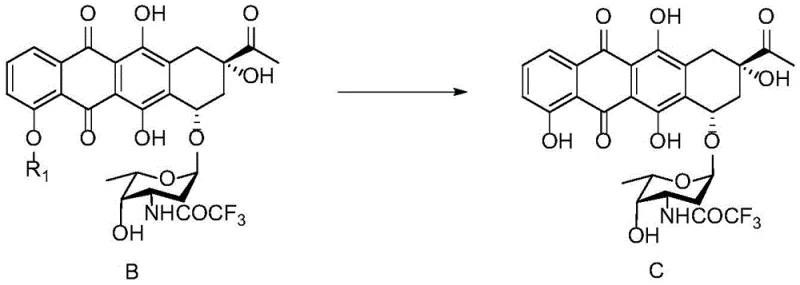
The pharmaceutical industry continuously seeks robust manufacturing routes for complex oncology agents, particularly anthracyclines used in treating hematological malignancies. Patent CN112010913B introduces a significant technological advancement in the preparation of 4-deoxy daunorubicin, a first-line therapy for relapsed and refractory adult Acute Myeloid Leukemia (AML). This patent details a novel synthetic pathway focusing on the critical conversion of Compound B to Compound C, a key intermediate in the anthracycline family. Unlike traditional methods that often struggle with scalability and impurity profiles, this invention leverages a specific combination of anhydrous magnesium salts and phase transfer catalysts to achieve superior reaction control. For R&D directors and procurement specialists, understanding this process is vital as it promises not only higher purity but also a more economically viable production model for high-value cancer therapeutics.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of anthracycline intermediates like Compound C has been plagued by significant challenges related to reaction amplification and impurity management. Conventional demethylation protocols often require excessive amounts of salt reagents to drive the reaction to completion, which inadvertently leads to complex workup procedures and increased waste generation. More critically, as these reactions are scaled from laboratory to industrial reactors, the heat transfer and mixing dynamics change, frequently causing a sharp decline in yield. Furthermore, traditional methods often fail to adequately suppress the formation of specific structural analogs, referred to in the patent as Impurity I and Impurity II. These impurities are structurally similar to the target molecule, making their removal via chromatography or crystallization both difficult and costly, thereby impacting the overall cost of goods sold (COGS) and extending the manufacturing lead time.
The Novel Approach
The methodology disclosed in CN112010913B represents a paradigm shift by optimizing the reagent stoichiometry and addition protocol. Instead of a single bulk addition of reagents, the novel approach employs a fed-batch strategy where a mixture of anhydrous magnesium chloride, potassium iodide, and tetrabutylammonium iodide is added in multiple portions (preferably 4 to 7 batches). This controlled addition maintains a steady state of reagent concentration, preventing exothermic runaways and ensuring uniform reaction kinetics throughout the vessel. Additionally, the process integrates strict moisture control, utilizing activated molecular sieves to keep water content below 1%, which is crucial for preventing hydrolysis side reactions. This refined approach not only reduces the total salt usage in the system but also effectively inhibits the generation of Impurity I and Impurity II, resulting in a cleaner crude product that requires less downstream purification.
Mechanistic Insights into MgCl2-Mediated Demethylation
The core chemical transformation in this patent is the selective demethylation of the methoxy group on the anthraquinone ring of Compound B to yield the phenolic hydroxyl group in Compound C. Mechanistically, anhydrous magnesium chloride acts as a mild yet effective Lewis acid, coordinating with the oxygen atom of the methoxy group to weaken the carbon-oxygen bond. The presence of potassium iodide (KI) and tetrabutylammonium iodide (TBAI) plays a synergistic role; the iodide ion serves as a potent nucleophile that attacks the methyl group, facilitating its removal as methyl iodide, while the quaternary ammonium salt acts as a phase transfer catalyst to enhance the solubility of the inorganic salts in the organic solvent, tetrahydrofuran. This specific reagent combination allows the reaction to proceed at moderate temperatures (50-60°C), avoiding the harsh conditions often required by stronger Lewis acids like boron tribromide, which can damage sensitive functional groups on the complex tetracyclic scaffold.
Impurity control is achieved through a deep understanding of the reaction environment's sensitivity to moisture and concentration gradients. Impurity I and Impurity II likely arise from over-reaction or hydrolytic degradation of the sensitive glycosidic or quinone moieties when water is present or when local concentrations of the demethylating agent are too high. By maintaining the water content within 1% using molecular sieves, the process eliminates the nucleophilic competition from water molecules. Furthermore, the fed-batch addition ensures that the concentration of the reactive magnesium species never exceeds the optimal threshold, thereby kinetically favoring the desired demethylation pathway over the degradation pathways that lead to impurities. This mechanistic precision ensures that the final API meets the stringent purity specifications required for oncology drugs.
How to Synthesize Compound C Efficiently
The synthesis of Compound C from Compound B is a critical unit operation in the manufacturing of 4-deoxy daunorubicin, requiring precise control over reaction parameters to ensure high yield and purity. The patented process outlines a specific sequence involving the preparation of anhydrous conditions, the precise weighing of magnesium chloride and iodide salts, and the implementation of a multi-stage feeding protocol. Operators must ensure that the reaction vessel is thoroughly dried and purged with nitrogen to maintain the inert atmosphere necessary for the stability of the anhydrous reagents. The detailed standardized synthetic steps, including specific stirring rates, temperature ramping profiles, and quenching procedures, are outlined in the technical guide below to assist process engineers in replicating this high-efficiency route.
- Charge anhydrous tetrahydrofuran and Compound B into a reaction vessel, optionally adding activated molecular sieves to control water content below 1%.
- Prepare a mixture of anhydrous magnesium chloride, potassium iodide, and tetrabutylammonium iodide for fed-batch addition.
- Heat the reaction mixture to 50-60°C and maintain for 11-15 hours while monitoring conversion to minimize Impurity I and II formation.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the adoption of this novel synthesis route offers tangible strategic benefits beyond mere technical feasibility. The primary advantage lies in the significant simplification of the supply chain for raw materials; by utilizing common inorganic salts like magnesium chloride and potassium iodide instead of exotic or hazardous reagents, the process reduces dependency on specialized chemical suppliers and mitigates the risk of raw material shortages. Moreover, the reduction in salt usage directly translates to lower waste disposal costs and simplified effluent treatment, aligning with increasingly strict environmental regulations in pharmaceutical manufacturing hubs. The robustness of the fed-batch process also means that production schedules are more predictable, with fewer batches rejected due to out-of-specification impurity profiles, thereby enhancing the overall reliability of the supply chain for this critical cancer medication.
- Cost Reduction in Manufacturing: The optimized stoichiometry and reduced salt consumption inherently lower the direct material costs associated with each batch. By minimizing the formation of difficult-to-remove impurities, the process drastically reduces the burden on downstream purification steps such as column chromatography or recrystallization, which are typically the most expensive phases of API manufacturing. This efficiency gain allows for a substantial reduction in the overall cost of goods, providing a competitive pricing advantage in the global market for oncology intermediates without compromising on quality standards.
- Enhanced Supply Chain Reliability: The use of stable, commercially available reagents like MgCl2 and TBAI ensures a secure supply base that is less susceptible to geopolitical or logistical disruptions compared to processes relying on custom-synthesized catalysts. The scalability of the fed-batch method means that production volume can be increased linearly without the need for extensive process re-validation, allowing suppliers to respond rapidly to surges in demand for 4-deoxy daunorubicin. This flexibility is crucial for maintaining continuous supply to pharmaceutical partners who require just-in-time delivery for their clinical and commercial programs.
- Scalability and Environmental Compliance: The process is explicitly designed for commercial scale-up, addressing the common pitfall where laboratory yields fail to translate to tonnage production. By controlling exotherms and impurity formation through fed-batch addition, the method ensures consistent quality from 100 kg to multi-ton scales. Furthermore, the reduction in chemical waste and the avoidance of harsh corrosive reagents contribute to a greener manufacturing footprint, helping pharmaceutical companies meet their sustainability goals and comply with rigorous environmental, health, and safety (EHS) audits.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this demethylation technology. These answers are derived directly from the experimental data and embodiments provided in the patent documentation, offering clarity on reaction conditions, impurity profiles, and scalability factors. Understanding these details is essential for technical teams evaluating the feasibility of integrating this route into their existing manufacturing infrastructure.
Q: How does the new process control impurities compared to conventional methods?
A: The process strictly controls water content within 1% using molecular sieves and employs a fed-batch addition strategy for reagents. This prevents local concentration spikes and hydrolysis, significantly inhibiting the formation of Impurity I and Impurity II.
Q: What are the key reagents used in this demethylation reaction?
A: The reaction utilizes anhydrous magnesium chloride (MgCl2) as the primary demethylating agent, assisted by potassium iodide (KI) and tetrabutylammonium iodide (TBAI) to enhance reactivity and solubility in tetrahydrofuran.
Q: Is this synthesis method suitable for large-scale commercial production?
A: Yes, the patent explicitly designs the method for scale-up by using a fed-batch mode (4-7 batches) which mitigates yield reduction often seen during reaction amplification, ensuring consistent quality from pilot to commercial scale.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 4-Deoxy Daunorubicin Intermediate Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical importance of high-quality intermediates in the development of life-saving oncology therapies. Our team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the transition from laboratory discovery to market availability is seamless and efficient. We adhere to stringent purity specifications and operate rigorous QC labs equipped with advanced analytical instrumentation to guarantee that every batch of 4-deoxy daunorubicin intermediate meets the highest international standards. Our commitment to technical excellence allows us to navigate the complexities of anthracycline synthesis, delivering products that support the global fight against leukemia.
We invite pharmaceutical partners to collaborate with us to leverage this advanced demethylation technology for your supply chain needs. By engaging with our technical procurement team, you can request a Customized Cost-Saving Analysis tailored to your specific volume requirements. We encourage you to reach out today to obtain specific COA data and route feasibility assessments, ensuring that your project benefits from the most efficient and reliable manufacturing solutions available in the industry.
