Revolutionizing Steroid Intermediate Production via Continuous Flow Tubular Reactor Technology
The pharmaceutical industry is constantly seeking robust methodologies to enhance the production efficiency of critical steroid intermediates, particularly those serving as precursors for life-saving medications like dexamethasone. Patent CN112608359B introduces a groundbreaking continuous flow synthesis strategy for preparing 17 alpha-hydroxyandrosta-4, 9-diene-3, 20-dione, a pivotal intermediate in the manufacture of corticosteroids used to treat severe allergies, asthma, and rheumatoid arthritis. This innovation marks a significant departure from traditional batch processing by implementing a continuous tubular reaction system that integrates Lewis acid catalysis and solid-phase deprotection. The technical breakthrough lies in its ability to automate process control, facilitate uninterrupted production cycles, and dramatically improve the stability of the final product quality. For global procurement teams and R&D directors, this patent represents a viable pathway to securing a reliable pharmaceutical intermediate supplier capable of meeting stringent purity specifications while optimizing operational expenditures through advanced engineering.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of 17 alpha-hydroxyandrosta-4, 9-diene-3, 20-dione has relied heavily on interval kettle-type reactions, which are plagued by inherent inefficiencies and scalability challenges. As illustrated in the prior art referenced by Chinese patent CN105440094B, the conventional route involves a cumbersome sequence of 3-ketocarbonyl protection, 17-hydroxyl protection, Grignard reaction, and subsequent deprotection steps. 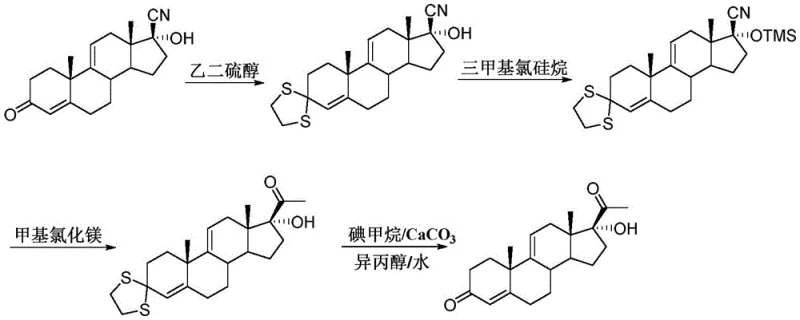 This multi-step batch process suffers from long production periods and low total yields due to the accumulation of losses at each isolation stage. Furthermore, the batch mode creates significant difficulties in controlling reaction parameters; the large volume of online material in kettle reactors leads to serious back-mixing, which promotes unwanted side reactions and results in unstable product quality. The poor reactivity of the cyano group in these traditional setups necessitates the use of excessive amounts of Grignard reagents to ensure complete conversion, thereby inflating raw material costs and complicating waste management protocols.
This multi-step batch process suffers from long production periods and low total yields due to the accumulation of losses at each isolation stage. Furthermore, the batch mode creates significant difficulties in controlling reaction parameters; the large volume of online material in kettle reactors leads to serious back-mixing, which promotes unwanted side reactions and results in unstable product quality. The poor reactivity of the cyano group in these traditional setups necessitates the use of excessive amounts of Grignard reagents to ensure complete conversion, thereby inflating raw material costs and complicating waste management protocols.
The Novel Approach
In stark contrast, the novel approach disclosed in CN112608359B leverages continuous flow chemistry to overcome these thermodynamic and kinetic barriers. The new process replaces the static kettle environment with a dynamic tubular reactor system where the ketal protector (I) and Grignard reagent are continuously pumped through a catalyst bed. 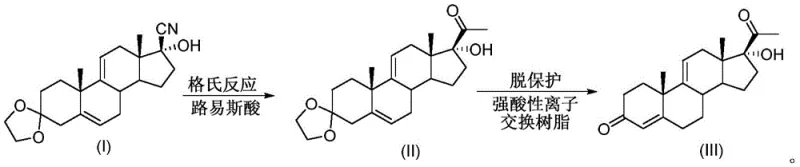 This transition to a plug flow regime ensures that reactants move through the system with minimal axial dispersion, effectively eliminating the back-mixing phenomena that degrade yield in batch reactors. The integration of a Lewis acid catalyst directly within the flow path activates the cyano group, allowing for precise stoichiometric control and a substantial reduction in reagent consumption. Additionally, the subsequent deprotection step is performed in a second tubular reactor packed with strong acid ion exchange resin, streamlining the workflow and enabling automatic control of the entire process. This modernization not only enhances safety by reducing the inventory of reactive intermediates but also paves the way for cost reduction in steroid manufacturing through improved atom economy and reduced solvent usage.
This transition to a plug flow regime ensures that reactants move through the system with minimal axial dispersion, effectively eliminating the back-mixing phenomena that degrade yield in batch reactors. The integration of a Lewis acid catalyst directly within the flow path activates the cyano group, allowing for precise stoichiometric control and a substantial reduction in reagent consumption. Additionally, the subsequent deprotection step is performed in a second tubular reactor packed with strong acid ion exchange resin, streamlining the workflow and enabling automatic control of the entire process. This modernization not only enhances safety by reducing the inventory of reactive intermediates but also paves the way for cost reduction in steroid manufacturing through improved atom economy and reduced solvent usage.
Mechanistic Insights into Lewis Acid-Catalyzed Continuous Grignard Reaction
The core mechanistic advantage of this patented process lies in the synergistic combination of Lewis acid catalysis and continuous flow dynamics within the tubular reactor R1. In traditional batch systems, the nucleophilic attack of the Grignard reagent on the nitrile group is often sluggish, requiring harsh conditions and large excesses of reagent. However, by packing the tubular reactor with Lewis acids such as magnesium chloride, lithium chloride, or lithium bromide, the electrophilicity of the cyano carbon is significantly enhanced. This activation lowers the energy barrier for the addition reaction, facilitating a rapid and selective transformation even at moderate temperatures ranging from 25°C to 110°C. The continuous flow environment further amplifies this effect by maintaining a high surface-area-to-volume ratio, which ensures efficient heat dissipation and prevents local hot spots that could lead to decomposition. Consequently, the reaction proceeds with high fidelity, minimizing the formation of by-products and ensuring that the intermediate ketone (II) is generated with exceptional purity before moving to the next stage.
Following the Grignard addition, the process employs a heterogeneous catalysis strategy for the deprotection step using strong acid ion exchange resin in tubular reactor R2. This mechanistic design is crucial for impurity control and environmental compliance. Unlike homogeneous acid deprotection which generates large volumes of acidic wastewater requiring neutralization, the solid-phase resin allows for a clean cleavage of the ketal protecting group. The plug flow nature of the reactor ensures that every molecule of the intermediate experiences the same residence time, typically between 10 minutes and 2 hours, leading to uniform deprotection. This consistency is vital for preventing over-reaction or incomplete hydrolysis, which are common sources of impurities in batch processes. The result is a crude product that requires minimal purification, often yielding white crystals with content exceeding 99% after simple recrystallization, thereby demonstrating the robustness of the catalytic system in maintaining high-purity pharmaceutical intermediate standards.
How to Synthesize 17 Alpha-Hydroxyandrosta-4, 9-Diene-3, 20-Dione Efficiently
The implementation of this continuous synthesis route requires precise engineering of the flow system to maximize the benefits of the tubular reactor design. Operators must first prepare the feed solutions by dissolving the ketal protector (I) in a suitable organic solvent such as tetrahydrofuran, toluene, or ethyl acetate and loading it into reservoir A, while the Grignard reagent is placed in reservoir B. These streams are then metered into a mixer and pumped into the catalyst-filled reactor R1, where the critical carbon-carbon bond formation occurs under controlled thermal conditions. The effluent is subsequently directed into reactor R2 for deprotection before undergoing a straightforward workup involving neutralization, extraction, and recrystallization. For a detailed breakdown of the specific operational parameters, residence times, and catalyst loading procedures, please refer to the standardized synthesis guide below.
- Dissolve ketal protector (I) in organic solvent (THF, toluene, etc.) and load into reservoir A; load Grignard reagent into reservoir B.
- Pump solutions into a mixer and feed into tubular reactor R1 filled with Lewis acid catalyst (e.g., MgCl2) at 25-110°C for Grignard reaction.
- Pass the effluent through tubular reactor R2 filled with strong acid ion exchange resin for deprotection, followed by neutralization and recrystallization.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the adoption of this continuous flow technology offers transformative benefits that extend far beyond simple yield improvements. The shift from batch to continuous processing fundamentally alters the cost structure of producing complex steroid intermediates by addressing key pain points related to raw material efficiency, operational safety, and production throughput. By eliminating the need for excessive Grignard reagents through Lewis acid catalysis, the process achieves significant cost reduction in pharmaceutical intermediates manufacturing without compromising on reaction completeness. Furthermore, the ability to run the reaction in a continuous manner rather than discrete batches allows for a dramatic increase in facility utilization rates, effectively decoupling production capacity from reactor volume constraints and ensuring a more reliable supply chain for downstream API manufacturers.
- Cost Reduction in Manufacturing: The economic impact of this technology is driven primarily by the optimization of reagent usage and the simplification of the workup process. In conventional methods, the poor activity of the cyano group forces manufacturers to use a large excess of expensive Grignard reagents, which not only increases material costs but also generates substantial waste upon quenching. The novel Lewis acid-catalyzed flow process drastically reduces the molar ratio of Grignard reagent required, leading to direct savings on raw material procurement. Additionally, the use of reusable solid acid catalysts in the deprotection step eliminates the recurring cost of purchasing liquid acids and the associated expenses of neutralizing and treating acidic wastewater. These cumulative efficiencies result in a leaner production model that substantially lowers the cost of goods sold (COGS) for high-value steroid intermediates.
- Enhanced Supply Chain Reliability: Supply chain continuity is often threatened by the variability inherent in batch processing, where slight deviations in mixing or temperature can ruin an entire batch, causing delays. The continuous tubular reactor system mitigates this risk by providing a highly stable and reproducible reaction environment. The plug flow characteristics ensure consistent product quality batch after batch, reducing the frequency of out-of-specification results that disrupt delivery schedules. Moreover, the compact footprint of tubular reactors allows for modular scaling; manufacturers can increase output by running the system for longer durations or numbering up reactors rather than building massive new facilities. This flexibility enables suppliers to respond more agilely to fluctuations in market demand, ensuring that clients receive their orders of high-purity pharmaceutical intermediates on time and without interruption.
- Scalability and Environmental Compliance: Scaling up chemical processes from the laboratory to commercial production is notoriously difficult, especially for exothermic reactions like Grignard additions. The continuous flow approach inherently solves the heat transfer limitations of large batch reactors, making the commercial scale-up of complex steroid intermediates safer and more predictable. The enhanced mass and heat transfer efficiency prevents thermal runaway, a critical safety feature when handling reactive organometallic reagents. From an environmental perspective, the process aligns with green chemistry principles by reducing solvent consumption and minimizing the generation of hazardous waste. The use of recyclable catalysts and the reduction in auxiliary chemicals for workup contribute to a lower environmental footprint, helping manufacturers meet increasingly stringent regulatory standards for emissions and waste disposal while maintaining high production volumes.
Frequently Asked Questions (FAQ)
Understanding the technical nuances of this continuous flow process is essential for stakeholders evaluating its potential for integration into their supply chains. The following questions address common inquiries regarding the operational parameters, catalyst longevity, and product quality assurance associated with Patent CN112608359B. These insights are derived directly from the experimental data and technical disclosures within the patent, providing a factual basis for assessing the feasibility of this advanced manufacturing technique for your specific application needs.
Q: How does the continuous flow method improve yield compared to traditional batch processes?
A: The continuous tubular reaction technology eliminates the back-mixing issues common in kettle-type reactors. By adopting a plug flow sample injection mode, mass and heat transfer efficiency is significantly improved, reducing side reactions and increasing the yield from approximately 78% in batch processes to over 90% in continuous flow.
Q: What catalysts are used in this novel Grignard reaction process?
A: The process utilizes Lewis acid catalysts such as magnesium chloride, lithium chloride, or lithium bromide packed within the tubular reactor. This catalytic approach activates the cyano group, allowing for a drastic reduction in the dosage of expensive Grignard reagents while maintaining high conversion rates.
Q: Is the ion exchange resin reusable in the deprotection step?
A: Yes, the strong acid ion exchange resin packed in tubular reactor R2 is designed for reuse. This solid-phase catalysis not only improves reaction efficiency but also simplifies the workup process by avoiding the generation of large amounts of acidic wastewater associated with liquid acid deprotection methods.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 17 Alpha-Hydroxyandrosta-4, 9-Diene-3, 20-Dione Supplier
At NINGBO INNO PHARMCHEM, we recognize that the transition to advanced continuous flow technologies requires a partner with deep technical expertise and proven industrial capability. As a leading CDMO, we possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the theoretical benefits of patents like CN112608359B are fully realized in practical manufacturing settings. Our state-of-the-art facilities are equipped with the necessary flow chemistry infrastructure and rigorous QC labs to maintain stringent purity specifications for critical steroid intermediates. We are committed to delivering products that meet the highest international standards, leveraging our technical proficiency to optimize reaction conditions and maximize yield for our global clientele.
We invite pharmaceutical companies and chemical distributors to collaborate with us to explore the commercial potential of this innovative synthesis route. By partnering with our technical procurement team, you can gain access to a Customized Cost-Saving Analysis tailored to your specific volume requirements. We encourage you to reach out today to request specific COA data and route feasibility assessments, allowing us to demonstrate how our continuous flow capabilities can enhance your supply chain resilience and drive down manufacturing costs for your key therapeutic programs.
