Revolutionizing Steroid Intermediate Manufacturing via Continuous Tubular Reactor Technology
The pharmaceutical industry is constantly seeking robust manufacturing solutions for critical steroid intermediates, particularly those serving as precursors for essential corticosteroids like dexamethasone. Patent CN112608359A discloses a groundbreaking method for preparing 17 alpha-hydroxyandrosta-4, 9-diene-3, 20-dione, shifting the paradigm from traditional batch processing to advanced continuous flow chemistry. This technological leap addresses long-standing inefficiencies in steroid synthesis by employing a continuous tubular reactor system filled with specific catalysts. The innovation lies not merely in the chemical transformation but in the engineering architecture that facilitates precise control over reaction parameters such as residence time and temperature. By integrating Lewis acid catalysis for the Grignard reaction and solid acid ion exchange resins for deprotection, this process achieves a level of operational stability and product consistency that batch methods struggle to match. For global supply chains, this represents a move towards more predictable and scalable production of high-value pharmaceutical intermediates.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the production of 17 alpha-hydroxyandrosta-4, 9-diene-3, 20-dione has relied heavily on interval kettle-type reactions, a method fraught with inherent inefficiencies and quality control challenges. As illustrated in the prior art routes, the synthesis often involves cumbersome multi-step protection and deprotection sequences that extend the production cycle and accumulate impurities. In a batch reactor environment, the large online material amount leads to serious back-mixing, creating hot spots and concentration gradients that foster unwanted side reactions. Furthermore, the cyano group in the starting material typically exhibits poor reactivity, necessitating a substantial excess of Grignard reagent to drive the reaction to completion, which inflates raw material costs and complicates waste disposal. The inability to precisely control heat dissipation in large kettles often results in unstable yields and variable product quality, making it difficult to meet the stringent purity specifications required for downstream drug synthesis.
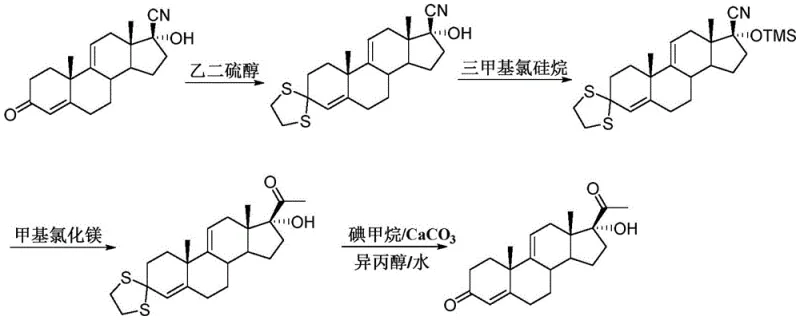
The Novel Approach
In stark contrast to these legacy methods, the novel approach detailed in the patent leverages continuous tubular reaction technology to fundamentally restructure the synthesis workflow. This method replaces the chaotic mixing of batch vessels with a disciplined plug flow sample injection mode, ensuring that every molecule experiences the same reaction history. The process utilizes a series of interconnected tubular reactors where the ketal protector and Grignard reagent are metered continuously into a mixer before entering a catalyst-filled tube. This setup allows for immediate and uniform heat transfer, effectively suppressing side reactions and enabling the use of milder conditions. The transition to a continuous flow system not only simplifies the operational complexity but also facilitates automatic control, which is crucial for maintaining the stability of the process over long production runs. This architectural shift is pivotal for manufacturers aiming to achieve cost reduction in pharmaceutical intermediates manufacturing while simultaneously enhancing throughput.
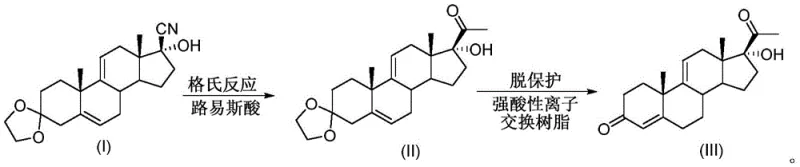
Mechanistic Insights into Lewis Acid-Catalyzed Continuous Grignard Reaction
The core chemical innovation in this process is the strategic application of Lewis acid catalysts within the confined geometry of a tubular reactor to activate the Grignard reaction. In traditional settings, the cyano group is notoriously sluggish, requiring harsh conditions or massive reagent excesses; however, the presence of catalysts such as magnesium chloride, lithium chloride, or lithium bromide alters the electronic environment of the substrate. These Lewis acids coordinate with the nitrogen atom of the cyano group, increasing its electrophilicity and making it more susceptible to nucleophilic attack by the Grignard reagent. Within the tubular reactor, this catalytic effect is amplified by the high surface-to-volume ratio, which ensures intimate contact between the solid catalyst and the flowing liquid stream. The result is a dramatic acceleration of the reaction rate, allowing for shorter residence times—ranging from 15 minutes to 2 hours—while maintaining high conversion efficiency. This mechanistic optimization is critical for commercial scale-up of complex pharmaceutical intermediates, as it decouples reaction speed from thermal runaway risks.
Furthermore, the integration of strong acid ion exchange resin for the subsequent deprotection step introduces a heterogeneous catalytic mechanism that streamlines purification. Unlike liquid acids which require neutralization and generate saline waste, the solid resin acts as a fixed bed through which the reaction mixture passes. The protons on the resin surface cleave the ketal protecting group efficiently, regenerating the ketone functionality without introducing soluble acidic contaminants into the product stream. This solid-phase catalysis not only improves reaction efficiency but also significantly reduces the generation of three wastes, aligning the process with modern green chemistry principles. The ability to reuse both the Lewis acid catalyst in the first reactor and the ion exchange resin in the second reactor adds a layer of economic sustainability, as the catalytic materials do not need to be replenished after every batch, thereby lowering the overall cost of goods sold.
How to Synthesize 17 alpha-hydroxyandrosta-4, 9-diene-3, 20-dione Efficiently
The synthesis protocol outlined in the patent provides a clear roadmap for implementing this continuous flow technology in an industrial setting. The procedure begins with the dissolution of the ketal protector in a suitable organic solvent such as tetrahydrofuran or toluene, which is then pumped alongside the Grignard reagent into the heated tubular reactor system. The precise control of flow rates determines the residence time, a critical parameter that must be optimized to balance conversion against potential degradation. Following the Grignard addition, the stream flows directly into the deprotection reactor, eliminating the need for intermediate isolation steps that often lead to yield losses. The detailed standardized synthesis steps, including specific temperature ranges and molar ratios, are essential for replicating the high purity and yield reported in the patent examples.
- Disperse ketal protector (I) in organic solvent and mix with Grignard reagent, feeding into a tubular reactor filled with Lewis acid catalyst.
- Pass the reacted mixture through a second tubular reactor packed with strong acid ion exchange resin for efficient deprotection.
- Perform post-treatment including neutralization, extraction, and recrystallization to isolate the high-purity target ketone.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain directors, the adoption of this continuous flow technology offers profound strategic advantages beyond mere technical novelty. The shift from batch to continuous processing inherently reduces the physical footprint of the manufacturing plant while increasing the volumetric productivity, allowing for greater output without proportional capital expenditure on new reactor vessels. The elimination of excessive Grignard reagent usage through Lewis acid catalysis directly translates to substantial raw material cost savings, as organometallic reagents are among the more expensive inputs in steroid synthesis. Moreover, the continuous nature of the process mitigates the risks associated with handling large quantities of reactive intermediates at once, enhancing overall plant safety and reducing insurance and compliance overheads. These factors combine to create a more resilient supply chain capable of responding rapidly to market demand fluctuations for critical steroid drugs.
- Cost Reduction in Manufacturing: The implementation of Lewis acid catalysis fundamentally alters the stoichiometry of the Grignard reaction, allowing for a significant reduction in the dosage of expensive Grignard reagents. By activating the cyano group, the process avoids the need for the massive excesses typically required in batch systems to ensure complete conversion. This optimization leads to direct savings on raw material procurement costs. Additionally, the reusability of the solid catalysts and ion exchange resins further drives down operational expenses, as the frequency of catalyst replacement is minimized. The reduction in solvent usage and energy consumption associated with shorter reaction times and eliminated intermediate workups also contributes to a leaner cost structure.
- Enhanced Supply Chain Reliability: Continuous flow systems are inherently more stable and easier to automate than batch processes, resulting in consistent product quality and reduced batch-to-batch variability. This reliability is crucial for maintaining uninterrupted supply lines to downstream API manufacturers who cannot afford delays due to failed batches. The plug flow nature of the reactor ensures that the product quality remains constant over time, reducing the need for extensive rework or rejection of off-spec material. Furthermore, the modular nature of tubular reactors allows for flexible capacity expansion; additional reactor tubes can be added in parallel to increase output without the long lead times associated with constructing new large-scale batch facilities.
- Scalability and Environmental Compliance: The transition to a continuous process using solid acid resins for deprotection drastically simplifies waste management protocols. By avoiding liquid mineral acids, the process eliminates the generation of large volumes of saline wastewater that require costly treatment before discharge. The high efficiency of the tubular reactors means that less solvent is required per unit of product, reducing the burden on solvent recovery systems and lowering volatile organic compound emissions. This alignment with environmental regulations not only prevents potential fines but also enhances the corporate sustainability profile, which is increasingly important for securing contracts with major multinational pharmaceutical companies committed to green supply chains.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this continuous flow synthesis method. Understanding these details is vital for technical teams evaluating the feasibility of adopting this technology for their own production lines. The answers are derived directly from the experimental data and process descriptions provided in the patent documentation, ensuring accuracy and relevance for decision-makers.
Q: How does the continuous tubular reactor improve yield compared to batch processes?
A: The continuous tubular reactor utilizes a plug flow mode which significantly reduces material back-mixing and improves mass and heat transfer efficiency. This controlled environment minimizes side reactions common in batch kettle systems, leading to higher conversion rates and product purity.
Q: What role does the Lewis acid catalyst play in the Grignard reaction?
A: The Lewis acid catalyst activates the cyano group, which typically exhibits poor reaction activity. By enhancing this activity, the process drastically reduces the required dosage of the expensive Grignard reagent, thereby lowering raw material costs and improving process safety.
Q: Why is strong acid ion exchange resin preferred for the deprotection step?
A: Using strong acid ion exchange resin in a fixed-bed tubular reactor allows for a heterogeneous catalytic process that is easily separable from the product stream. This eliminates the need for liquid acid neutralization steps, significantly reducing the generation of three wastes and simplifying downstream processing.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 17 alpha-hydroxyandrosta-4, 9-diene-3, 20-dione Supplier
At NINGBO INNO PHARMCHEM, we recognize the transformative potential of continuous flow chemistry in the production of high-value steroid intermediates. As a premier CDMO partner, we possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that innovative laboratory processes are successfully translated into robust industrial operations. Our facility is equipped with state-of-the-art continuous flow reactors and rigorous QC labs capable of meeting stringent purity specifications required by global regulatory bodies. We are committed to delivering high-purity pharmaceutical intermediates that adhere to the highest standards of quality and consistency, supporting our partners in bringing life-saving medications to market faster.
We invite you to collaborate with us to leverage these advanced manufacturing capabilities for your specific project needs. Our technical procurement team is ready to provide a Customized Cost-Saving Analysis tailored to your volume requirements, demonstrating how this continuous flow route can optimize your budget. Please contact us to request specific COA data and route feasibility assessments, and let us demonstrate how our expertise in process intensification can become a competitive advantage for your supply chain.
