Revolutionizing Thiocarbamate Manufacturing: A Safe, Metal-Free Photocatalytic Route for Global Supply Chains
The landscape of fine chemical manufacturing is undergoing a paradigm shift towards greener, safer, and more sustainable methodologies, a transition vividly exemplified by the technological breakthroughs detailed in patent CN108947880B. This pivotal intellectual property introduces a novel preparation method for thiocarbamate compounds based on visible light photocatalysis, addressing critical pain points that have long plagued the pharmaceutical and agrochemical industries. Traditionally, the synthesis of these vital structural motifs relied heavily on hazardous reagents and extreme conditions, creating significant bottlenecks for reliable thiocarbamate suppliers aiming to meet stringent global safety standards. The disclosed innovation leverages the power of visible light to drive the nucleophilic addition of thiols to isonitriles, utilizing environmentally benign organic dye catalysts instead of toxic heavy metals. By operating at room temperature and employing water as a co-solvent, this process not only enhances the safety profile of the manufacturing operation but also drastically simplifies the downstream purification workflows. For R&D directors and process chemists, this represents a transformative opportunity to redesign synthetic routes for high-purity pharmaceutical intermediates with improved atom economy and reduced environmental footprint.

Furthermore, the versatility of this photocatalytic system allows for a broad substrate scope, accommodating various substituted aryl, heteroaryl, and alkyl groups, which is essential for the diverse library synthesis required in modern drug discovery. The elimination of gaseous carbon monoxide and phosgene removes the need for specialized high-pressure equipment and rigorous gas handling protocols, thereby lowering the barrier to entry for commercial scale-up of complex thiocarbamates. As a leading entity in the fine chemical sector, understanding and adopting such cutting-edge methodologies is crucial for maintaining competitiveness in cost reduction in pharmaceutical intermediate manufacturing. The ability to produce these compounds under mild, metal-free conditions aligns perfectly with the increasing regulatory pressures regarding residual solvents and heavy metal impurities in active pharmaceutical ingredients. Consequently, this technology serves as a robust foundation for developing supply chains that are not only efficient but also resilient against regulatory changes and raw material volatility.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the industrial synthesis of thiocarbamate derivatives has been fraught with significant safety hazards and operational complexities that pose severe challenges for supply chain heads and procurement managers. The most prevalent traditional routes involve the use of phosgene, a notoriously toxic and corrosive gas, which reacts with amines to form amino acyl chlorides before subsequent substitution with thiophenols. Alternatively, other established methods utilize carbon monoxide gas in three-component reactions catalyzed by palladium or selenium, requiring high-pressure reactors and strict containment measures to prevent lethal leaks. These conventional approaches, as illustrated in older literature and prior art reaction schemes, inherently carry high risks of environmental pollution and acute human health hazards, necessitating expensive engineering controls and waste treatment facilities.
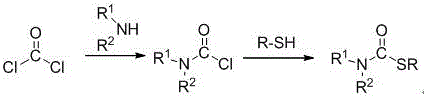
Beyond the immediate safety concerns, the reliance on transition metal catalysts in methods involving carbon monoxide introduces substantial downstream processing burdens. The removal of trace metal residues to meet the stringent purity specifications required for API intermediates often involves additional purification steps, such as scavenging resins or repeated recrystallizations, which inevitably lead to yield losses and increased production costs. Moreover, the use of stoichiometric amounts of bases or promoters in some older protocols generates large quantities of salt waste, complicating the environmental compliance landscape. For a procurement manager, these factors translate into higher raw material costs, longer lead times due to complex safety audits, and potential supply disruptions if regulatory bodies tighten restrictions on phosgene usage. The operational rigidity of these high-energy processes also limits the flexibility needed to respond quickly to market demands for diverse thiocarbamate analogs.
The Novel Approach
In stark contrast to these legacy methods, the photocatalytic strategy outlined in the patent data offers a streamlined, safe, and economically superior alternative that fundamentally reshapes the production logic. By utilizing visible light as the energy source and organic dyes like Rose Bengal as catalysts, the reaction proceeds under ambient pressure and near-room temperature conditions, effectively eliminating the need for high-energy inputs and specialized pressure vessels. This mild reaction environment not only ensures the safety of the operators but also preserves the integrity of sensitive functional groups that might decompose under the harsh thermal conditions of traditional synthesis. The substitution of toxic gases with stable solid or liquid reagents, specifically thiols and isonitriles, simplifies the logistics of raw material storage and handling, providing a distinct advantage in supply chain reliability.
From a commercial perspective, this novel approach significantly reduces the complexity of the manufacturing process, directly contributing to cost reduction in electronic chemical manufacturing and related sectors where purity is paramount. The absence of heavy metal catalysts means that the final product is inherently cleaner, reducing the burden on quality control laboratories and minimizing the risk of batch rejection due to metal contamination. Furthermore, the use of water as a co-solvent enhances the green chemistry profile of the process, aligning with corporate sustainability goals and potentially qualifying the manufacturing site for environmental incentives. This method empowers manufacturers to scale up production with greater confidence, knowing that the process is robust, safe, and adaptable to a wide range of substrates without requiring extensive re-optimization of reaction parameters.
Mechanistic Insights into Visible Light Photocatalytic Coupling
The core of this technological advancement lies in the intricate interplay between the photo-excited organic catalyst and the reactant molecules, a mechanism that offers profound implications for reaction selectivity and efficiency. Upon irradiation with visible light, typically from a blue or green LED source, the photocatalyst (such as Rose Bengal) absorbs photons and transitions to an excited state, possessing sufficient energy to initiate single-electron transfer (SET) processes or energy transfer pathways. In the context of thiocarbamate formation, the excited catalyst likely facilitates the generation of thiyl radicals from the thiol substrate or activates the isonitrile moiety through a radical addition mechanism. This radical-mediated pathway bypasses the high activation energy barriers associated with thermal nucleophilic attacks, allowing the reaction to proceed rapidly at room temperature. The presence of oxygen or air in the reaction headspace often plays a synergistic role, potentially acting as a terminal oxidant to regenerate the ground state of the catalyst, thus sustaining the catalytic cycle without the need for external chemical oxidants.
Understanding this mechanistic nuance is critical for R&D directors focused on impurity control and process optimization. The radical nature of the transformation suggests that side reactions involving radical dimerization or over-oxidation must be carefully managed through precise control of light intensity and reagent stoichiometry. However, the patent data indicates that the system is remarkably tolerant, yielding high-purity products with minimal byproduct formation across a diverse array of substrates. The use of a biphasic or mixed solvent system comprising an organic solvent like ethyl acetate and water further aids in stabilizing the reactive intermediates and facilitating the separation of the product post-reaction. This mechanistic elegance ensures that the process is not merely a laboratory curiosity but a viable industrial protocol capable of delivering consistent quality. By avoiding the formation of stable metal-ligand complexes that can trap intermediates, the organic photocatalyst ensures a clean turnover, which is essential for maintaining high throughput in a commercial setting.
How to Synthesize Thiocarbamate Efficiently
Implementing this photocatalytic protocol in a practical setting requires adherence to specific operational parameters to maximize yield and reproducibility. The general procedure involves the sequential addition of the thiol derivative, the isonitrile compound, and a catalytic amount of the organic dye into a reaction vessel equipped with a magnetic stirrer. A mixture of an organic solvent, such as ethyl acetate or acetonitrile, and water is then introduced to create the optimal reaction medium. The vessel is subsequently placed in a photoreactor illuminated by a visible light source, typically a 3W to 120W LED lamp, and stirred under air atmosphere for a duration ranging from 3 to 6 hours. Reaction progress is monitored via thin-layer chromatography (TLC), and upon completion, the mixture is subjected to a standard aqueous workup involving extraction with ethyl acetate. The combined organic layers are concentrated under reduced pressure, and the resulting crude residue is purified by flash column chromatography using a petroleum ether and ethyl acetate gradient to afford the target thiocarbamate in high purity.
- Combine thiophenol derivative, isonitrile compound, and a non-metal photocatalyst (e.g., Rose Bengal) in a reaction vessel containing an organic solvent and water mixture.
- Irradiate the reaction mixture with a visible light source (such as a 3W blue LED lamp) at room temperature while stirring in air for approximately 3 hours.
- Upon completion, extract the product with ethyl acetate, concentrate under vacuum, and purify the crude residue via silica gel column chromatography to obtain the pure thiocarbamate.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain strategists, the adoption of this photocatalytic methodology presents a compelling value proposition centered on risk mitigation and operational efficiency. The primary advantage lies in the drastic simplification of the safety infrastructure required for production. By eliminating the need for phosgene generators or high-pressure carbon monoxide lines, facilities can significantly reduce their capital expenditure on specialized containment systems and emergency response equipment. This reduction in overhead directly translates to lower manufacturing costs, allowing for more competitive pricing strategies in the global market. Additionally, the use of readily available, commodity-grade reagents like thiols and isonitriles ensures a stable supply base, reducing the vulnerability to shortages of exotic or highly regulated precursors. The robustness of the reaction conditions also implies a lower rate of batch failures, enhancing the overall reliability of supply for downstream customers.
- Cost Reduction in Manufacturing: The economic benefits of this process are multifaceted, stemming primarily from the elimination of expensive transition metal catalysts and the reduction of energy consumption. Traditional methods often require heating to elevated temperatures or the use of precious metal catalysts like palladium, which represent significant line items in the bill of materials. In contrast, the photocatalytic method utilizes inexpensive organic dyes and operates at room temperature, driven by energy-efficient LED lighting. Furthermore, the simplified workup procedure, which avoids complex metal scavenging steps, reduces the consumption of auxiliary chemicals and labor hours. These cumulative efficiencies result in a leaner manufacturing process that delivers substantial cost savings without compromising on the quality of the final intermediate.
- Enhanced Supply Chain Reliability: Supply chain continuity is often threatened by the regulatory scrutiny associated with hazardous chemicals. By shifting away from phosgene and carbon monoxide, manufacturers can navigate the regulatory landscape with greater ease, avoiding the delays and inspections that frequently accompany the transport and storage of toxic gases. The raw materials for this new method are stable solids or liquids that can be stored under standard conditions, simplifying inventory management and logistics. This stability ensures that production schedules can be maintained consistently, even in the face of external disruptions. Moreover, the scalability of the photochemical process means that capacity can be expanded by simply adding more light sources or reactors, rather than constructing massive high-pressure vessels, offering a flexible response to fluctuating market demand.
- Scalability and Environmental Compliance: As environmental regulations become increasingly stringent, the ability to demonstrate a green manufacturing process is a significant competitive advantage. This photocatalytic method generates minimal waste, particularly since it avoids the production of stoichiometric salt byproducts common in base-promoted reactions. The use of water as a co-solvent further reduces the volume of organic waste solvents that require disposal. From a scalability perspective, the modular nature of flow photochemistry or multi-batch parallel processing allows for seamless transition from pilot scale to full commercial production. This adaptability ensures that the supply of high-purity thiocarbamates can be ramped up quickly to meet the needs of large-scale API synthesis, all while maintaining a low environmental impact profile that satisfies corporate sustainability mandates.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this photocatalytic technology, derived from the specific advantages and operational details found in the patent literature. These insights are intended to clarify the feasibility of adopting this route for large-scale production and to highlight the specific benefits over legacy synthetic methods. Understanding these nuances is essential for technical teams evaluating the integration of this process into their existing manufacturing portfolios.
Q: Does this photocatalytic method require expensive transition metal catalysts?
A: No, the process utilizes inexpensive organic dyes such as Rose Bengal or Eosin B as photocatalysts, completely eliminating the need for costly palladium or selenium catalysts and avoiding heavy metal contamination in the final API intermediate.
Q: How does this method improve safety compared to traditional thiocarbamate synthesis?
A: Traditional methods often rely on highly toxic phosgene or carbon monoxide gas. This novel approach operates at room temperature using stable solid reagents (thiols and isonitriles), significantly reducing occupational health risks and environmental hazards.
Q: Is the reaction scalable for industrial production of agrochemical intermediates?
A: Yes, the reaction conditions are mild (room temperature, atmospheric pressure) and utilize water as a co-solvent, which simplifies heat management and waste treatment, making it highly suitable for commercial scale-up of complex thiocarbamates.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Thiocarbamate Supplier
The emergence of visible light photocatalysis as a viable industrial tool marks a significant milestone in the evolution of fine chemical synthesis, offering a pathway to safer and more sustainable manufacturing practices. NINGBO INNO PHARMCHEM stands at the forefront of this transformation, leveraging our extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production to bring these innovative methods to the global market. Our commitment to excellence is underpinned by stringent purity specifications and rigorous QC labs that ensure every batch of thiocarbamate intermediate meets the highest international standards. We understand that in the pharmaceutical and agrochemical industries, consistency and quality are non-negotiable, and our state-of-the-art facilities are designed to deliver exactly that, utilizing the latest green chemistry technologies to minimize environmental impact while maximizing output.
We invite potential partners to engage with our technical procurement team to discuss how this advanced photocatalytic route can be tailored to your specific project requirements. Whether you are looking for a Customized Cost-Saving Analysis for an existing product line or need to develop a new supply chain for a novel thiocarbamate derivative, we have the expertise to guide you. We encourage you to request specific COA data and route feasibility assessments to verify the superiority of this metal-free approach. By collaborating with us, you gain access to a supply chain that is not only cost-effective and reliable but also aligned with the future of sustainable chemical manufacturing, ensuring long-term success for your projects.
