Advanced Synthesis of 2-Methyl-8-Aminoquinoline via Redox-Coupled Cyclization for Commercial Scale-up
Advanced Synthesis of 2-Methyl-8-Aminoquinoline via Redox-Coupled Cyclization for Commercial Scale-up
The global demand for high-purity heterocyclic intermediates continues to surge, driven by the expanding pharmaceutical and agrochemical sectors. Patent CN102675201B introduces a transformative methodology for synthesizing 2-methyl-8-aminoquinoline, a critical building block for dyes and active pharmaceutical ingredients. Unlike legacy processes that struggle with isomer separation or rely on costly halogenated precursors, this invention leverages a clever redox-coupled cyclization strategy starting from readily available o-nitrophenol. By integrating the reduction of nitro groups directly into the oxidative ring-closing step, the process achieves superior atom economy and minimizes waste generation. For R&D directors and procurement strategists, this represents a significant opportunity to optimize supply chains for complex quinoline derivatives while adhering to stricter environmental compliance standards.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of 2-methyl-8-aminoquinoline has been plagued by significant selectivity and cost challenges. Traditional routes often begin with the direct nitration of 2-methylquinoline, a process that notoriously yields a complex mixture of 2-methyl-5-nitroquinoline and the desired 2-methyl-8-nitroquinoline isomers. As illustrated in the reaction scheme below, separating these structural analogs requires energy-intensive purification steps that drastically erode overall yield and increase production costs. Furthermore, alternative pathways involving halogenated intermediates, such as 2-methyl-8-bromoquinoline, necessitate the use of expensive palladium or tin reagents for subsequent amination, introducing heavy metal contamination risks that are unacceptable for modern pharmaceutical applications.
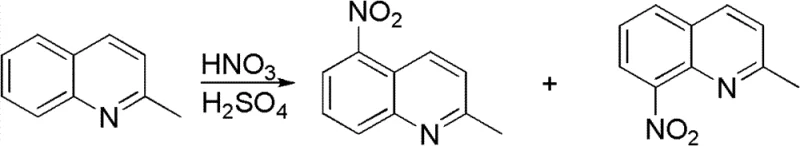
The Novel Approach
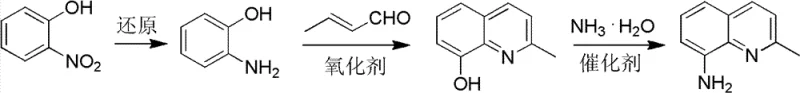
In stark contrast, the methodology disclosed in CN102675201B constructs the quinoline scaffold from the ground up using o-nitrophenol and crotonaldehyde. This approach completely bypasses the regioselectivity issues inherent in electrophilic aromatic substitution on pre-formed quinoline rings. The core innovation lies in a tandem sequence where o-nitrophenol serves a dual role: it acts as the primary carbon skeleton source and simultaneously functions as an internal oxidant to drive the dehydrogenative aromatization of the dihydroquinoline intermediate. This elegant design not only simplifies the operational workflow but also ensures that the amino group is introduced at the precise 8-position from the outset, delivering a cleaner crude product profile that facilitates downstream purification.
Mechanistic Insights into Redox-Coupled Cyclization
The chemical elegance of this process is best understood through its unique redox mechanism, which effectively recycles nitrogen-containing species within the reaction matrix. During the cyclization phase, the intermediate formed from o-aminophenol and crotonaldehyde undergoes oxidative dehydrogenation to achieve aromaticity. Crucially, the patent specifies that o-nitrophenol acts as the hydrogen acceptor in this step, getting reduced back into o-aminophenol in the process. This newly generated o-aminophenol then re-enters the condensation cycle with crotonaldehyde, creating a self-sustaining loop that amplifies the effective concentration of the reactive amine species. This mechanism explains the remarkably high yields reported, as it mitigates the loss of valuable amine intermediates to side reactions or polymerization.
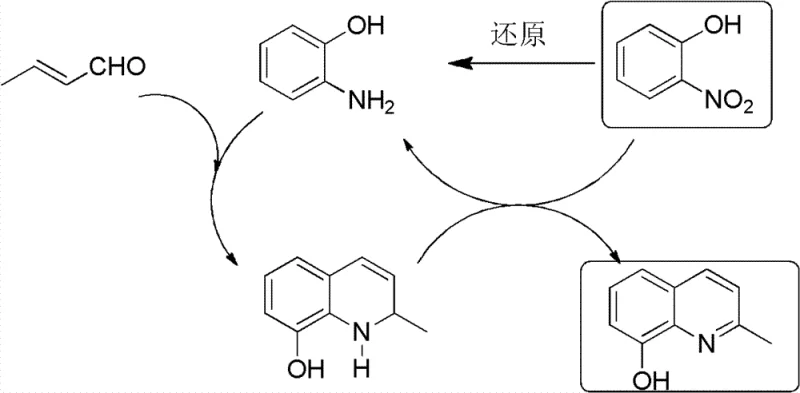
From an impurity control perspective, this mechanism offers distinct advantages over halogen-based routes. By avoiding the use of bromine or chlorine atoms on the aromatic ring prior to amination, the process eliminates the formation of dehalogenated byproducts and poly-aminated impurities that are common in nucleophilic aromatic substitution reactions. The final amination step utilizes a robust cobalt-chloride catalyst system under high-pressure ammonia conditions, which selectively displaces the hydroxyl group without affecting the methyl substituent or the quinoline nitrogen. This specificity ensures a narrow impurity profile, reducing the burden on quality control laboratories and enabling the production of material that meets stringent pharmacopeial standards with minimal recrystallization.
How to Synthesize 2-Methyl-8-Aminoquinoline Efficiently
The synthesis protocol outlined in the patent provides a clear roadmap for transitioning from laboratory benchtop to pilot plant operations. The procedure begins with the reduction of o-nitrophenol to o-aminophenol, followed by the key acid-catalyzed condensation with crotonaldehyde in the presence of the nitro-oxidant. The final transformation involves a high-temperature ammonolysis step that requires precise control of pressure and temperature to maximize conversion. While the chemical principles are straightforward, successful implementation relies on optimizing the molar ratios of the oxidant and managing the exothermic nature of the cyclization. For detailed operational parameters and safety guidelines, please refer to the standardized synthesis steps provided below.
- Reduce o-nitrophenol to o-aminophenol using catalytic hydrogenation or chemical reduction agents like sodium sulfide.
- Perform oxidative cyclization with crotonaldehyde and hydrochloric acid, utilizing o-nitrophenol as a recyclable oxidant to form 2-methyl-8-hydroxyquinoline.
- Execute high-temperature ammonolysis of the hydroxy-intermediate using ammonia water and a cobalt-based catalyst system to yield the final amino product.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain directors, the adoption of this synthetic route offers compelling economic and logistical benefits. The primary advantage stems from the substitution of expensive, specialized halogenated starting materials with commodity chemicals like o-nitrophenol and crotonaldehyde, which are available in bulk quantities from multiple global suppliers. This diversification of the raw material base significantly mitigates supply risk and protects against price volatility associated with niche intermediates. Additionally, the elimination of precious metal catalysts such as palladium, which are typically required for cross-coupling reactions in alternative routes, results in substantial cost savings and removes the need for complex heavy metal scavenging processes.
- Cost Reduction in Manufacturing: The integrated redox mechanism significantly lowers the consumption of oxidizing agents by recycling the nitro-group reduction product back into the reaction cycle. This intrinsic efficiency reduces the total mass of reagents required per kilogram of product, directly translating to lower variable manufacturing costs. Furthermore, the avoidance of chromatographic separation steps needed for isomer mixtures in traditional nitration routes streamlines the production workflow, reducing solvent usage and energy consumption associated with distillation and purification.
- Enhanced Supply Chain Reliability: By relying on o-nitrophenol as the foundational feedstock, manufacturers can leverage a well-established global supply chain for nitro-aromatics, ensuring consistent availability and shorter lead times. The robustness of the cobalt-catalyzed amination step also means that the process is less sensitive to minor fluctuations in reagent quality compared to sensitive palladium-catalyzed couplings, thereby reducing the frequency of batch failures and ensuring more predictable delivery schedules for downstream customers.
- Scalability and Environmental Compliance: The process is designed for scalability, utilizing standard industrial unit operations such as reflux condensers and high-pressure autoclaves that are common in fine chemical facilities. From an environmental standpoint, the "green" nature of the redox cycle minimizes the generation of nitro-containing waste streams, simplifying wastewater treatment protocols. This alignment with green chemistry principles facilitates easier regulatory approval and reduces the environmental compliance burden for manufacturing sites operating under strict emission limits.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this patented technology. These insights are derived directly from the experimental data and comparative analysis presented in CN102675201B, providing a factual basis for evaluating the feasibility of this route for your specific application requirements. Understanding these nuances is critical for making informed decisions about process adoption and vendor qualification.
Q: How does this patent's route improve upon traditional nitration methods?
A: Traditional nitration of 2-methylquinoline produces difficult-to-separate mixtures of 5-nitro and 8-nitro isomers. This patent avoids that by building the quinoline ring from o-nitrophenol, ensuring regioselectivity and higher purity without complex chromatographic separation.
Q: What are the cost advantages of using o-nitrophenol as an oxidant?
A: The process utilizes o-nitrophenol not just as a feedstock but as an internal oxidant that gets reduced to o-aminophenol during the reaction. This dual functionality reduces the need for external expensive oxidizing agents and maximizes atom economy, significantly lowering raw material costs.
Q: Is the high-pressure amination step scalable for industrial production?
A: Yes, the amination step operates at 300-320°C and 1.8-2.2 MPa, conditions that are standard for industrial autoclave reactors. The use of robust cobalt-based catalysts ensures the reaction is suitable for large-scale continuous or batch processing without requiring exotic equipment.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 2-Methyl-8-Aminoquinoline Supplier
At NINGBO INNO PHARMCHEM, we recognize the strategic value of efficient synthetic routes like the one described in CN102675201B for securing the supply of critical pharmaceutical intermediates. Our technical team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the transition from lab-scale optimization to full-scale manufacturing is seamless and risk-mitigated. We maintain stringent purity specifications and operate rigorous QC labs equipped with advanced analytical instrumentation to guarantee that every batch of 2-methyl-8-aminoquinoline meets the exacting standards required for drug substance synthesis.
We invite you to collaborate with us to leverage this advanced technology for your supply chain. Contact our technical procurement team today to request a Customized Cost-Saving Analysis tailored to your volume requirements. We are prepared to provide specific COA data and comprehensive route feasibility assessments to demonstrate how our manufacturing capabilities can enhance your operational efficiency and reduce your total cost of ownership.
