Advanced Manufacturing of 2-Methyl-8-Aminoquinoline via O-Nitrophenol Cyclization Technology
Advanced Manufacturing of 2-Methyl-8-Aminoquinoline via O-Nitrophenol Cyclization Technology
The global demand for high-purity heterocyclic intermediates continues to surge, driven by the expanding pharmaceutical and agrochemical sectors. A pivotal breakthrough in this domain is documented in patent CN102675201A, which introduces a revolutionary synthetic pathway for 2-methyl-8-aminoquinoline starting from the readily available feedstock o-nitrophenol. This technology represents a paradigm shift from traditional destructive functionalization methods to a constructive build-up strategy that inherently controls regioselectivity. By leveraging a unique redox mechanism where o-nitrophenol serves simultaneously as a substrate and an oxidant, the process achieves superior atom economy and minimizes waste generation. For R&D directors and procurement specialists, this innovation offers a compelling alternative to legacy routes that suffer from low yields and prohibitive purification costs. The following analysis dissects the technical merits and commercial viability of this approach, highlighting its potential to redefine supply chain standards for quinoline derivatives.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of 2-methyl-8-aminoquinoline has been plagued by significant structural challenges inherent to electrophilic aromatic substitution on the quinoline nucleus. The most common legacy approach involves the direct nitration of 2-methylquinoline, a reaction that notoriously lacks regioselectivity. As illustrated in the reaction scheme below, nitration typically yields a complex mixture of 2-methyl-5-nitroquinoline and the desired 2-methyl-8-nitroquinoline isomers.
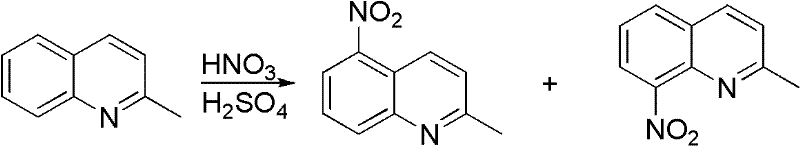
Separating these isomers is thermodynamically difficult and economically draining, often requiring energy-intensive fractional distillation or repeated recrystallization that drastically erodes overall process yield. Alternative routes, such as the nucleophilic substitution of 2-methyl-8-bromoquinoline with ammonia, introduce different bottlenecks. While chemically feasible, the preparation of the bromo-intermediate itself involves harsh conditions and expensive halogenating agents, creating a cost structure that is unsustainable for large-scale manufacturing. Furthermore, these multi-step sequences accumulate impurities at every stage, complicating the final purification and jeopardizing the stringent purity specifications required for pharmaceutical applications.
The Novel Approach
In stark contrast to these fragmented strategies, the methodology disclosed in CN102675201A constructs the quinoline skeleton de novo from o-nitrophenol, ensuring the correct substitution pattern is established from the outset. This constructive approach bypasses the isomer separation nightmare entirely by building the ring system around the pre-functionalized benzene precursor. The core innovation lies in a tandem cyclization-oxidation sequence where o-aminophenol reacts with crotonaldehyde in the presence of o-nitrophenol. This not only forms the heterocyclic ring but also aromatizes it in a single pot, streamlining the operation. The subsequent conversion of the resulting 2-methyl-8-hydroxyquinoline to the target amine via high-pressure ammonolysis is a robust, scalable transformation. This route transforms a complex separation problem into a straightforward synthesis challenge, offering a clear path to cost reduction in fine chemical manufacturing.
Mechanistic Insights into Redox-Mediated Cyclization
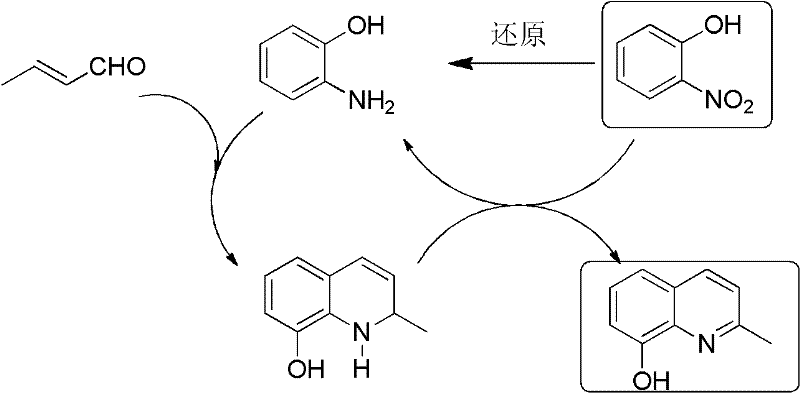
The heart of this technological advancement is the ingenious utilization of o-nitrophenol as a dual-function reagent during the ring-closing step. Mechanistically, the reaction begins with the condensation of o-aminophenol and crotonaldehyde to form an intermediate Schiff base or enamine species. Under acidic reflux conditions, this intermediate undergoes intramolecular cyclization. However, the resulting dihydroquinoline species requires oxidation to achieve aromatic stability. Traditionally, this would require the addition of stoichiometric external oxidants like nitrobenzene or metal salts, which generate substantial waste. In this novel system, added o-nitrophenol acts as the hydrogen acceptor. It oxidizes the dihydro-intermediate to 2-methyl-8-hydroxyquinoline while being reduced itself back to o-aminophenol.
This regenerated o-aminophenol then re-enters the cyclization cycle with more crotonaldehyde, creating a catalytic-like loop that significantly boosts the effective yield of the cyclization step. The detailed redox cycle is depicted in the schematic below, highlighting how the nitro group facilitates dehydrogenation without external reagents. This self-sustaining mechanism not only improves the mass balance of the process but also simplifies the workup procedure by reducing the load of inorganic salts in the waste stream. For process chemists, understanding this redox interplay is crucial for optimizing the molar ratios of o-nitrophenol to o-aminophenol, which the patent suggests should be maintained around 1:3 for optimal performance. This level of mechanistic control ensures consistent quality and reproducibility, key factors for reliable pharmaceutical intermediate supplier operations.
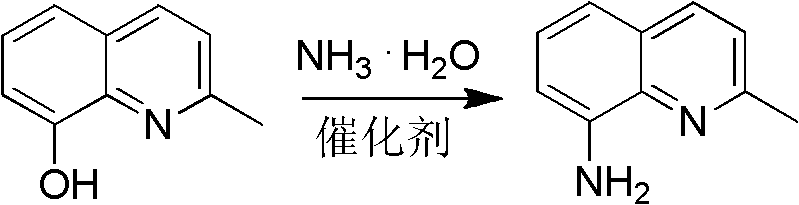
Furthermore, the final amination step employs a cobalt-based catalyst system, likely facilitating the nucleophilic attack of ammonia on the electron-deficient C-8 position of the quinoline ring. The use of ammonium chloride and cobalt chloride creates a Lewis acidic environment that activates the hydroxyl leaving group, allowing the substitution to proceed at elevated temperatures (300-320°C) and pressures. This high-energy activation is necessary to overcome the kinetic barrier of displacing the phenolic hydroxyl group, a transformation that is otherwise sluggish under ambient conditions. The result is a clean conversion to the primary amine with minimal formation of secondary or tertiary amine byproducts, ensuring a high-purity profile suitable for downstream drug synthesis.
How to Synthesize 2-Methyl-8-Aminoquinoline Efficiently
Implementing this synthesis route requires precise control over reaction parameters, particularly during the high-pressure ammonolysis phase. The process is divided into three distinct operational stages: the initial reduction of the nitro group, the oxidative cyclization with crotonaldehyde, and the final nucleophilic substitution. Each stage demands specific equipment capabilities, such as hydrogenation reactors for the first step and autoclaves capable of withstanding high temperatures and pressures for the last. The patent provides detailed embodiments that serve as a blueprint for scaling this chemistry from gram to kilogram quantities. Operators must pay close attention to the stoichiometry of the oxidant in the cyclization step to maximize the redox efficiency described earlier. For a comprehensive guide on the specific operational parameters, safety protocols, and workup procedures, please refer to the standardized synthesis instructions below.
- Reduce o-nitrophenol to o-aminophenol using catalytic hydrogenation or chemical reduction agents.
- Perform oxidative cyclization with crotonaldehyde, utilizing o-nitrophenol as an internal oxidant to form 2-methyl-8-hydroxyquinoline.
- Execute high-temperature ammonolysis of 2-methyl-8-hydroxyquinoline using ammonia water and a cobalt-based catalyst to yield the final amine.
Commercial Advantages for Procurement and Supply Chain Teams
From a strategic sourcing perspective, the adoption of this o-nitrophenol-based route offers profound advantages over legacy technologies, primarily driven by the simplification of the supply chain and the elimination of costly purification bottlenecks. Traditional methods relying on isomer separation create unpredictable yields and extended lead times, as the ratio of 5-nitro to 8-nitro products can fluctuate with minor changes in feedstock quality or reaction conditions. By switching to a constructive synthesis where the substitution pattern is predetermined by the starting material, manufacturers can guarantee consistent output rates. This predictability is invaluable for supply chain heads who need to manage inventory levels and meet just-in-time delivery schedules for API production. The ability to source commodity chemicals like o-nitrophenol and crotonaldehyde, rather than specialized halogenated quinolines, further insulates the production cost from volatility in niche reagent markets.
- Cost Reduction in Manufacturing: The most significant economic driver here is the removal of the isomer separation step. In conventional nitration routes, a substantial portion of the raw material ends up as the unwanted 5-nitro isomer, which is often discarded or requires expensive recycling processes. By avoiding this loss entirely, the effective cost per kilogram of the active intermediate is drastically lowered. Additionally, the internal redox cycle reduces the consumption of external oxidizing agents, cutting down on reagent costs and waste disposal fees. The streamlined workflow also means fewer unit operations, which translates to lower labor costs and reduced energy consumption per batch. These cumulative savings allow for a much more competitive pricing structure without compromising on margin.
- Enhanced Supply Chain Reliability: Relying on complex multi-step syntheses involving hazardous halogenation reagents introduces multiple points of failure in the supply chain. The new route utilizes robust, well-understood chemical transformations that are less sensitive to minor operational variances. The starting materials, o-nitrophenol and crotonaldehyde, are produced on a massive industrial scale globally, ensuring a stable and continuous supply even during market disruptions. This abundance of feedstock reduces the risk of production stoppages due to raw material shortages. Furthermore, the simplified purification process means that production cycles are shorter, allowing for faster turnaround times and increased flexibility to respond to sudden spikes in demand from downstream pharmaceutical clients.
- Scalability and Environmental Compliance: As regulatory pressure mounts regarding chemical waste and emissions, the greener profile of this synthesis becomes a major asset. The reduction in inorganic salt waste from avoiding external oxidants and the elimination of halogenated byproducts aligns perfectly with modern environmental, health, and safety (EHS) standards. The process is inherently scalable; the high-pressure amination step is a standard unit operation in the fine chemical industry, meaning that scaling from pilot plant to commercial production involves linear engineering rather than fundamental process redesign. This ease of scale-up ensures that capacity can be expanded rapidly to meet growing market needs for high-purity pharmaceutical intermediates without requiring massive capital expenditure on new, specialized infrastructure.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this patented technology. They are derived from the specific advantages and operational details outlined in the patent documentation, providing clarity for potential partners evaluating this synthesis route. Understanding these nuances is critical for making informed decisions about process adoption and supply agreements. We encourage technical teams to review these points carefully to assess the fit for their specific manufacturing requirements.
Q: Why is the o-nitrophenol route superior to direct nitration of 2-methylquinoline?
A: Direct nitration produces a difficult-to-separate mixture of 5-nitro and 8-nitro isomers, whereas the o-nitrophenol route constructs the specific substitution pattern directly, eliminating complex purification steps.
Q: What is the unique role of o-nitrophenol in the cyclization step?
A: In this novel process, o-nitrophenol acts dually as both a reactant precursor and an oxidizing agent, creating a redox cycle that improves atom economy and reaction yield significantly.
Q: Is this synthesis method suitable for large-scale industrial production?
A: Yes, the process utilizes commodity chemicals like crotonaldehyde and ammonia, and the reaction conditions, including high-pressure amination, are well-established in industrial organic synthesis settings.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 2-Methyl-8-Aminoquinoline Supplier
At NINGBO INNO PHARMCHEM, we recognize that the transition from laboratory innovation to commercial reality requires more than just a patent; it demands deep process engineering expertise and rigorous quality control. Our team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the theoretical benefits of the o-nitrophenol route are fully realized in practice. We have invested heavily in state-of-the-art high-pressure reactors and advanced analytical instrumentation to support the stringent purity specifications required for this intermediate. Our commitment to excellence means that every batch of 2-methyl-8-aminoquinoline is subjected to comprehensive testing in our rigorous QC labs, guaranteeing consistency and reliability for your downstream synthesis.
We invite you to collaborate with us to optimize your supply chain for quinoline derivatives. By leveraging this advanced synthesis technology, we can offer a Customized Cost-Saving Analysis tailored to your specific volume requirements. We encourage you to contact our technical procurement team to request specific COA data and route feasibility assessments. Let us demonstrate how our manufacturing capabilities can enhance your product competitiveness and secure your supply of this critical pharmaceutical intermediate.
Engineering Bottleneck?
Can't scale up this synthesis? Upload your target structure or CAS, and our CDMO team will evaluate the industrial feasibility within 24 hours. Request Evaluation →
