Scaling High-Purity Methylprednisolone Intermediates via Advanced Biological Dehydrogenation
Introduction to Next-Generation Steroid Synthesis
The production of high-value corticosteroids, such as Methylprednisolone, relies heavily on the efficiency and purity of key intermediate synthesis steps, particularly the 1,2-dehydrogenation of the steroid A-ring. This critical transformation introduces the conjugated enone system essential for the anti-inflammatory potency of the final drug. Traditional chemical methods have long struggled with toxicity and yield limitations, creating bottlenecks for reliable pharmaceutical intermediate suppliers aiming for green manufacturing standards. Patent CN101760495A presents a groundbreaking solution by leveraging the metabolic capabilities of Arthrobacter simplex to achieve this dehydrogenation biologically. This approach not only circumvents the use of hazardous reagents but also aligns with modern regulatory demands for cleaner synthetic routes. By shifting from stoichiometric chemical oxidants to biocatalysis, manufacturers can secure a more robust supply chain for complex steroid scaffolds.
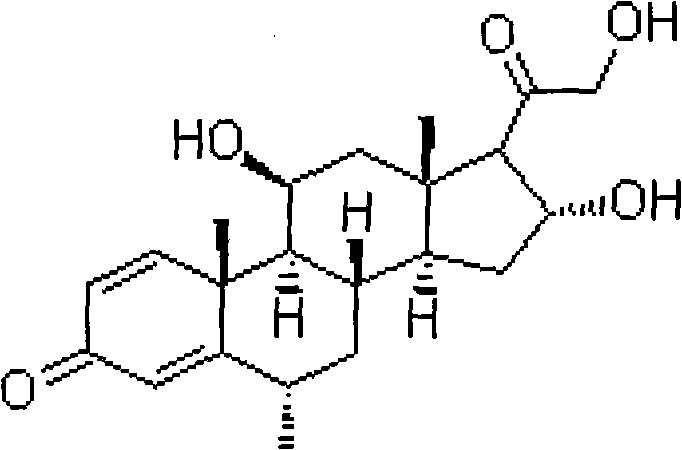
The strategic implementation of this biological pathway allows for the processing of diverse substrate analogues, including 6-alpha-methyl variants, which are pivotal for enhancing glucocorticoid activity while minimizing mineralocorticoid side effects. For R&D teams evaluating process scalability, this patent offers a validated framework that transitions seamlessly from laboratory shake flasks to industrial fermenters. The ability to utilize specific bacterial strains to perform regio-selective oxidation under mild conditions represents a significant leap forward in cost reduction in steroid manufacturing. As the industry moves towards sustainable chemistry, adopting such enzymatic or whole-cell catalytic processes becomes not just an option, but a necessity for maintaining competitiveness in the global API market.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the introduction of the 1,2-double bond in steroid intermediates has been predominantly achieved using Selenium Dioxide (SeO2) as the oxidizing agent. While chemically effective, this reagent poses severe challenges for large-scale production due to its high toxicity and environmental impact. The handling of SeO2 requires specialized containment and safety protocols, driving up operational costs and complicating waste management procedures. Moreover, the chemical dehydrogenation process typically suffers from modest yields, often hovering around 55%, which necessitates the processing of larger volumes of starting material to achieve target output. A critical quality concern is the difficulty in completely removing trace selenium residues from the final product, which can lead to failed batch releases due to strict heavy metal limits in pharmaceutical specifications. These factors combined create a fragile supply chain vulnerable to regulatory scrutiny and raw material volatility.
The Novel Approach
In stark contrast, the novel approach detailed in the patent utilizes a whole-cell biocatalytic system driven by Arthrobacter simplex. This biological method operates under mild physiological conditions, typically between 30°C and 34°C, eliminating the need for harsh chemical oxidants and extreme temperatures. The biotransformation exhibits remarkable specificity, targeting the 1,2-position of the steroid nucleus without affecting other sensitive functional groups such as hydroxyls or ketones elsewhere in the molecule. Most importantly, the conversion efficiency is substantially improved, with reported rates ranging from 70% to 90%, representing a dramatic increase in material throughput. By replacing a toxic chemical step with a fermentation process, the technology inherently simplifies the downstream purification workflow, as there is no need for complex heavy metal scavenging steps. This shift not only enhances the safety profile of the manufacturing site but also significantly improves the overall economic viability of producing high-purity corticosteroid intermediates.
Mechanistic Insights into Arthrobacter Simplex Biocatalysis
The core of this technological advancement lies in the specific enzymatic machinery of Arthrobacter simplex, which possesses steroid 1,2-dehydrogenase activity. When cultured in a nutrient-rich medium containing glucose, yeast extract, and corn steep liquor, the bacteria express the necessary enzymes to abstract hydrogen atoms from the C1 and C2 positions of the steroid substrate. The patent highlights the importance of strain selection, specifically citing strains AS 1.754 and AS 1.94*, which have been optimized for high transformation efficiency. The mechanism involves the transfer of electrons from the substrate to the bacterial electron transport chain, ultimately reducing oxygen or other electron acceptors in the medium. This bio-oxidation is highly stereoselective, ensuring that the resulting double bond maintains the correct geometry required for subsequent synthetic steps. Understanding this biological catalyst's behavior is crucial for R&D directors aiming to replicate these results, as factors like dissolved oxygen levels and pH (maintained at 7.0-7.2) play pivotal roles in enzyme activity.
Furthermore, the process demonstrates exceptional versatility regarding substrate tolerance. The biological system is capable of accepting a wide array of steroid precursors, including those with varying substituents at the C6, C11, C16, and C17 positions. Whether the substrate is a 11-beta-hydroxy compound or a 16-alpha,17-alpha-epoxy derivative, the Arthrobacter strain effectively performs the dehydrogenation. This broad substrate scope is a massive advantage for process chemists who need to synthesize multiple analogues within the same facility without changing the core production infrastructure. The impurity profile generated by this biological route is distinctly different from chemical oxidation, primarily consisting of unreacted starting material and minor metabolites rather than toxic heavy metal complexes. This cleaner impurity profile simplifies the crystallization and purification stages, directly contributing to higher final yields and reduced solvent consumption during workup.
How to Synthesize 6 Alpha-Methylprednisolone Intermediate Efficiently
Implementing this biological dehydrogenation process requires precise control over fermentation parameters to maximize the conversion of Formula 1 compounds into the desired Formula 2 dehydrogenated products. The protocol begins with the activation of the bacterial culture through a multi-stage seed train, ensuring a high density of active cells before the substrate is introduced. The substrate, often dissolved in a co-solvent like ethanol or DMF to improve solubility in the aqueous fermentation broth, is fed into the reactor at a controlled concentration, typically between 1% and 3% w/v. Maintaining the temperature within the narrow window of 30-34°C is critical, as deviations can inhibit enzyme activity or promote the growth of contaminating microbes. Detailed standardized synthesis steps for replicating this high-efficiency transformation are provided in the guide below.
- Prepare the Arthrobacter simplex strain (e.g., AS 1.94*) through slant culture, primary seed cultivation, and secondary seed cultivation in a glucose and yeast extract-based medium at 30-34°C.
- Introduce the steroid substrate (Formula 1) into the fermentation tank containing the cultivated bacteria, maintaining a substrate concentration of 1-3% and a temperature of 30-34°C for 36-72 hours.
- Terminate the reaction by heating the fermented liquid to 70-90°C to deactivate the bacteria, followed by solvent extraction (e.g., ethyl acetate) and purification to isolate the dehydrogenated product (Formula 2).
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the transition to this biological manufacturing route offers compelling strategic benefits that extend beyond simple yield improvements. The elimination of Selenium Dioxide removes a significant hazardous material from the supply chain, reducing the regulatory burden and insurance costs associated with storing and transporting toxic oxidants. This shift also mitigates the risk of supply disruptions caused by environmental crackdowns on chemical plants handling heavy metals. By adopting a fermentation-based process, manufacturers can leverage existing biotech infrastructure, which is often more scalable and flexible than specialized chemical reactors. The ability to produce intermediates with a cleaner impurity profile reduces the load on quality control laboratories and shortens the release time for batches, enhancing overall supply chain agility.
- Cost Reduction in Manufacturing: The biological route fundamentally alters the cost structure by eliminating the expensive and complex steps required to remove selenium residues. In traditional chemical synthesis, significant resources are allocated to activated carbon treatments and filtration processes to meet heavy metal specifications; this bio-method renders those steps obsolete. Additionally, the higher conversion efficiency means that less starting material is wasted, directly lowering the cost of goods sold (COGS). The reduction in hazardous waste disposal fees further contributes to substantial operational savings, making the final intermediate more price-competitive in the global market without compromising on quality standards.
- Enhanced Supply Chain Reliability: Relying on biological fermentation decouples production from the volatility of specialty chemical reagent markets. The nutrients required for the Arthrobacter culture, such as glucose and corn steep liquor, are commodity chemicals with stable and abundant global supplies. This ensures that production schedules are not held hostage by the availability of niche oxidants like SeO2. Furthermore, the robustness of the bacterial strains allows for consistent batch-to-batch performance, reducing the incidence of failed runs that can disrupt delivery timelines. This reliability is crucial for maintaining continuous API production lines and meeting the rigorous Just-In-Time delivery expectations of multinational pharmaceutical clients.
- Scalability and Environmental Compliance: Fermentation processes are inherently scalable, allowing for a smooth transition from pilot-scale 5L reactors to multi-ton industrial fermenters without significant re-engineering. This scalability supports the commercial scale-up of complex pharmaceutical intermediates to meet surging market demand. From an environmental perspective, the process generates significantly less toxic waste, aligning with increasingly stringent global environmental regulations. The absence of heavy metal effluent simplifies wastewater treatment requirements, reducing the environmental footprint of the manufacturing site and enhancing the company's sustainability profile, which is a key metric for modern ESG-conscious investors and partners.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this biological dehydrogenation technology. These insights are derived directly from the experimental data and claims presented in the patent literature, providing a factual basis for decision-making. Understanding these nuances helps stakeholders evaluate the feasibility of integrating this route into their existing manufacturing portfolios. The answers reflect the specific advantages of using Arthrobacter simplex over traditional chemical methodologies.
Q: What are the advantages of using Arthrobacter simplex over Selenium Dioxide for dehydrogenation?
A: The biological method using Arthrobacter simplex eliminates the use of highly toxic Selenium Dioxide (SeO2), thereby removing the risk of heavy metal residues in the final API. Furthermore, it significantly improves conversion rates from approximately 55% (chemical method) to 70-90% (biological method), reducing raw material waste.
Q: Which specific bacterial strains are recommended for this synthesis?
A: The patent specifically recommends using Arthrobacter simplex strains, particularly AS 1.754 and AS 1.94* (provided by the Institute of Microorganism, Academia Sinica), which have demonstrated high transformation efficiency for various steroid substrates.
Q: How is the reaction terminated and the product isolated?
A: The biotransformation is terminated by heating the fermentation broth to 70-90°C to deactivate the microbial cells. The product is then extracted using organic solvents such as ethyl acetate, followed by concentration and refining to obtain the high-purity dehydrogenated steroid.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Methylprednisolone Intermediate Supplier
At NINGBO INNO PHARMCHEM, we recognize the transformative potential of biological dehydrogenation in the synthesis of high-value steroid intermediates. As a leading CDMO partner, we possess the technical expertise to adapt and optimize such advanced fermentation pathways for industrial application. Our facilities are equipped to handle complex biocatalytic processes, ensuring that the transition from lab-scale innovation to commercial reality is seamless and efficient. We bring extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, guaranteeing that your supply needs are met with consistency and precision. Our commitment to quality is underscored by our stringent purity specifications and rigorous QC labs, which ensure that every batch of intermediate meets the highest international pharmacopoeia standards.
We invite procurement leaders and R&D directors to collaborate with us on optimizing their steroid synthesis routes. By leveraging our expertise in biocatalysis, we can help you achieve significant process improvements and cost efficiencies. We encourage you to contact our technical procurement team to request a Customized Cost-Saving Analysis tailored to your specific project requirements. Our team is ready to provide specific COA data and route feasibility assessments to demonstrate how our capabilities can enhance your supply chain resilience and product quality.
Engineering Bottleneck?
Can't scale up this synthesis? Upload your target structure or CAS, and our CDMO team will evaluate the industrial feasibility within 24 hours. Request Evaluation →
