Scalable Metal-Free Synthesis of Aromatic Amines for Pharmaceutical Intermediates
The pharmaceutical and fine chemical industries are constantly seeking more efficient, sustainable, and cost-effective pathways to synthesize critical building blocks. Patent CN109134267B introduces a groundbreaking methodology for the synthesis of aromatic amine compounds, addressing long-standing challenges in organic synthesis. This technology enables the direct conversion of bulk alkyl aromatic compounds or aromatic alcohol derivatives into valuable aromatic amines without the reliance on transition metal catalysts. For R&D directors and procurement managers alike, this represents a paradigm shift away from traditional, waste-intensive processes towards a cleaner, more streamlined manufacturing protocol. The ability to utilize readily available starting materials while achieving high selectivity positions this technology as a cornerstone for next-generation API intermediate production.
Aromatic amines serve as indispensable precursors in the manufacture of local anesthetics like procaine and benzocaine, as well as polyurethane raw materials such as diphenylmethane diisocyanate (MID). The global demand for these compounds exceeds millions of tons annually, yet conventional synthesis methods often suffer from significant drawbacks. Traditional routes typically involve multi-step nitration followed by reduction, or cross-coupling reactions requiring precious metal catalysts. These legacy processes generate substantial hazardous waste, incur high purification costs to remove metal residues, and often require harsh reaction conditions that limit scalability. The innovation disclosed in CN109134267B circumvents these issues by employing a direct amination strategy that is both operationally simple and environmentally superior.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the industrial preparation of substituted anilines has relied heavily on the nitration of aromatic rings followed by reduction, a sequence that poses severe safety and environmental hazards. The use of mixed acids for nitration generates large volumes of acidic wastewater, while the subsequent reduction step often necessitates high-pressure hydrogenation or stoichiometric amounts of reducing agents like iron powder. Alternatively, modern cross-coupling strategies, such as Buchwald-Hartwig amination, depend on palladium or copper catalysts. While effective, these methods introduce the risk of heavy metal contamination in the final active pharmaceutical ingredient, necessitating expensive scavenging steps and rigorous quality control testing. Furthermore, the substrate scope for these coupling reactions can be limited by steric hindrance or the availability of specific halogenated precursors, which are often more costly than their alkyl counterparts.
The Novel Approach
In stark contrast, the novel approach detailed in the patent utilizes a metal-free catalytic system that leverages oxidative cleavage or acid-mediated substitution to install the amino group directly. Method One employs an oxidant, such as 2,3-dichloro-5,6-dicyano-1,4-benzoquinone (DDQ), to facilitate the oxidative cleavage of the benzylic carbon-carbon bond in alkyl aromatics in the presence of an azide source. Method Two utilizes acid additives like trifluoroacetic acid to activate aromatic alcohol derivatives for nucleophilic substitution. This dual-pathway flexibility allows manufacturers to select the most economically viable starting material for their specific target molecule. By eliminating the need for transition metals, the process drastically simplifies downstream processing and ensures a cleaner impurity profile, which is critical for regulatory compliance in pharmaceutical manufacturing.
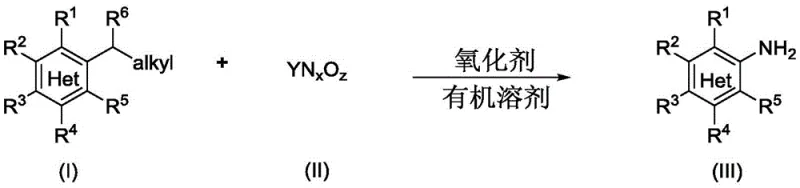
The versatility of this synthetic strategy is further evidenced by its compatibility with a broad spectrum of functional groups and heterocyclic systems. As illustrated in the reaction schemes, the methodology tolerates substituents such as halogens, ethers, esters, and even sensitive groups like nitro and cylohexyl moieties. This robustness means that complex intermediates can be synthesized without extensive protecting group manipulations, thereby reducing the overall step count and improving the cumulative yield of the synthesis. For supply chain managers, this translates to a more resilient production schedule with fewer potential points of failure compared to multi-step traditional routes.
Mechanistic Insights into Metal-Free Oxidative Amination
The core mechanism driving Method One involves the activation of the benzylic position by a strong oxidant, leading to the formation of a reactive intermediate that undergoes nucleophilic attack by the azide species. Specifically, oxidants like DDQ or hypervalent iodine reagents abstract hydride equivalents or facilitate single-electron transfer processes that weaken the benzylic C-C or C-H bonds. This activation allows for the subsequent displacement or oxidative functionalization with the nitrogen source, such as sodium azide or trimethylsilyl azide. The reaction proceeds through a concerted pathway that avoids the formation of free radical chains which could lead to polymerization or side reactions, ensuring high chemoselectivity for the primary amine product.

For Method Two, the mechanism relies on the protonation of the hydroxyl group in the aromatic alcohol derivative by a strong acid additive, converting it into a superior leaving group. This generates a benzylic carbocation or a tightly ion-paired intermediate that is highly susceptible to nucleophilic attack by the azide anion. The use of acids like trifluoroacetic acid or methanesulfonic acid provides the necessary acidity to drive this equilibrium forward without degrading the aromatic core. This mechanistic pathway is particularly advantageous for substrates where oxidative conditions might be detrimental to other functional groups on the ring. Understanding these distinct mechanisms allows process chemists to fine-tune reaction parameters, such as temperature and solvent polarity, to maximize efficiency for specific substrate classes.
Impurity control is inherently superior in this metal-free system. In traditional palladium-catalyzed reactions, trace metals can coordinate with product amines, forming stable complexes that are difficult to separate. In this new protocol, the byproducts are primarily organic salts or reduced forms of the oxidant, which can be easily removed via aqueous workup or standard crystallization techniques. This results in a final product with exceptionally low levels of inorganic impurities, meeting the stringent specifications required for GMP manufacturing of pharmaceutical intermediates. The absence of metal catalysts also removes the need for specialized reactor linings or dedicated equipment to prevent cross-contamination, further enhancing operational flexibility.
How to Synthesize Aromatic Amines Efficiently
Implementing this synthesis route in a laboratory or pilot plant setting requires careful attention to reagent stoichiometry and reaction conditions to ensure optimal yields. The patent provides extensive experimental data demonstrating that molar ratios of the nitrogen source to the substrate can vary widely, typically ranging from 1:1.5 to 1:20, allowing for optimization based on cost and conversion rates. Reaction temperatures are generally mild, operating effectively between 40°C and 100°C, which reduces energy consumption compared to high-temperature nitration processes. Solvent selection is also critical, with options ranging from non-polar hydrocarbons like n-hexane to polar protic solvents like trifluoroacetic acid, depending on the solubility of the specific starting materials.
- Mix the alkyl aromatic compound or aromatic alcohol derivative with a nitrogen-containing compound such as sodium azide in an organic solvent.
- Add an oxidant like DDQ for alkyl aromatics or an acid additive like trifluoroacetic acid for alcohol derivatives to initiate the reaction.
- Stir the mixture at temperatures between 40°C and 100°C for 4 to 36 hours, then quench with base and extract the product.
Commercial Advantages for Procurement and Supply Chain Teams
From a commercial perspective, the adoption of this metal-free synthesis technology offers profound advantages for cost reduction in pharmaceutical intermediate manufacturing. The primary driver of cost savings is the elimination of precious metal catalysts, which represent a significant portion of raw material expenses in traditional cross-coupling routes. By replacing palladium or copper with inexpensive organic oxidants or mineral acids, the direct material cost per kilogram of product is substantially lowered. Additionally, the removal of metal scavenging resins and the associated filtration steps reduces both consumable costs and labor time, leading to a more lean and efficient production process that enhances overall margin potential.
- Cost Reduction in Manufacturing: The economic benefits extend beyond simple reagent costs to include significant savings in waste treatment and purification. Traditional nitration processes generate acidic waste streams that require neutralization and specialized disposal, incurring high environmental compliance fees. This new method generates significantly less hazardous waste, and the organic byproducts are often easier to treat or recycle. Furthermore, the simplified workup procedure reduces the volume of solvents required for extraction and chromatography, lowering the total cost of ownership for the manufacturing campaign and improving the facility's environmental footprint.
- Enhanced Supply Chain Reliability: Relying on bulk alkyl aromatic compounds or simple aromatic alcohols as starting materials mitigates supply chain risks associated with specialized halogenated precursors. These bulk chemicals are produced on a massive scale for other industries, ensuring consistent availability and price stability even during market fluctuations. This reliability is crucial for long-term supply agreements with major pharmaceutical clients who require guaranteed continuity of supply. The robustness of the reaction conditions also means that production is less susceptible to delays caused by equipment maintenance or strict safety protocols associated with high-pressure hydrogenation.
- Scalability and Environmental Compliance: The scalability of this process is demonstrated by its compatibility with standard glass-lined or stainless steel reactors commonly found in multipurpose chemical plants. There is no need for exotic equipment capable of handling pyrophoric catalysts or high-pressure gases. This ease of scale-up from gram to ton scale accelerates the timeline from process development to commercial production. Moreover, the alignment with green chemistry principles, such as atom economy and hazard reduction, positions manufacturers favorably regarding increasingly strict global environmental regulations, future-proofing the production asset against regulatory tightening.
Frequently Asked Questions (FAQ)
To assist technical teams in evaluating the feasibility of this technology for their specific projects, we have compiled answers to common inquiries regarding the process capabilities and limitations. These insights are derived directly from the experimental data and scope defined within the patent documentation, providing a realistic view of what can be achieved in a production environment. Understanding these nuances is essential for accurate process design and risk assessment.
Q: Does this synthesis method leave transition metal residues in the final product?
A: No, the method described in patent CN109134267B is explicitly metal-free, eliminating the need for expensive palladium or copper catalysts and avoiding difficult-to-remove metal residues.
Q: What types of substrates are compatible with this amination process?
A: The process supports a wide range of substrates including alkyl benzenes, aromatic alcohol derivatives, and various heterocycles like pyridine, indole, and thiophene, allowing for diverse functional group tolerance.
Q: How does this method compare to traditional nitration-reduction routes in terms of safety?
A: This method avoids the use of strong nitrating acids and high-pressure hydrogenation steps, significantly reducing safety risks and waste disposal costs associated with traditional aniline synthesis.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Aromatic Amines Supplier
At NINGBO INNO PHARMCHEM, we recognize the transformative potential of metal-free synthetic methodologies in modernizing the supply chain for critical chemical intermediates. Our team of expert process chemists possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that innovative laboratory discoveries are successfully translated into robust industrial processes. We maintain stringent purity specifications and operate rigorous QC labs equipped with state-of-the-art analytical instrumentation to guarantee that every batch of aromatic amine meets the highest quality standards required by the global pharmaceutical industry.
We invite you to collaborate with us to leverage this advanced synthesis technology for your upcoming projects. Our technical procurement team is ready to provide a Customized Cost-Saving Analysis tailored to your specific molecule, demonstrating how switching to this metal-free route can optimize your budget. Please contact us to request specific COA data for our existing catalog of aniline derivatives or to discuss route feasibility assessments for your custom synthesis needs. Together, we can build a more sustainable and efficient supply chain for the next generation of life-saving medicines.
