Advanced Non-Halide Synthesis of Substituted Aromatic Amines for Commercial Scale-Up
Advanced Non-Halide Synthesis of Substituted Aromatic Amines for Commercial Scale-Up
The chemical industry is constantly seeking more sustainable and economically viable pathways for producing high-value intermediates, and the technology disclosed in patent CN1121706A represents a significant leap forward in this domain. This patent details a robust process for preparing substituted aromatic amines, specifically targeting the synthesis of 4-aminodiphenylamine (4-ADPA) and its derivatives without relying on traditional halide-based substitution mechanisms. By utilizing a nucleophilic compound and an azo-containing compound in a controlled solvent system, this method effectively bypasses the corrosive and environmentally taxing steps associated with conventional halide chemistry. For R&D directors and procurement specialists alike, understanding this shift from halide-dependent routes to azo-coupling strategies is crucial for optimizing supply chains and reducing long-term operational costs in the manufacture of pharmaceutical intermediates and rubber additives.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the preparation of substituted aromatic amines like 4-ADPA has relied heavily on nucleophilic aromatic substitution mechanisms where an amino functional group nucleophile replaces a halide, typically using substrates like parachloronitrobenzene. This conventional approach presents severe drawbacks, primarily because substituted halides are highly corrosive to reaction apparatus, leading to increased maintenance costs and potential safety hazards in large-scale manufacturing facilities. Furthermore, the halide byproducts generated during these reactions end up in waste streams, necessitating expensive and complex disposal procedures to meet stringent environmental regulations. The reliance on separate production units to generate starting materials like aniline and nitrobenzene further complicates the process flow, increasing the overall capital expenditure and throughput requirements for manufacturers attempting to scale these traditional methods.
The Novel Approach
In stark contrast, the novel approach outlined in the patent utilizes a non-halide methodology that contacts a nucleophilic compound with an azo-containing compound, such as azobenzene or its derivatives, in the presence of a suitable base. This reaction occurs within a confined zone at temperatures ranging from 10 °C to 150 °C, critically eliminating the generation of corrosive halide waste streams entirely. By shifting the synthetic strategy to an azo-coupling followed by reduction, the process not only mitigates equipment corrosion but also simplifies the purification workflow, as there is no need for expensive halide removal steps. This fundamental change in chemical architecture allows for a more streamlined production process that is inherently safer and more aligned with modern green chemistry principles, offering a distinct competitive advantage for suppliers of high-purity aromatic amines.
Mechanistic Insights into Azo-Coupling and Reduction
The core of this innovative synthesis lies in the precise interaction between the nucleophilic compound and the azo-containing species under basic conditions. The reaction mechanism involves the nucleophilic attack on the azo linkage, facilitated by a suitable solvent system and a controlled amount of protic substance. The molar ratio of the protic substance to the base is a critical parameter, maintained between 0:1 to about 5:1, to ensure optimal reaction kinetics without suppressing the nucleophilicity. This delicate balance allows for the formation of intermediate azo-compounds which are subsequently reduced to the final aromatic amine. The versatility of this mechanism is highlighted by the wide range of acceptable nucleophiles, including anilines, aliphatic amines, and various amides, allowing for the tailored synthesis of diverse substituted derivatives.
Furthermore, the structural diversity of the nucleophilic compounds plays a pivotal role in determining the final product profile and purity. For instance, diamides can serve as effective nucleophiles in this system, expanding the scope of accessible chemical space beyond simple anilines. 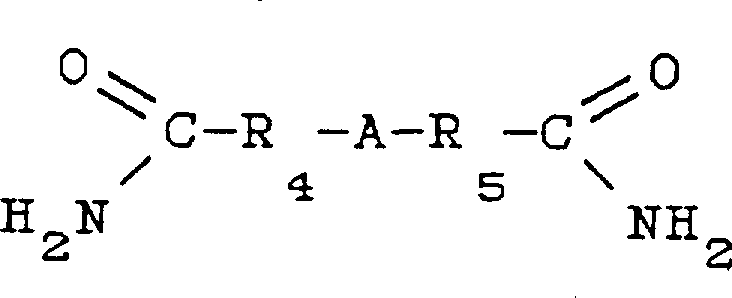 . The ability to incorporate complex structures like the diamide shown above demonstrates the robustness of the catalytic system in handling sterically demanding substrates. Following the coupling step, the reduction of the azo-intermediate is typically achieved via catalytic hydrogenation using metals like palladium or platinum, ensuring high conversion rates and minimizing the formation of unwanted byproducts, which is essential for meeting the stringent purity specifications required by pharmaceutical and electronic chemical clients.
. The ability to incorporate complex structures like the diamide shown above demonstrates the robustness of the catalytic system in handling sterically demanding substrates. Following the coupling step, the reduction of the azo-intermediate is typically achieved via catalytic hydrogenation using metals like palladium or platinum, ensuring high conversion rates and minimizing the formation of unwanted byproducts, which is essential for meeting the stringent purity specifications required by pharmaceutical and electronic chemical clients.
How to Synthesize Substituted Aromatic Amines Efficiently
Implementing this synthesis route requires careful attention to reaction conditions, particularly regarding the control of protic substances and the selection of the base. The process begins with contacting the selected nucleophile with the azo compound in a solvent such as dimethyl sulfoxide or aniline itself, followed by the addition of a base like potassium hydroxide or potassium tert-butoxide. Maintaining the reaction temperature within the optimal range of 50 °C to 90 °C is crucial for maximizing yield while minimizing side reactions. Once the coupling is complete, the mixture undergoes a reduction step, often in the same vessel, to yield the final amine product. For a comprehensive breakdown of the standardized operating procedures and specific reagent quantities, please refer to the detailed synthesis guide below.
- Contact a nucleophilic compound such as aniline or amide with an azo-containing compound in a suitable solvent system.
- React the mixture in the presence of a suitable base and a controlled amount of protic substance at 10 °C to 150 °C.
- Reduce the resulting reaction product under catalytic hydrogenation conditions to produce the final substituted aromatic amine.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the transition to this non-halide process offers tangible benefits that extend beyond mere chemical elegance. The elimination of halide starting materials directly translates to a reduction in raw material volatility and cost, as commodity chemicals like aniline and azobenzene are generally more stable and widely available than specialized halogenated intermediates. Additionally, the removal of corrosive halides from the process flow significantly extends the lifespan of reactor vessels and piping, leading to substantial capital savings on equipment maintenance and replacement over the long term. This process stability ensures a more reliable supply of critical intermediates, reducing the risk of production stoppages due to equipment failure or regulatory hurdles associated with hazardous waste disposal.
- Cost Reduction in Manufacturing: The primary driver for cost optimization in this process is the complete avoidance of halide waste treatment. Traditional methods incur significant expenses in neutralizing and disposing of halide salts, whereas this novel route generates a much cleaner waste profile. By eliminating the need for expensive halide scrubbing systems and reducing the corrosion-related downtime of manufacturing assets, the overall cost of goods sold is significantly lowered. Furthermore, the ability to perform the coupling and reduction in potentially fewer steps or simplified workups contributes to lower energy consumption and labor costs per kilogram of product produced.
- Enhanced Supply Chain Reliability: Sourcing halogenated intermediates can often be a bottleneck due to their classification as hazardous materials and the limited number of qualified suppliers. By shifting the feedstock requirement to non-halogenated precursors like azobenzene and aniline, manufacturers can tap into a broader and more resilient supply base. This diversification reduces the risk of supply disruptions caused by regulatory changes or logistical issues specific to hazardous cargo. Consequently, lead times for high-purity substituted aromatic amines can be stabilized, ensuring consistent delivery schedules for downstream customers in the pharmaceutical and agrochemical sectors.
- Scalability and Environmental Compliance: The process is designed with industrial scalability in mind, operating at moderate temperatures and pressures that are easily manageable in standard stainless steel reactors. The absence of corrosive halides simplifies the engineering requirements for scale-up, allowing for larger batch sizes without proportional increases in safety risks. From an environmental perspective, the reduction in hazardous waste generation aligns perfectly with increasingly strict global environmental standards, facilitating easier permitting and community acceptance for manufacturing sites. This compliance advantage future-proofs the supply chain against tightening regulations on chemical emissions and waste disposal.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this non-halide synthesis technology. These answers are derived directly from the experimental data and claims presented in the patent literature, providing a factual basis for evaluating the feasibility of this route for your specific application needs. Understanding these nuances is essential for making informed decisions about adopting this technology for large-scale production.
Q: How does this non-halide process improve environmental compliance compared to traditional methods?
A: Traditional methods often utilize substituted halides which are corrosive to reactors and generate hazardous halide waste streams requiring expensive disposal. This patented process eliminates the use of halide starting materials entirely, thereby removing the need for costly halide removal from waste streams and significantly reducing reactor corrosion risks.
Q: What is the critical role of the protic substance in this reaction mechanism?
A: The controlled amount of protic substance, such as water or alcohol, is vital for maintaining reaction selectivity. The molar ratio of protic substance to base must be carefully managed, typically between 0:1 to 5:1, to prevent the suppression of the nucleophilic reaction while ensuring high conversion efficiency of the azo-containing compound.
Q: Can this process be scaled for the production of antioxidant derivatives?
A: Yes, the process is highly scalable and specifically designed for industrial feasibility. The resulting substituted aromatic amines, such as 4-ADPA, can undergo standard reductive alkylation to produce alkylated diamines which serve as effective antioxidants and antiozonants in rubber and polymer applications.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Substituted Aromatic Amines Supplier
At NINGBO INNO PHARMCHEM, we recognize the transformative potential of the non-halide synthesis route described in CN1121706A for producing high-quality substituted aromatic amines. As a leading CDMO partner, we possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the theoretical benefits of this patent are fully realized in practical manufacturing settings. Our state-of-the-art facilities are equipped with rigorous QC labs capable of meeting stringent purity specifications, guaranteeing that every batch of 4-ADPA or related intermediates meets the exacting standards required by global pharmaceutical and specialty chemical companies.
We invite you to collaborate with us to leverage this advanced chemistry for your next project. Our technical team is ready to provide a Customized Cost-Saving Analysis tailored to your specific volume requirements, demonstrating how this non-halide route can optimize your budget. Please contact our technical procurement team today to request specific COA data and route feasibility assessments, and let us help you secure a sustainable and cost-effective supply of these critical chemical building blocks.
