Revolutionizing Secondary Amine Production via Metal-Free One-Pot Alkyne Hydration
The landscape of organic synthesis for nitrogen-containing heterocycles is undergoing a significant transformation, driven by the urgent need for greener, safer, and more cost-effective manufacturing processes. A pivotal development in this domain is documented in patent CN108358793B, which discloses a novel, metal-free methodology for synthesizing secondary amine compounds directly from alkynes. This innovation represents a paradigm shift away from traditional transition-metal catalyzed hydrogenation, offering a robust alternative that aligns perfectly with the stringent purity and safety standards required by modern pharmaceutical supply chains. The core of this technology lies in a sophisticated "three-step one-pot" strategy that seamlessly integrates alkyne hydration, imine condensation, and boron-mediated reduction into a single operational sequence. By leveraging inexpensive ethynyl-substituted benzenes as starting materials, this process not only democratizes access to complex amine scaffolds but also drastically reduces the environmental footprint associated with their production. For R&D teams seeking reliable pathways to high-value intermediates, this patent provides a blueprint for efficiency that bypasses the logistical nightmares of handling hazardous gases and removing trace heavy metals.
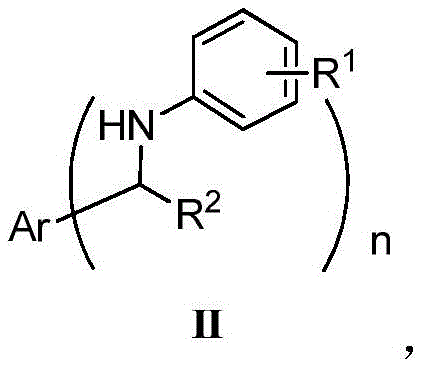
Furthermore, the structural versatility enabled by this synthetic route is exceptional, accommodating a wide array of substituents on both the aromatic ring and the amine nitrogen. As illustrated in the general formula, the method tolerates diverse functional groups including halogens, alkoxy groups, and nitro groups, making it an indispensable tool for the rapid generation of chemical libraries for drug discovery. The ability to synthesize such diverse structures under mild conditions without compromising yield is a testament to the robustness of the underlying chemistry. For procurement managers and supply chain heads, the implications are profound: a move towards this technology promises a more resilient supply chain less dependent on volatile precious metal markets and specialized high-pressure equipment. This report delves deep into the mechanistic nuances and commercial viability of this breakthrough, positioning it as a cornerstone for future cost reduction in API manufacturing.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the industrial synthesis of secondary amines has been dominated by reductive amination protocols that rely heavily on transition metal catalysts such as palladium on carbon or Raney nickel, often requiring high-pressure hydrogen gas. These conventional methods present a myriad of operational challenges that inflate costs and complicate regulatory compliance. The use of hydrogen gas necessitates specialized high-pressure reactors and rigorous safety protocols to mitigate explosion risks, creating a significant barrier to entry for smaller manufacturing facilities and increasing capital expenditure. Moreover, the reliance on precious metal catalysts introduces a critical vulnerability in the supply chain, as the prices of metals like palladium are subject to extreme market volatility. Beyond the economic factors, the presence of metal residues in the final product is a major concern for pharmaceutical applications, where strict limits on heavy metal impurities are enforced by global health authorities. Removing these trace metals often requires additional purification steps, such as scavenging or recrystallization, which inevitably lead to yield losses and increased waste generation. The cumulative effect of these inefficiencies is a manufacturing process that is both economically burdensome and environmentally unsustainable.
The Novel Approach
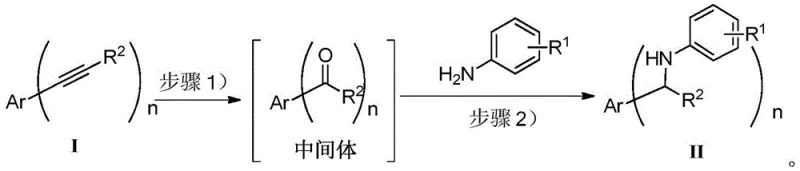
In stark contrast, the methodology described in patent CN108358793B offers a revolutionary "three-step one-pot" solution that elegantly circumvents these traditional bottlenecks. This novel approach utilizes a Brønsted acid catalyst, specifically trifluoromethanesulfonic acid, to drive the initial hydration of the alkyne, followed by a spontaneous condensation with an aromatic amine and a final reduction using a boron reagent. The elimination of transition metals from the catalytic cycle is a game-changer, effectively removing the need for expensive metal scavengers and the associated risk of metal contamination in the final active pharmaceutical ingredient. The reaction proceeds under relatively mild thermal conditions, typically ranging from 40°C to 80°C, and at atmospheric pressure, which significantly lowers the energy consumption and safety risks compared to high-pressure hydrogenation. The use of tetrahydroxydiboron as a reducing agent is particularly ingenious; it is a stable, non-pyrophoric solid that is easy to handle and store, unlike many traditional hydride reagents. This streamlined process not only enhances the overall atom economy but also simplifies the workflow by consolidating three distinct chemical transformations into a single vessel, thereby reducing solvent usage and processing time.
Mechanistic Insights into Brønsted Acid-Catalyzed Hydration and Boron Reduction
The success of this metal-free synthesis hinges on a delicate interplay between acid catalysis and the unique properties of fluorinated solvents. The mechanism initiates with the activation of the alkyne triple bond by the strong Brønsted acid, trifluoromethanesulfonic acid, which facilitates the nucleophilic attack of water to form an enol intermediate that rapidly tautomerizes to the corresponding ketone. This hydration step is critical and is significantly accelerated by the use of fluorinated alcohols like trifluoroethanol as co-solvents. These fluorinated solvents are not merely inert media; they participate actively in the reaction by forming extensive hydrogen-bonding networks that stabilize the charged transition states and enhance the acidity of the catalyst. Following the formation of the ketone, the aromatic amine is introduced directly into the reaction mixture, where it undergoes condensation to form an imine intermediate. This step is driven by the removal of water and the thermodynamic stability of the resulting C=N bond. The final and perhaps most crucial step is the reduction of this imine to the secondary amine using tetrahydroxydiboron. Unlike traditional hydride sources that might reduce other functional groups indiscriminately, the boron reagent exhibits high chemoselectivity for the imine bond, ensuring that sensitive substituents on the aromatic rings remain intact. This precise control over reactivity is essential for maintaining the integrity of complex molecular architectures often found in drug candidates.
From an impurity control perspective, this mechanism offers distinct advantages over metal-catalyzed routes. In traditional hydrogenation, side reactions such as over-reduction or dehalogenation can occur, leading to difficult-to-separate impurities that compromise the purity profile of the final product. The boron-mediated reduction described here operates under much milder conditions, minimizing the potential for such degradative pathways. Furthermore, the absence of metal catalysts eliminates the formation of metal-organic complexes that can act as persistent impurities. The byproducts of the boron reduction are primarily boric acid derivatives, which are water-soluble and can be easily removed during the aqueous workup phase, leaving behind a high-purity organic product. This inherent cleanliness of the reaction profile translates directly into simplified downstream processing, as fewer chromatographic separations or recrystallizations are required to meet pharmaceutical grade specifications. For quality control teams, this means a more predictable and robust impurity profile, reducing the analytical burden and accelerating the release of batches for clinical or commercial use.
How to Synthesize N-(1-phenylethyl)aniline Efficiently
The practical implementation of this synthesis is remarkably straightforward, designed to be accessible even to laboratories with limited infrastructure. The process begins with the dissolution of the ethynyl-substituted benzene starting material in a mixture of trifluoroethanol and water, followed by the addition of the acid catalyst. Detailed standardized synthesis steps are provided below to guide the replication of this high-yielding protocol. The reaction conditions are optimized to balance reaction rate with selectivity, ensuring that the hydration proceeds to completion before the amine is added. Once the ketone intermediate is formed, the temperature is raised to facilitate the condensation with the aromatic amine, a step that typically requires only a short duration to reach equilibrium. The final reduction step is the longest, requiring extended heating to ensure complete conversion of the imine to the amine, but this is a passive process that requires minimal operator intervention. The workup involves a simple extraction with ethyl acetate, leveraging the solubility differences between the organic product and the inorganic boron byproducts. This ease of execution makes the method highly attractive for both small-scale discovery chemistry and larger pilot plant operations.
- Hydrate ethynyl-substituted benzene using trifluoromethanesulfonic acid in a fluorinated alcohol/water mixture at 40°C to form a ketone intermediate.
- Directly add aromatic amine to the reaction system and heat to 80°C to generate the imine species in situ.
- Introduce tetrahydroxydiboron to the mixture and maintain at 80°C for 14 hours to reduce the imine to the final secondary amine product.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain directors, the adoption of this metal-free synthesis protocol offers a compelling value proposition centered on cost stability and operational resilience. The primary driver of cost reduction in manufacturing stems from the complete elimination of precious metal catalysts. By replacing expensive palladium or platinum systems with inexpensive Brønsted acids and boron reagents, the direct material costs are significantly lowered, and the volatility associated with precious metal pricing is entirely avoided. Additionally, the one-pot nature of the process reduces the number of unit operations required, which translates to lower labor costs, reduced energy consumption for heating and cooling between steps, and minimized solvent waste disposal fees. The simplicity of the equipment requirements—standard glass-lined reactors capable of moderate heating rather than specialized high-pressure vessels—further lowers the barrier to entry and allows for greater flexibility in manufacturing site selection. These factors combine to create a manufacturing process that is not only cheaper but also more agile and responsive to market demands.
- Cost Reduction in Manufacturing: The economic benefits of this technology are multifaceted, extending beyond simple reagent costs to encompass the entire production lifecycle. By eliminating the need for transition metal catalysts, manufacturers avoid the substantial expenses associated with catalyst recovery, recycling, or disposal, which can be a significant portion of the operating budget in traditional processes. Furthermore, the high atom economy of the reaction ensures that a maximum proportion of the starting materials ends up in the final product, minimizing waste and maximizing yield. The use of commodity chemicals like trifluoroethanol and tetrahydroxydiboron ensures a stable and predictable supply chain for raw materials, shielding the production process from the geopolitical and market fluctuations that often affect specialty reagents. This comprehensive approach to cost optimization makes the process highly competitive for the commercial production of high-volume pharmaceutical intermediates.
- Enhanced Supply Chain Reliability: In an era where supply chain disruptions are a constant threat, the robustness of this synthetic route provides a strategic advantage. The raw materials required, such as phenylacetylene derivatives and anilines, are widely available from multiple global suppliers, reducing the risk of single-source dependency. The mild reaction conditions and lack of hazardous gases like hydrogen mean that the process can be safely operated in a wider range of facilities, increasing the available manufacturing capacity and reducing lead times for high-purity intermediates. The stability of the reagents also allows for longer shelf lives and easier logistics, ensuring that production schedules are not delayed by the degradation of sensitive catalysts. This reliability is crucial for maintaining continuous supply to downstream customers and meeting the just-in-time delivery expectations of the modern pharmaceutical industry.
- Scalability and Environmental Compliance: Scaling chemical processes from the bench to the plant floor is often fraught with challenges, but this metal-free method is inherently scalable due to its simplicity and safety profile. The absence of exothermic hydrogenation events and high-pressure requirements simplifies the engineering controls needed for large-scale production, reducing the time and cost associated with process validation. From an environmental perspective, the process aligns with the principles of green chemistry by reducing waste generation and avoiding the use of toxic heavy metals. The aqueous workup generates waste streams that are easier to treat and dispose of compared to those containing heavy metal residues, facilitating compliance with increasingly stringent environmental regulations. This sustainability angle is not just a regulatory checkbox but a market differentiator that appeals to eco-conscious partners and end-users.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this synthesis method, based on the detailed data provided in the patent literature. These answers are designed to clarify the operational parameters and strategic benefits for stakeholders evaluating this technology for their own portfolios. Understanding the nuances of the reaction conditions and the rationale behind the reagent choices is key to successful technology transfer and scale-up.
Q: What are the primary advantages of this metal-free synthesis over traditional hydrogenation?
A: This method eliminates the need for expensive transition metal catalysts like palladium or platinum and avoids the safety hazards associated with high-pressure hydrogen gas, significantly simplifying equipment requirements and downstream purification.
Q: Why is tetrahydroxydiboron preferred as the reducing agent in this protocol?
A: Tetrahydroxydiboron acts as a mild, non-toxic, and cost-effective reducing agent that selectively reduces the imine intermediate without affecting other sensitive functional groups, ensuring high chemoselectivity and yield.
Q: Can this process be scaled for industrial production of pharmaceutical intermediates?
A: Yes, the one-pot nature of the reaction minimizes solvent usage and unit operations, while the use of commercially available raw materials and mild reaction conditions makes it highly suitable for commercial scale-up of complex pharmaceutical intermediates.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Secondary Amine Supplier
The technological advancements detailed in patent CN108358793B underscore the immense potential of metal-free synthesis in modernizing the production of critical chemical building blocks. At NINGBO INNO PHARMCHEM, we recognize the transformative power of such innovations and have integrated similar cutting-edge methodologies into our own manufacturing capabilities. As a premier CDMO partner, we possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that our clients receive consistent, high-quality supply regardless of volume. Our commitment to excellence is backed by stringent purity specifications and rigorous QC labs that utilize state-of-the-art analytical instrumentation to verify every batch against the highest industry standards. We understand that in the pharmaceutical sector, purity is not just a metric but a mandate, and our processes are designed to deliver products that exceed these exacting requirements.
We invite you to explore how our expertise in green chemistry and process optimization can drive value for your organization. Whether you are looking to optimize an existing route or develop a new pathway for a complex intermediate, our team is ready to provide a Customized Cost-Saving Analysis tailored to your specific needs. We encourage you to contact our technical procurement team today to request specific COA data and route feasibility assessments. By partnering with us, you gain access to a supply chain that is not only reliable and compliant but also forward-thinking and dedicated to sustainable innovation. Let us help you navigate the complexities of chemical manufacturing with confidence and precision.
