Advanced Ruxolitinib Synthesis: Scalable Routes for Commercial API Production
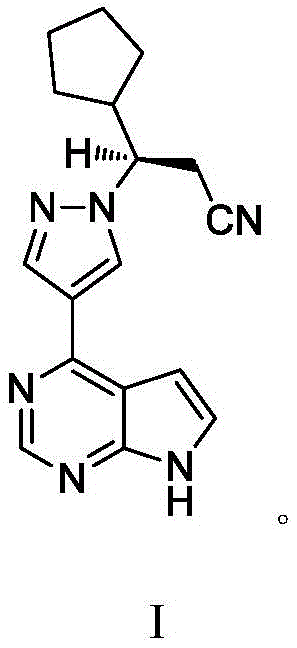
The pharmaceutical industry continuously seeks robust manufacturing pathways for high-value kinase inhibitors, and Patent CN111763209B presents a significant advancement in the synthesis of Ruxolitinib, also known as Luccotinib. This specific intellectual property outlines a preparation method that strategically bypasses the limitations of traditional asymmetric catalysis, offering a streamlined approach to constructing the critical chiral center. By leveraging chiral resolution techniques using D-tartaric acid instead of costly rhodium or palladium catalysts, the process achieves exceptional stereoselectivity with ee values reaching 99.4%. This technical breakthrough is particularly relevant for a reliable pharmaceutical intermediate supplier aiming to secure long-term supply chains for oncology treatments. The methodology encompasses a series of well-defined transformations, including acyl halogenation, amidation, and dehydration, all optimized for industrial feasibility. Understanding the nuances of this patent is essential for R&D directors evaluating process robustness and procurement managers analyzing cost structures in API manufacturing.
The limitations of conventional methods for synthesizing JAK inhibitors often revolve around the reliance on precious metal catalysts and complex chiral ligands, which introduce significant cost volatility and supply chain risks. Traditional routes described in prior art, such as those utilizing rhodium catalysts, frequently require harsh reaction conditions and extensive purification steps to remove trace metal impurities, which is a critical concern for regulatory compliance. Furthermore, the chiral purity obtained through direct asymmetric synthesis can sometimes be inconsistent, necessitating additional recrystallization steps that reduce overall yield. In contrast, the novel approach detailed in this patent utilizes a chiral pool strategy combined with resolution, which inherently stabilizes the stereochemical outcome. This shift from catalytic asymmetry to resolution-based chirality introduction simplifies the process control parameters and reduces the dependency on specialized reagents that may face supply constraints. The new route ensures that the critical pyrazole ring formation occurs under mild acidic or basic conditions, preserving the integrity of sensitive functional groups throughout the synthesis.
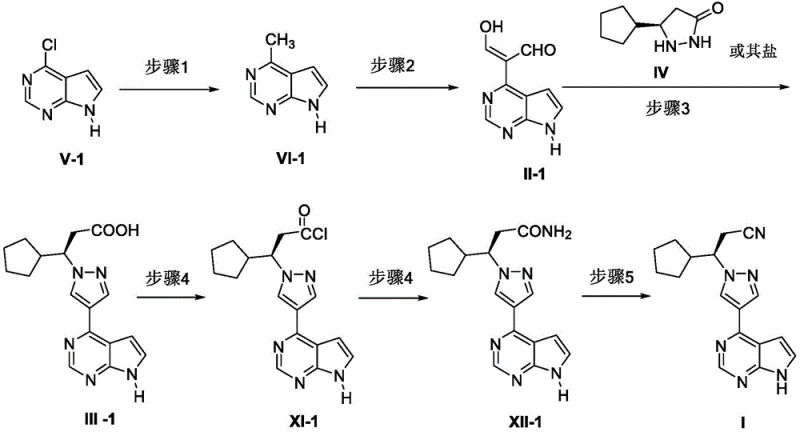
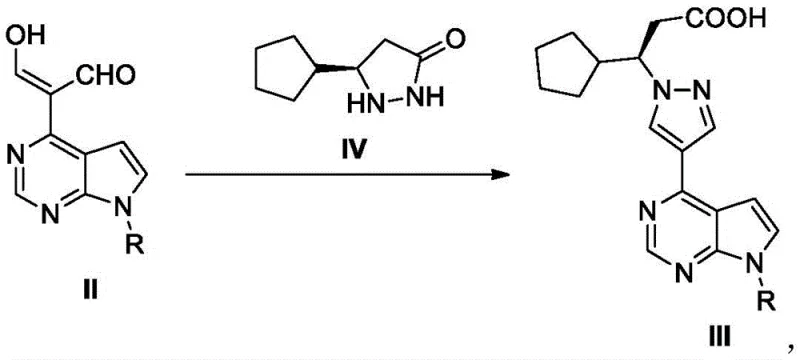
Mechanistic insights into the coupling reaction reveal a sophisticated interplay between the electrophilic aldehyde intermediate and the nucleophilic chiral hydrazine derivative. The formation of the pyrazole ring, a cornerstone of the Ruxolitinib structure, proceeds through a condensation mechanism that is highly sensitive to pH and solvent polarity. The patent specifies the use of solvents like acetic acid or ethanol, which facilitate the proton transfer necessary for cyclization while maintaining the solubility of the intermediates. This careful selection of reaction media prevents the formation of unwanted regioisomers, thereby enhancing the purity profile of the crude product. Additionally, the subsequent conversion of the carboxylic acid to the amide and finally to the nitrile involves activation via acyl chlorides, typically using oxalyl chloride or phosphorus oxychloride. This activation strategy ensures high atom utilization and minimizes the generation of hazardous waste streams. For R&D teams, understanding these mechanistic details is vital for troubleshooting potential scale-up issues and ensuring that the impurity profile remains within strict pharmacopeial limits.
How to Synthesize Ruxolitinib Efficiently
The synthesis of Ruxolitinib described in this patent offers a clear pathway for laboratories aiming to replicate or license this technology for production. The process begins with the preparation of the chiral pyrazolidinone intermediate, which serves as the source of stereochemistry for the final molecule. Following the resolution step, the coupling with the pyrrolopyrimidine aldehyde must be conducted under controlled thermal conditions to ensure complete conversion. The detailed standardized synthesis steps see the guide below, which outlines the specific molar ratios and solvent systems required to achieve the reported yields. Adhering to these parameters is crucial for maintaining the high purity specifications demanded by regulatory bodies for oncology APIs. This section serves as a technical reference for process chemists looking to implement this route in a pilot or commercial plant setting.
- Prepare the chiral pyrazolidinone intermediate via condensation of cyclopentylacrylic acid and hydrazine, followed by D-tartaric acid resolution.
- Execute Vilsmeier-Haack formylation on 4-methyl-7H-pyrrolo[2,3-d]pyrimidine to generate the key aldehyde intermediate.
- Couple the chiral pyrazolidinone with the pyrrolopyrimidine aldehyde, followed by amidation and dehydration to yield the final nitrile.
Commercial Advantages for Procurement and Supply Chain Teams
From a commercial perspective, this synthesis route offers substantial benefits for procurement and supply chain teams focused on cost reduction in API manufacturing. By eliminating the need for expensive asymmetric transition metal catalysts, the process significantly lowers the raw material costs associated with each batch. This reduction in catalyst expense is compounded by the simplified downstream processing, as there is no need for specialized metal scavenging resins or extensive washing protocols to meet heavy metal specifications. Furthermore, the use of common industrial solvents such as dichloromethane, NMP, and ethanol enhances supply chain reliability, as these materials are readily available from multiple global vendors. The robustness of the chiral resolution step ensures consistent quality, reducing the risk of batch failures that can disrupt production schedules. These factors collectively contribute to a more resilient supply chain capable of meeting the demands of high-purity pharmaceutical intermediates without compromising on delivery timelines.
- Cost Reduction in Manufacturing: The elimination of precious metal catalysts such as rhodium or palladium removes a major cost driver from the bill of materials. Additionally, the high yields reported in the patent examples indicate efficient material usage, which minimizes waste disposal costs. The process avoids complex protection and deprotection sequences where possible, further reducing the number of unit operations and associated labor and utility costs. This streamlined approach allows for a more competitive pricing structure for the final active pharmaceutical ingredient.
- Enhanced Supply Chain Reliability: Relying on commodity chemicals and solvents rather than specialized chiral ligands mitigates the risk of supply disruptions. The chiral resolving agent, D-tartaric acid, is a bulk chemical with a stable global supply, ensuring that production can continue uninterrupted. The mild reaction conditions also reduce the wear and tear on reactor equipment, leading to lower maintenance costs and higher equipment availability. This reliability is critical for maintaining continuous supply to downstream formulation partners.
- Scalability and Environmental Compliance: The process is designed with industrial production in mind, utilizing solvents and reagents that are manageable at large scales. The avoidance of heavy metals simplifies environmental compliance and waste treatment, as the effluent streams are less hazardous. The high atom utilization rate mentioned in the patent suggests a greener chemistry profile, which aligns with increasing regulatory pressure for sustainable manufacturing practices. This scalability ensures that the process can be adapted from kilogram to ton-scale production seamlessly.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this synthesis technology. These answers are derived directly from the technical specifications and experimental data provided in the patent documentation. They are intended to clarify the operational advantages and chemical feasibility of the route for potential partners and licensees. Understanding these details helps in making informed decisions regarding technology transfer and process adoption.
Q: How does this patent improve chiral purity compared to traditional catalytic methods?
A: The patent utilizes chiral resolution with D-tartaric acid rather than expensive asymmetric metal catalysts, achieving ee values exceeding 99% without complex ligand systems.
Q: What are the key reagents for the dehydration step in this process?
A: The process employs phosphorus oxychloride or cyanuric chloride as dehydrating agents to convert the amide intermediate to the critical cyano group under mild conditions.
Q: Is this synthesis route suitable for large-scale commercial manufacturing?
A: Yes, the route avoids harsh reaction conditions and expensive transition metals, utilizing commercially available solvents like dichloromethane and NMP, facilitating commercial scale-up of complex pharmaceutical intermediates.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Ruxolitinib Supplier
At NINGBO INNO PHARMCHEM, we possess the technical expertise to translate complex patent methodologies like CN111763209B into commercial reality. Our team has extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the transition from lab to plant is smooth and efficient. We maintain stringent purity specifications and operate rigorous QC labs to guarantee that every batch of Ruxolitinib intermediate meets the highest global standards. Our commitment to quality and compliance makes us a trusted partner for pharmaceutical companies seeking a reliable Ruxolitinib supplier.
We invite you to contact our technical procurement team to discuss how we can support your supply chain needs. Request a Customized Cost-Saving Analysis to understand the economic benefits of this specific synthesis route for your project. We are ready to provide specific COA data and route feasibility assessments to help you move forward with confidence. Partner with us to secure a stable and cost-effective supply of high-quality oncology intermediates.
