Advanced Synthesis of Brexpiprazole Intermediate Using Phthalimide Protection for Commercial Scale
Introduction to the Novel Brexpiprazole Intermediate Synthesis
The pharmaceutical industry constantly seeks robust synthetic routes for complex antipsychotic agents, and the preparation of brexpiprazole intermediates remains a critical challenge for supply chain stability. Patent CN114181145A introduces a groundbreaking preparation method that addresses longstanding issues regarding yield and purity in the synthesis of 7-[4-(piperazin-1-yl)butoxy]quinolin-2(1H)-one derivatives. This innovation is particularly significant for manufacturers aiming to secure a reliable pharmaceutical intermediates supplier capable of delivering high-quality materials consistently. By shifting away from traditional direct alkylation methods that suffer from poor selectivity, this new approach utilizes a phthalimide protection strategy to ensure precise mono-substitution. The technical breakthrough lies in the strategic use of potassium phthalimide to mask the amine functionality during the initial chain extension, effectively preventing the formation of bis-alkylated byproducts that plague conventional routes. This level of control is essential for maintaining the stringent purity specifications required for downstream API synthesis.
Furthermore, the economic implications of this synthetic advancement cannot be overstated for procurement teams focused on cost reduction in API manufacturing. The method relies on readily available starting materials such as 1,4-dibromobutane and 7-hydroxyquinolinone, which are commodity chemicals with stable pricing structures compared to specialized protected piperazines. The process eliminates the need for expensive and hazardous reagents like methanesulfonyl chloride, which not only reduces raw material costs but also minimizes equipment corrosion and maintenance expenses. For supply chain heads, the operational simplicity of this route—operating at moderate temperatures between 60°C and 100°C without high-pressure requirements—translates to enhanced supply chain reliability and reduced lead time for high-purity pharmaceutical intermediates. The ability to recycle solvents like acetone and ethanol further aligns with modern green chemistry initiatives, ensuring long-term sustainability for large-scale production facilities.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of brexpiprazole intermediates has been hindered by significant chemical inefficiencies that impact both yield and operational safety. Prior art, such as the methods described in WO2018172463, typically involves reacting Boc-protected piperazine directly with 1,4-disubstituted butanes. A major drawback of this approach is the lack of selectivity; the piperazine nitrogen can attack both ends of the dihaloalkane simultaneously, leading to the formation of dimeric impurities that are chemically similar to the desired product and extremely difficult to separate. This side reaction drastically reduces the overall yield and necessitates complex purification steps, such as column chromatography, which are impractical for multi-ton commercial production. Additionally, other reported routes, like those in WO2017/025987, rely on the activation of hydroxyl groups using methanesulfonyl chloride. This reagent is highly corrosive to stainless steel reactors, posing severe risks to equipment integrity and requiring specialized lining materials that increase capital expenditure. The toxicity and environmental burden of handling such aggressive sulfonylating agents also complicate waste treatment protocols, making these conventional methods increasingly obsolete in a regulated manufacturing environment.
The Novel Approach
The innovative route disclosed in the patent data fundamentally reimagines the construction of the butoxy-piperazine linker by employing a Gabriel Synthesis-inspired strategy. Instead of starting with the sensitive piperazine ring, the process begins with the alkylation of potassium phthalimide with 1,4-dibromobutane. This step is highly selective for mono-substitution due to the steric and electronic properties of the phthalimide anion, effectively acting as a robust protecting group for the future amine. As illustrated in the comprehensive reaction scheme below, this intermediate is then coupled with 7-hydroxyquinolinone to establish the ether linkage before the piperazine ring is even formed.
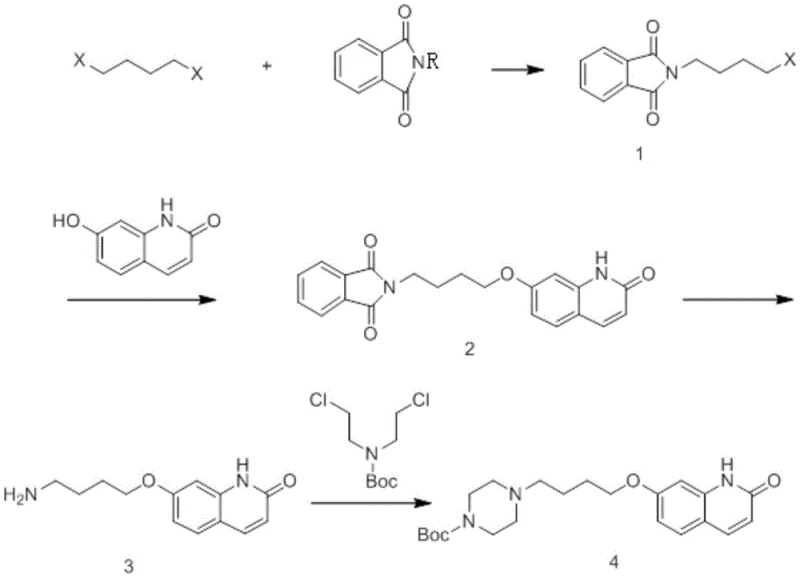
Subsequent deprotection with hydrazine hydrate reveals the primary amine, which is then cyclized with Boc-bis-2-chloroethylamine to construct the piperazine ring in a controlled manner. This sequence completely bypasses the issue of double alkylation associated with direct piperazine substitution. The final deprotection step using hydrochloric acid in methanol is mild and efficient, yielding the target intermediate with exceptional purity. By decoupling the chain extension from the ring formation, this novel approach offers a modular and scalable pathway that significantly simplifies post-reaction processing and improves the overall mass balance of the synthesis.
Mechanistic Insights into Phthalimide-Mediated Alkylation and Cyclization
The core mechanistic advantage of this process lies in the nucleophilic substitution dynamics of the phthalimide anion. In the initial step, potassium phthalimide acts as a strong nucleophile in a polar aprotic solvent like acetone or DMF, attacking the terminal carbon of 1,4-dibromobutane via an SN2 mechanism. The resulting imide is stable and unreactive towards further alkylation under these conditions, effectively capping one end of the butyl chain. This prevents the 'polymerization' or dimerization often seen when using primary amines directly. Following the etherification with 7-hydroxyquinolinone, which proceeds via base-mediated phenoxide formation, the phthalimide group is cleaved using hydrazine hydrate. This hydrazinolysis reaction is highly specific, converting the cyclic imide into a soluble phthalhydrazide byproduct while releasing the free primary amine (Compound 3) in high yield. The absence of competing side reactions at this stage ensures that the amine functionality is available exclusively for the subsequent ring-closing step.
The formation of the piperazine ring itself is a critical transformation that dictates the final quality of the intermediate. This cyclization involves the reaction of the linear amine with Boc-bis-2-chloroethylamine. To facilitate this intramolecular SN2 reaction, the patent specifies the addition of a catalytic amount of iodide salt, such as potassium iodide. The iodide ion acts as a nucleophilic catalyst, performing a Finkelstein-type halogen exchange to convert the less reactive chloro-groups into more reactive iodo-groups in situ. This enhances the electrophilicity of the ethyl chains, allowing the amine to displace the halide and close the ring efficiently at moderate temperatures (around 70°C).
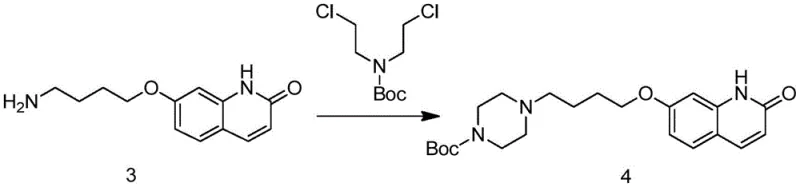
This catalytic cycle ensures that the reaction proceeds to completion without requiring excessive heat that could degrade the sensitive quinolinone moiety. Furthermore, the use of the Boc group on the bis-chloroethylamine precursor protects the second nitrogen atom, ensuring that the cyclization occurs only at the intended site. This precise control over regiochemistry is vital for minimizing structural impurities. The final product, after acid-mediated Boc removal, exhibits a clean impurity profile, as the robust protection-deprotection sequence filters out potential side products at each stage, resulting in a material that meets the rigorous standards expected from a high-purity OLED material or pharmaceutical intermediate supplier.
How to Synthesize Brexpiprazole Intermediate Efficiently
Implementing this synthesis requires careful attention to stoichiometry and solvent selection to maximize the benefits of the phthalimide strategy. The process is divided into five distinct chemical transformations, each optimized for high conversion and ease of isolation. The initial alkylation is best performed in acetone with reflux, allowing for simple filtration of inorganic salts. The subsequent etherification requires a higher boiling solvent like DMF to drive the reaction with the phenolic substrate. Deprotection with hydrazine is exothermic and should be managed with controlled addition, while the cyclization step benefits from the iodide catalyst to lower the activation energy. Finally, the acid deprotection is a straightforward salt formation and hydrolysis that can be conducted at room temperature.
- React 1,4-dibromobutane with potassium phthalimide in acetone with potassium carbonate to form the mono-substituted phthalimide derivative (Compound 1).
- Couple Compound 1 with 7-hydroxyquinolin-2(1H)-one in DMF using potassium carbonate to attach the quinolinone moiety (Compound 2).
- Deprotect the phthalimide group using hydrazine hydrate in ethanol to reveal the primary amine (Compound 3).
- Cyclize the amine with Boc-bis-2-chloroethylamine using potassium iodide and carbonate to form the protected piperazine ring (Compound 4).
- Remove the Boc protecting group using saturated HCl in methanol to obtain the final brexpiprazole intermediate (Compound 5).
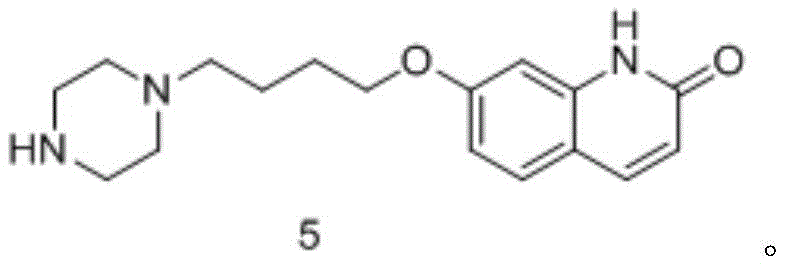
For detailed operational parameters, including specific molar ratios, temperature ramps, and workup procedures, operators should refer to the standardized protocol derived from the patent examples. The following guide outlines the critical control points for each stage to ensure reproducibility and safety during scale-up.
Commercial Advantages for Procurement and Supply Chain Teams
From a commercial perspective, this synthetic route offers transformative advantages for organizations managing the supply of complex heterocyclic intermediates. The shift from specialized, protected piperazines to commodity chemicals like phthalimide and dibromobutane represents a significant strategic pivot in raw material sourcing. This change mitigates the risk of supply disruptions caused by the limited availability of niche reagents and insulates the production cost from the volatility of the fine chemical market. Moreover, the elimination of corrosive reagents like methanesulfonyl chloride extends the lifespan of reactor vessels and piping, leading to substantial cost savings in capital maintenance and reducing the frequency of plant shutdowns for repairs. The environmental profile of the process is also markedly improved, as the solvents used (acetone, ethanol, methanol) are widely recycled in industrial settings, lowering waste disposal fees and aligning with corporate sustainability goals.
- Cost Reduction in Manufacturing: The economic efficiency of this method is driven by the high atom economy of the Gabriel synthesis step and the avoidance of expensive chromatographic purifications. By preventing the formation of difficult-to-remove bis-alkylated impurities, the process reduces the loss of valuable quinolinone starting material, which is often the cost-driving component of the molecule. Additionally, the use of catalytic potassium iodide in the cyclization step allows for faster reaction times and lower energy consumption compared to non-catalyzed thermal methods. These factors combine to lower the overall cost of goods sold (COGS), making the final API more competitive in the generic pharmaceutical market without compromising on quality standards.
- Enhanced Supply Chain Reliability: Supply chain resilience is bolstered by the use of globally available raw materials that are produced by multiple vendors, reducing dependency on single-source suppliers. The robustness of the chemical steps, which tolerate minor variations in temperature and mixing without significant yield loss, ensures consistent batch-to-batch quality. This reliability is crucial for maintaining continuous production schedules and meeting the just-in-time delivery requirements of downstream API manufacturers. The simplified workup procedures, primarily involving filtration and crystallization rather than complex extractions, also reduce the turnaround time between batches, effectively increasing the throughput capacity of existing manufacturing facilities.
- Scalability and Environmental Compliance: The process is inherently scalable because it avoids unit operations that are difficult to translate from lab to plant, such as low-temperature cryogenic reactions or high-pressure hydrogenations. Operating at atmospheric pressure and moderate temperatures simplifies the engineering requirements for large-scale reactors. From an environmental compliance standpoint, the absence of heavy metal catalysts and toxic sulfonyl chlorides simplifies the effluent treatment process. The aqueous waste streams generated are easier to treat biologically or chemically, reducing the burden on wastewater treatment plants and ensuring adherence to increasingly strict environmental regulations in major pharmaceutical manufacturing hubs.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this phthalimide-based synthesis route. These answers are derived directly from the experimental data and comparative analysis provided in the patent documentation, offering clarity on the practical benefits of adopting this technology. Understanding these nuances is essential for technical teams evaluating the feasibility of technology transfer and for procurement professionals assessing the long-term value proposition of this supply chain solution.
Q: Why is the phthalimide protection strategy superior to direct Boc-piperazine alkylation?
A: Direct alkylation of Boc-piperazine with dihaloalkanes often leads to double substitution, where both ends of the alkane react with piperazine, creating difficult-to-remove impurities and lowering yield. The phthalimide strategy (Gabriel Synthesis) ensures mono-substitution initially, providing better control over the reaction pathway and higher purity.
Q: What are the environmental advantages of this new synthesis method?
A: Unlike previous methods that utilized highly corrosive methanesulfonyl chloride or required harsh conditions, this process uses common solvents like acetone and ethanol which can be recycled. It avoids toxic reagents and operates under mild temperatures (60-100°C), making it safer for industrial operators and easier to treat waste streams.
Q: Can this process be scaled for commercial API production?
A: Yes, the process is designed for industrialization. It avoids special conditions like high pressure or cryogenic temperatures. The use of robust reagents like potassium carbonate and standard solvents facilitates easy scale-up from kilogram to multi-ton production without significant process redesign.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Brexpiprazole Intermediate Supplier
At NINGBO INNO PHARMCHEM, we recognize that the successful commercialization of antipsychotic therapies depends on the availability of high-quality, cost-effective intermediates. Our technical team has extensively analyzed the phthalimide protection strategy and is fully prepared to implement this advanced route for our global partners. We possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the transition from laboratory optimization to full-scale manufacturing is seamless and efficient. Our state-of-the-art facilities are equipped with the necessary corrosion-resistant reactors and solvent recovery systems to handle this chemistry safely and sustainably. We maintain stringent purity specifications through our rigorous QC labs, utilizing advanced analytical techniques to verify that every batch meets the critical quality attributes required for API synthesis.
We invite pharmaceutical companies and contract manufacturers to collaborate with us to leverage this innovative synthesis method for their brexpiprazole supply chains. By partnering with us, you gain access to a Customized Cost-Saving Analysis that quantifies the specific economic benefits of switching to this route for your volume requirements. We encourage you to contact our technical procurement team to request specific COA data from our pilot batches and to discuss route feasibility assessments tailored to your project timelines. Together, we can optimize the production of this vital psychiatric medication, ensuring patient access while driving operational excellence in the pharmaceutical supply chain.
