Advanced Synthesis of 3-(1-Hydroxyphenyl-1-alkoxyiminomethyl)dioxazines for Antifungal Applications
The pharmaceutical and agrochemical industries continuously demand more efficient pathways for synthesizing complex heterocyclic intermediates, particularly those serving as precursors for antifungal agents. Patent CN1184811A discloses a groundbreaking methodology for the preparation of 3-(1-hydroxyphenyl-1-alkoxyiminomethyl)dioxazines, a class of compounds critical for developing next-generation fungicides. This technical insight report analyzes the novel rearrangement strategy detailed in the patent, which offers a robust alternative to traditional multi-step syntheses. By leveraging a unique base-catalyzed rearrangement of O-hydroxyethyl-O'-alkyl-benzofurandione dioximes, manufacturers can achieve superior purity profiles and operational simplicity. For procurement leaders seeking a reliable pharmaceutical intermediate supplier, understanding the mechanistic advantages of this route is essential for evaluating long-term supply chain resilience and cost-effectiveness in the production of high-value antifungal active ingredients.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Prior art methods, such as those described in WO 95-04728, typically rely on a cumbersome sequence starting from hydroxyphenyl acetates. This conventional approach necessitates the protection of the phenolic hydroxyl group, often using dihydropyran to form a tetrahydropyranyl ether, followed by multiple functionalization steps including oximation, alkylation, and amidation. The critical bottleneck in this legacy workflow is the sheer number of unit operations required, each introducing potential yield losses and impurity carryover. Furthermore, the final deprotection step to remove the tetrahydropyranyl group requires acidic conditions that can compromise the stability of the sensitive oxime and dioxazine rings. These inefficiencies result in occasional low yields and a decisive adverse effect on the cost ratio, making the conventional route less attractive for commercial scale-up of complex pharmaceutical intermediates where margin compression is a constant pressure.
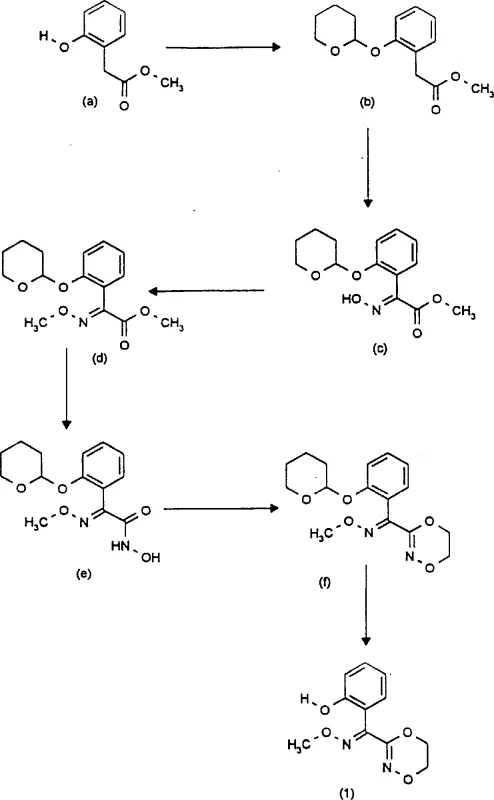
The Novel Approach
In stark contrast, the process disclosed in CN1184811A circumvents the need for protecting groups entirely by utilizing a direct rearrangement strategy. The core innovation lies in the conversion of O-hydroxyethyl-O'-alkyl-benzofurandione dioximes (Formula II) directly into the target 3-(1-hydroxyphenyl-1-alkoxyiminomethyl)dioxazines (Formula I). This transformation can be effected in the presence of diluents and bases or acids, often proceeding through a mixture of stereoisomers that are subsequently isomerized to the thermodynamically stable E-isomer. By eliminating the protection-deprotection cycle, the novel approach drastically simplifies the synthetic tree, reducing solvent consumption and waste generation. This streamlining not only enhances the overall throughput but also aligns with modern green chemistry principles, offering significant cost reduction in pharmaceutical intermediate manufacturing by minimizing raw material intensity and processing time.
Mechanistic Insights into Base-Catalyzed Rearrangement and Cyclization
The heart of this technology is the rearrangement of the benzofurandione dioxime scaffold. As illustrated in the general formulas, the reaction involves the cleavage of the benzofuran ring and the concurrent formation of the six-membered 1,4,2-dioxazine ring. When Process (a) is conducted in the presence of bases such as alkali metal hydroxides or alkoxides, the O-hydroxyethyl side chain participates in an intramolecular nucleophilic attack, facilitating the ring expansion. The reaction conditions are remarkably mild, typically ranging from 0°C to 80°C, which preserves the integrity of the labile N-O bonds. The ability to perform this rearrangement in aqueous or alcoholic media further underscores its practical utility, as it avoids the need for expensive anhydrous solvents. This mechanistic pathway ensures that the resulting product is obtained in high purity, often crystallizing directly from the reaction mixture, which simplifies downstream isolation and reduces the burden on purification infrastructure.
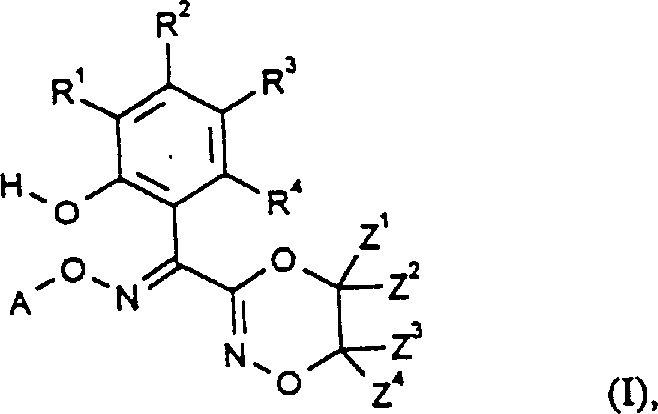
Impurity control is inherently built into this process design through the selective formation of the E-isomer. While the initial rearrangement may yield a mixture of E and Z stereoisomers, the patent details a highly effective isomerization protocol using mineral acids like hydrochloric acid in organic solvents. This step drives the equilibrium towards the desired E-configuration, which is crucial for the biological activity of the final antifungal compound. The process demonstrates surprising stability; contrary to literature expectations where benzofurandione monoximes might decompose into salicylic acid derivatives under basic conditions, the specific substitution pattern in Formula II directs the reaction exclusively towards the dioxazine product. This high chemoselectivity minimizes the formation of difficult-to-remove byproducts, ensuring that the final API intermediate meets stringent purity specifications required by regulatory bodies for pharmaceutical registration.
How to Synthesize 3-(1-Hydroxyphenyl-1-alkoxyiminomethyl)dioxazines Efficiently
The synthesis begins with the preparation of the key intermediate, O-alkyl-benzofurandione dioxime (Formula III), which can be accessed via several versatile routes including the cyclization of omega-nitro-2-hydroxyacetophenone oximes or the nitrosation of benzofuranone oximes. Once Formula III is secured, it is reacted with an ethane derivative, such as ethylene oxide, under basic conditions to install the hydroxyethyl group, yielding Formula II. The final and most critical step involves the base-catalyzed rearrangement of Formula II in water or alcohol to close the dioxazine ring. Detailed standardized synthetic steps for implementing this pathway in a GMP environment are provided below to assist R&D teams in rapid technology transfer.
- Prepare O-alkyl-benzofurandione dioxime (Formula III) via cyclization of omega-nitro-2-hydroxyacetophenone oxime or nitrosation of benzofuranone oxime.
- React Formula III with an ethane derivative like ethylene oxide under basic conditions to form O-hydroxyethyl-O'-alkyl-benzofurandione dioxime (Formula II).
- Perform the key rearrangement of Formula II using a base such as potassium hydroxide in aqueous or alcoholic media to yield the target dioxazine (Formula I).
Commercial Advantages for Procurement and Supply Chain Teams
For supply chain heads and procurement managers, the adoption of this novel synthesis route presents compelling economic and logistical benefits. The elimination of the tetrahydropyranyl protecting group removes the need for purchasing and handling dihydropyran, as well as the associated reagents for its removal, thereby simplifying the bill of materials. This reduction in chemical complexity translates directly into substantial cost savings by lowering the total volume of raw materials required per kilogram of finished product. Additionally, the process utilizes common, commodity-grade solvents like water, methanol, and ethyl acetate, which are readily available globally, mitigating the risk of supply disruptions associated with specialty solvents. The robustness of the reaction conditions allows for flexible manufacturing schedules, enhancing the reliability of delivery for high-purity pharmaceutical intermediates.
- Cost Reduction in Manufacturing: The streamlined process significantly lowers the cost of goods sold (COGS) by reducing the total number of synthetic steps. By avoiding the protection and deprotection sequences inherent in older methods, manufacturers save on reagent costs, solvent recovery expenses, and labor hours. The high yields reported in the patent examples, such as the 91.3% yield in the cyclization step, indicate a highly efficient use of starting materials. Furthermore, the ability to isolate products via simple filtration rather than complex chromatography reduces processing costs and equipment downtime, driving down the overall manufacturing expense without compromising quality.
- Enhanced Supply Chain Reliability: The starting materials for this process, including omega-nitro-2-hydroxyacetophenones and benzofuranones, are well-established chemicals with stable global supply chains. Unlike proprietary catalysts or exotic reagents that may face sourcing bottlenecks, these precursors can be procured from multiple vendors, ensuring continuity of supply. The process tolerance for water and common alcohols means that production is not dependent on strictly anhydrous conditions, reducing the risk of batch failures due to moisture sensitivity. This resilience makes the supply of these critical antifungal intermediates more predictable and secure for downstream drug manufacturers.
- Scalability and Environmental Compliance: The reaction conditions described, operating at atmospheric pressure and moderate temperatures, are inherently safer and easier to scale from pilot plant to commercial production. The use of aqueous workups and the generation of fewer organic waste streams align with increasingly strict environmental regulations. The process avoids the use of heavy metal catalysts or hazardous chlorinated solvents in key steps, simplifying waste treatment and disposal. This environmental compatibility not only reduces compliance costs but also supports the sustainability goals of modern pharmaceutical companies, making it an attractive option for long-term commercial partnerships.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this patented technology. These answers are derived directly from the experimental data and claims within CN1184811A, providing clarity on process feasibility and product quality. Understanding these details is vital for technical teams evaluating the integration of this route into existing manufacturing portfolios.
Q: What are the primary advantages of this new synthesis route over conventional methods?
A: The novel process eliminates the need for tetrahydropyranyl (THP) protecting groups, significantly reducing the number of synthetic steps and avoiding harsh deprotection conditions, which leads to higher overall yields and simplified purification.
Q: How is stereochemical purity managed in the production of these dioxazines?
A: The process allows for the formation of stereoisomer mixtures which can be efficiently isomerized to the desired E-isomer using acid catalysis, ensuring high stereochemical purity suitable for downstream pharmaceutical applications.
Q: Are the starting materials for this process commercially viable for large-scale production?
A: Yes, the key starting materials such as omega-nitro-2-hydroxyacetophenones and benzofuranones are well-known compounds that can be sourced readily or synthesized via established industrial methods, ensuring supply chain stability.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 3-(1-Hydroxyphenyl-1-alkoxyiminomethyl)dioxazine Supplier
At NINGBO INNO PHARMCHEM, we recognize the strategic importance of efficient intermediate synthesis in the competitive antifungal market. Our team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the theoretical benefits of patent CN1184811A are fully realized in practice. We operate stringent purity specifications and maintain rigorous QC labs to guarantee that every batch of 3-(1-hydroxyphenyl-1-alkoxyiminomethyl)dioxazine meets the exacting standards required for pharmaceutical grade applications. Our commitment to technical excellence allows us to navigate the complexities of rearrangement chemistry, delivering consistent quality that supports your regulatory filings and product launches.
We invite you to collaborate with us to optimize your supply chain for these critical intermediates. Contact our technical procurement team today to request a Customized Cost-Saving Analysis tailored to your specific volume requirements. We are prepared to provide specific COA data and route feasibility assessments to demonstrate how our manufacturing capabilities can enhance your project's economics and timeline. Let us be your partner in transforming innovative chemistry into commercial success.
