Scalable Synthesis of 1,1'-Deoxygossypol: A Cost-Effective Route for Anti-HIV Intermediates
Scalable Synthesis of 1,1'-Deoxygossypol: A Cost-Effective Route for Anti-HIV Intermediates
The pharmaceutical industry is constantly seeking robust, scalable pathways for complex bioactive molecules, particularly those with antiviral potential. A recent breakthrough detailed in patent CN112679319B introduces a highly efficient method for synthesizing 1,1'-deoxygossypol, a derivative of the natural polyphenol gossypol known for its anti-HIV activity. This innovation represents a paradigm shift from reliance on scarce, expensive synthetic precursors to utilizing abundant, renewable cottonseed derivatives. By re-engineering the synthetic backbone, the inventors have achieved a total yield of 45% across eight steps, effectively more than doubling the efficiency of previous literature methods which struggled to reach 16.9%. For R&D directors and procurement strategists, this development signals a new era of accessibility for this critical intermediate, promising not only economic advantages but also enhanced supply chain stability through the use of commoditized starting materials.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Prior to this invention, the synthesis of 1,1'-deoxygossypol was hindered by a convoluted and economically unviable route described in early medicinal chemistry literature (J. Med. Chem. 1995). The traditional approach relied heavily on 1-bromo-2-isopropyl-3,4-dimethoxybenzene as the foundational building block. This starting material is not only synthetically challenging to produce but also commands an exorbitant market price due to its scarcity, making large-scale production financially prohibitive. Furthermore, the legacy nine-step sequence necessitates the use of highly reactive and dangerous reagents such as n-BuLi, requiring stringent cryogenic control at temperatures as low as -78°C. Such harsh conditions impose severe limitations on reactor design and operational safety, creating significant bottlenecks for commercial scale-up. The cumulative effect of these factors results in a dismal overall yield, rendering the conventional method unsuitable for meeting the growing demand for anti-HIV research materials.
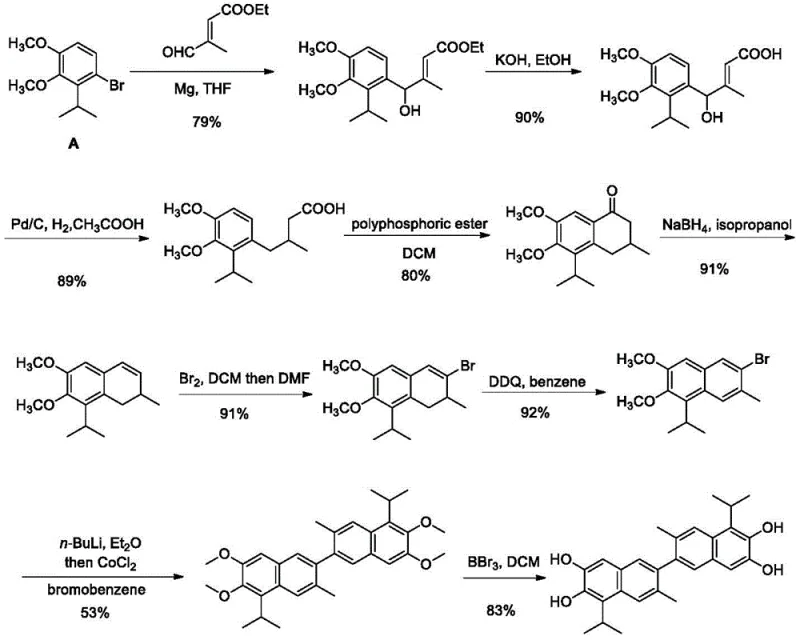
The Novel Approach
In stark contrast, the methodology disclosed in patent CN112679319B leverages gossypol acetate as the initial raw material, a compound derived directly from cottonseed processing. This strategic pivot transforms the economic landscape of the synthesis, replacing a rare, high-cost aromatic halide with a plentiful, low-cost agricultural byproduct. The new eight-step pathway is designed around a logical sequence of protection, functional group manipulation, and selective reduction, avoiding the need for extreme cryogenic temperatures. Instead, reactions proceed under mild thermal conditions ranging from room temperature to 90°C, utilizing standard laboratory equipment that is readily available in most pilot and production plants. This streamlined approach not only simplifies the operational workflow but also drastically reduces the energy consumption and safety risks associated with handling pyrophoric reagents, thereby establishing a far more sustainable and industrially viable manufacturing process.
Mechanistic Insights into Protective Group Strategy and Pd-Catalyzed Reduction
The core chemical ingenuity of this synthesis lies in its sophisticated management of the polyphenolic scaffold inherent to the gossypol structure. The process initiates with the removal of aldehyde groups from gossypol acetate using aqueous sodium hydroxide, followed by a global acetylation to protect all phenolic hydroxyls as acetates. This creates a stable intermediate (Compound III) that can withstand subsequent harsh conditions. A critical mechanistic step involves the selective deacetylation using potassium carbonate in a methanol/DCM mixture, which differentiates the hydroxyl groups based on their steric and electronic environments. Following this differentiation, specific phenolic positions are methylated to form methoxy ethers, while others are reserved for activation. The pivotal transformation occurs in the later stages where specific hydroxyl groups are converted into triflates (Compound VII) using triflic anhydride. These triflate groups serve as excellent leaving groups, enabling a palladium-catalyzed reductive cleavage using triethylsilane (Et3SiH) and a Pd(0) catalyst generated in situ. This step effectively removes the oxygen functionality at the 1,1' positions, achieving the "deoxy" status of the target molecule with high chemoselectivity.
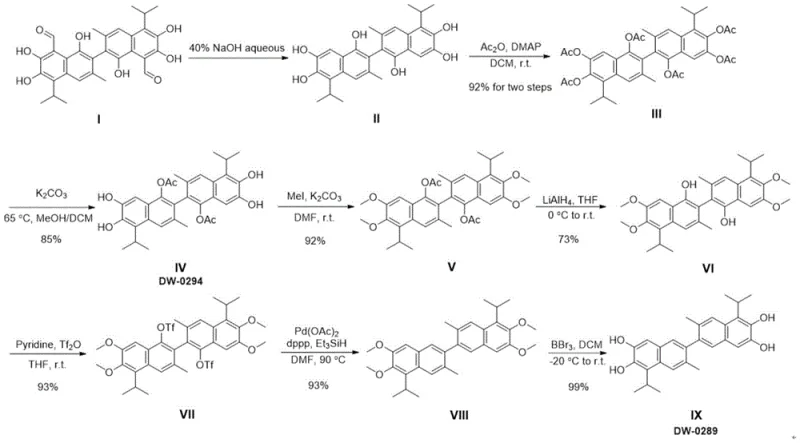
Impurity control is meticulously managed throughout this sequence through the strategic use of orthogonal protecting groups. The interplay between acetyl, methyl, and triflate groups allows chemists to direct reactivity to specific sites on the binaphthalene core without affecting neighboring functionalities. For instance, the use of lithium aluminum hydride (LAH) in tetrahydrofuran (THF) is carefully controlled at 0°C to reduce ester functionalities to alcohols without disrupting the methyl ethers. This precision minimizes the formation of side products such as over-reduced species or scrambled isomers, which are common pitfalls in polyphenol synthesis. The final demethylation step utilizes boron tribromide (BBr3) to reveal the free phenolic hydroxyls, yielding the target 1,1'-deoxygossypol (Compound IX) as a high-purity gray solid. The robustness of this mechanism ensures that the impurity profile remains clean, facilitating easier downstream purification and ensuring the material meets the stringent specifications required for biological testing.
How to Synthesize 1,1'-Deoxygossypol Efficiently
The synthesis of 1,1'-deoxygossypol via this patented route offers a clear, step-by-step protocol that balances chemical efficiency with operational simplicity. The process begins with the conversion of gossypol acetate to apogossypol, followed by a series of protection and activation steps that culminate in a palladium-catalyzed reduction. Each stage has been optimized to maximize yield and minimize waste, making it an ideal candidate for technology transfer. While the detailed experimental conditions regarding stoichiometry and workup procedures are extensive, the general workflow provides a reliable blueprint for producing this valuable intermediate. For a comprehensive breakdown of the specific reaction parameters, temperatures, and isolation techniques required to replicate this success, please refer to the standardized synthesis guide below.
- Convert gossypol acetate to apogossypol via alkaline de-aldehydation, followed by per-acetylation to protect phenolic hydroxyls.
- Perform selective deacetylation and methylation to differentiate hydroxyl groups, preparing the scaffold for activation.
- Activate phenolic positions with triflic anhydride, followed by palladium-catalyzed reductive removal of the triflate groups and final demethylation.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the adoption of this synthesis route offers transformative benefits that extend far beyond simple chemistry. The most immediate impact is seen in the drastic reduction of raw material costs. By shifting the supply base from specialized, imported aromatic halides to domestically available cottonseed derivatives, manufacturers can insulate themselves from the volatility of the fine chemical market. This localization of the supply chain significantly enhances reliability, ensuring that production schedules are not disrupted by the scarcity of niche reagents. Furthermore, the elimination of cryogenic steps reduces the capital expenditure required for specialized cooling infrastructure, allowing existing facilities to adapt to this process with minimal retrofitting. These factors combine to create a leaner, more agile manufacturing model that is better equipped to respond to fluctuating market demands.
- Cost Reduction in Manufacturing: The substitution of expensive starting materials with gossypol acetate fundamentally alters the cost structure of the final product. Since gossypol acetate is a byproduct of the massive cottonseed oil industry, its availability is virtually limitless and its price point is a fraction of synthetic alternatives. Additionally, the higher overall yield of 45% means that less raw material is wasted per kilogram of product, further driving down the cost of goods sold (COGS). The avoidance of exotic reagents like n-BuLi also eliminates the need for costly quenching and disposal protocols associated with hazardous waste, contributing to substantial operational savings.
- Enhanced Supply Chain Reliability: Relying on agricultural byproducts rather than multi-step synthetic intermediates diversifies the supply risk profile. Cottonseed is a globally traded commodity, ensuring a consistent and predictable flow of raw materials regardless of geopolitical tensions or synthetic capacity constraints. This stability is crucial for long-term project planning and securing supply contracts with major pharmaceutical partners. Moreover, the simplified reaction conditions reduce the likelihood of batch failures due to equipment malfunction or operator error, leading to more consistent delivery times and improved customer satisfaction.
- Scalability and Environmental Compliance: The mild reaction conditions and the use of common solvents like DCM, THF, and ethyl acetate make this process inherently scalable from gram to ton quantities. The absence of extreme temperatures and pyrophoric reagents simplifies the safety assessment and regulatory approval process for new manufacturing sites. From an environmental perspective, the higher atom economy and reduced waste generation align with green chemistry principles, helping companies meet increasingly strict sustainability targets. This eco-friendly profile not only reduces disposal costs but also enhances the brand reputation of the manufacturer as a responsible partner in the pharmaceutical value chain.
Frequently Asked Questions (FAQ)
Understanding the technical nuances of this new synthesis method is essential for stakeholders evaluating its potential for integration into their supply chains. The following questions address common concerns regarding scalability, purity, and regulatory compliance, drawing directly from the data provided in the patent documentation. These insights are intended to clarify the practical implications of adopting this technology and to assist decision-makers in assessing its fit for their specific operational needs.
Q: What is the primary advantage of using gossypol acetate over traditional starting materials?
A: Gossypol acetate is a abundant, low-cost byproduct of the cottonseed oil industry, whereas traditional routes rely on scarce and expensive synthetic bromobenzenes, drastically reducing raw material costs.
Q: How does the new synthesis route improve overall yield compared to literature methods?
A: The patented 8-step sequence achieves a total yield of approximately 45%, which is a substantial improvement over the previously reported 16.9% yield from the 9-step Grignard-based route.
Q: Does this process require hazardous cryogenic conditions?
A: No, unlike the prior art which requires n-BuLi at -78°C, this method operates under mild conditions (mostly 0°C to 90°C), significantly enhancing operational safety and ease of scale-up.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 1,1'-Deoxygossypol Supplier
The successful implementation of complex synthetic routes like the one described in CN112679319B requires a partner with deep technical expertise and a proven track record in process development. At NINGBO INNO PHARMCHEM, we possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that your transition from lab bench to market is seamless. Our state-of-the-art facilities are equipped with rigorous QC labs capable of verifying stringent purity specifications for sensitive polyphenolic intermediates, guaranteeing that every batch meets the highest international standards. We understand the critical nature of antiviral research and are committed to providing a supply of 1,1'-deoxygossypol that supports your scientific breakthroughs without compromise.
We invite you to collaborate with us to unlock the full potential of this cost-effective synthesis. Our technical team is ready to provide a Customized Cost-Saving Analysis tailored to your specific volume requirements, demonstrating exactly how this new route can optimize your budget. Please contact our technical procurement team today to request specific COA data and comprehensive route feasibility assessments. Let us help you secure a sustainable, high-quality supply of 1,1'-deoxygossypol that drives your projects forward with confidence and efficiency.
