Advanced 3-Step Synthesis of 2-Acylamino-3-Biphenylpropionic Acid for Commercial Scale-Up
Advanced 3-Step Synthesis of 2-Acylamino-3-Biphenylpropionic Acid for Commercial Scale-Up
The pharmaceutical industry constantly seeks more efficient pathways for producing complex amino acid derivatives, particularly those serving as critical building blocks for peptide therapeutics. Patent CN101555211B introduces a groundbreaking chemical synthesis method for 2-acylamino-3-biphenylpropionic acid, a compound of significant value in the production of advanced drugs such as octreotide. This innovation addresses long-standing inefficiencies in traditional manufacturing by streamlining the reaction sequence from four steps down to three, thereby enhancing both economic viability and operational simplicity. The structural versatility of this molecule, as depicted in the general formula below, allows for various R-group modifications, making it a highly adaptable intermediate for diverse medicinal chemistry applications.
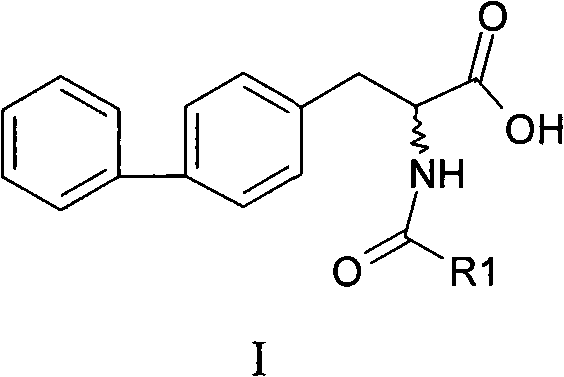
For R&D directors and process chemists, the implications of this patent are profound, offering a route that not only improves yield but also simplifies purification protocols. By leveraging robust organic transformations such as Schiff base alkylation and selective hydrolysis, the method ensures high product purity without the need for exotic catalysts. This technical advancement positions the compound as a reliable candidate for large-scale commercial production, meeting the stringent quality standards required by global regulatory bodies for active pharmaceutical ingredient (API) precursors.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of 2-acylamino-3-biphenylpropionic acid has been plagued by inefficient multi-step sequences that detrimentally impact overall process economics. As illustrated in the prior art reaction scheme, traditional methods often necessitate a four-step pathway where the acyl protecting group must be laboriously removed in an intermediate stage only to be re-introduced later in the synthesis. This redundant manipulation of functional groups not only extends the production timeline but also exposes the sensitive molecular scaffold to multiple potential degradation pathways, resulting in significant material loss at each stage.
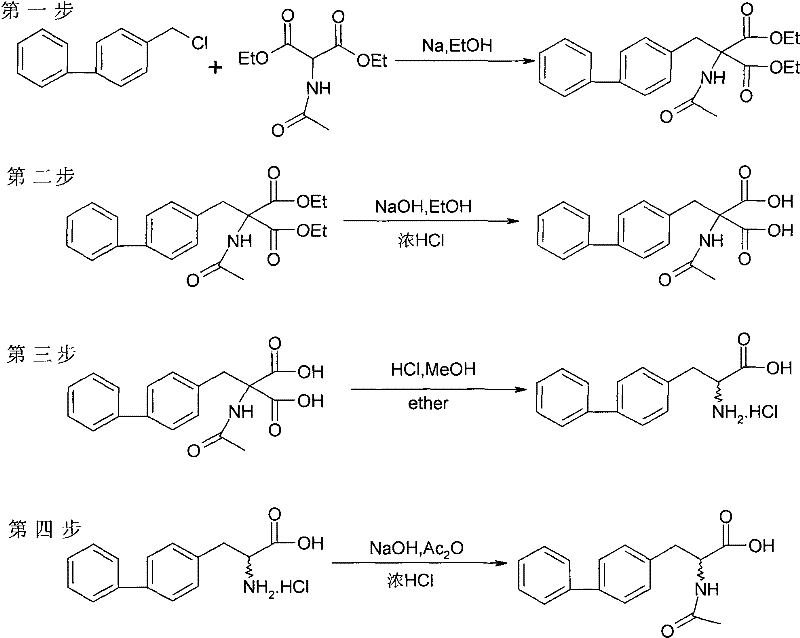
Furthermore, the reliance on numerous reagents and solvents in these legacy processes complicates waste management and increases the environmental footprint of the manufacturing operation. The cumulative yield of such conventional routes typically hovers around 64%, which is suboptimal for high-value pharmaceutical intermediates where margin compression is a constant concern. The operational complexity associated with isolating and purifying intermediates after every single transformation adds substantial labor and equipment costs, rendering these older methods less competitive in a market that demands rapid, cost-effective supply chain solutions.
The Novel Approach
In stark contrast, the novel methodology disclosed in the patent revolutionizes the production landscape by condensing the synthesis into a concise three-step sequence that maximizes atom economy and operational efficiency. The new route, shown in the comprehensive reaction diagram below, strategically bypasses the unnecessary deprotection-reprotection cycle, allowing the acyl group to remain intact throughout the critical carbon-carbon bond-forming stages. This strategic simplification directly translates to a dramatic improvement in overall yield, pushing performance metrics to over 88%, which represents a substantial gain in productivity for any manufacturing facility.
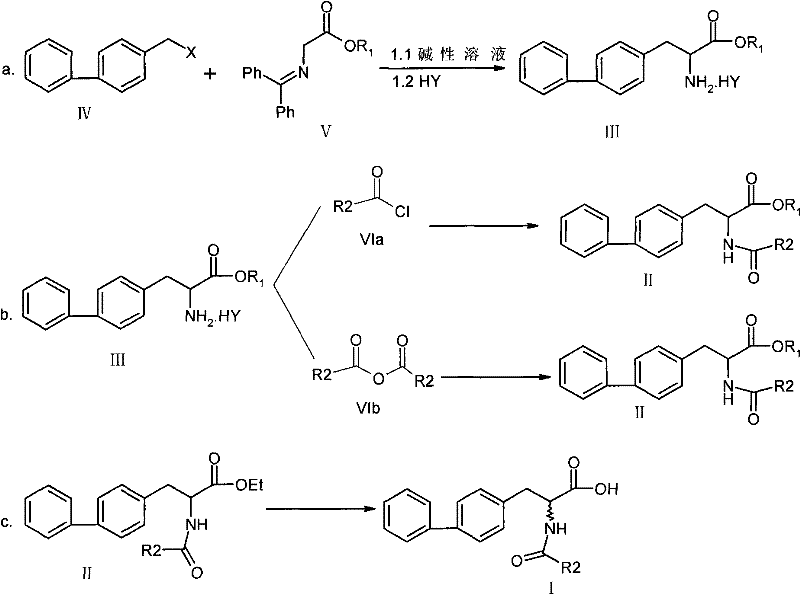
Beyond yield improvements, this approach utilizes widely available and cost-effective reagents such as simple inorganic bases and common organic solvents, eliminating the dependency on expensive or hazardous specialty chemicals. The streamlined workflow reduces the number of isolation and purification events, which not only lowers solvent consumption but also minimizes the risk of cross-contamination between batches. For procurement managers, this translates to a more stable and predictable supply of raw materials, while for supply chain heads, it意味着 a faster time-to-market for the final drug product due to reduced manufacturing lead times.
Mechanistic Insights into Schiff Base Alkylation and Hydrolysis
The core of this synthetic breakthrough lies in the elegant utilization of a Schiff base derivative, specifically N-(diphenylmethylenyl)aminoacetate, as a glycine equivalent for the alkylation step. In the initial phase, the methylene protons of the Schiff base are activated by a mild base, generating a nucleophilic enolate species that attacks the electrophilic benzylic carbon of the biphenyl halide. This carbon-carbon bond formation is highly selective and proceeds under relatively mild thermal conditions, preserving the integrity of the sensitive biphenyl moiety. The subsequent acidic hydrolysis cleaves the imine bond to reveal the free amine, which is immediately captured as a stable salt, preventing racemization and ensuring high optical purity of the resulting amino acid ester.
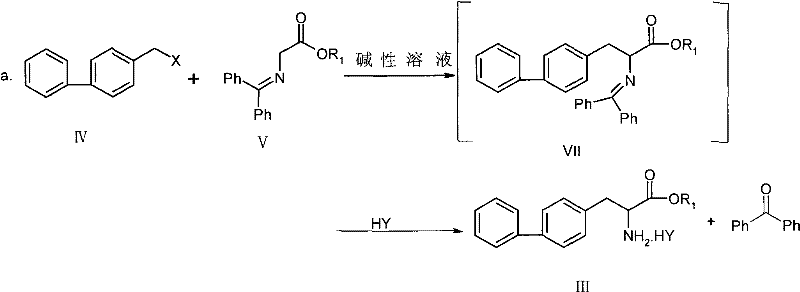
Impurity control is inherently built into this mechanism due to the high chemoselectivity of the reagents employed. The use of specific molar ratios of base to substrate ensures complete conversion of the starting material while minimizing side reactions such as elimination or over-alkylation. Furthermore, the choice of acid for the hydrolysis step, such as hydrochloric acid or oxalic acid, allows for the precipitation of the intermediate salt in high purity, effectively filtering out non-basic organic impurities before they can propagate through the subsequent acylation and hydrolysis steps. This rigorous control over the reaction pathway ensures that the final API intermediate meets the strict impurity profiles required for downstream peptide synthesis.
How to Synthesize 2-Acylamino-3-Biphenylpropionic Acid Efficiently
Implementing this synthesis requires precise control over reaction parameters to achieve the reported high yields and purity levels. The process begins with the alkylation of the Schiff base in a suitable solvent like toluene or DMF, followed by careful pH adjustment to isolate the amino ester salt. The second stage involves acylation using acid chlorides or anhydrides in the presence of a tertiary amine base, a standard procedure that benefits from the high reactivity of the free amine generated in situ. Finally, the ester is hydrolyzed under alkaline conditions to yield the target acid, which precipitates upon acidification.
- Step 1: Alkylation of N-(diphenylmethylenyl)aminoacetate with 4-halomethylbiphenyl under basic conditions, followed by acid hydrolysis to form the amino acid ester salt.
- Step 2: Acylation of the amino acid ester salt using acid chlorides or anhydrides in the presence of a base to form the protected ester intermediate.
- Step 3: Alkaline hydrolysis of the ester intermediate to yield the final 2-acylamino-3-biphenylpropionic acid product with high purity.
Commercial Advantages for Procurement and Supply Chain Teams
From a commercial perspective, the adoption of this patented synthesis route offers transformative benefits for procurement strategies and supply chain resilience. The drastic reduction in synthetic steps directly correlates to a significant decrease in manufacturing costs, as fewer unit operations mean lower energy consumption, reduced labor hours, and diminished solvent usage. By eliminating the need for specialized catalysts and complex protection group chemistry, the process relies on commodity chemicals that are readily available in the global market, shielding the supply chain from volatility associated with niche reagent sourcing.
- Cost Reduction in Manufacturing: The elimination of redundant chemical steps and the use of inexpensive, bulk-available reagents like sodium hydroxide and hydrochloric acid fundamentally lower the cost of goods sold. The increase in overall yield from roughly 64% to over 88% means that less raw material is required to produce the same amount of final product, effectively maximizing the value extracted from every kilogram of input. This efficiency gain allows for more competitive pricing structures without compromising on quality margins.
- Enhanced Supply Chain Reliability: Simplifying the synthesis to three robust steps reduces the number of potential failure points in the manufacturing process, leading to higher batch success rates and more consistent delivery schedules. The reliance on stable, non-hazardous solvents and reagents minimizes regulatory hurdles related to transportation and storage, ensuring a smoother flow of materials through the logistics network. This reliability is crucial for maintaining continuous production lines for downstream peptide drugs.
- Scalability and Environmental Compliance: The process is designed with scalability in mind, utilizing reaction conditions that are easily transferable from laboratory to pilot and commercial scales. The reduction in waste generation, driven by higher yields and fewer purification steps, aligns with modern green chemistry principles and helps manufacturers meet increasingly stringent environmental regulations. This sustainability aspect not only reduces disposal costs but also enhances the corporate social responsibility profile of the supply chain.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the synthesis and application of 2-acylamino-3-biphenylpropionic acid. These insights are derived directly from the technical specifications and experimental data provided in the patent documentation, ensuring accuracy and relevance for industry professionals seeking to implement this technology.
Q: How does the new synthesis route improve yield compared to traditional methods?
A: The patented method increases overall yield from approximately 64% in conventional four-step routes to over 88% by eliminating unnecessary deprotection and re-protection steps, significantly reducing material loss.
Q: What are the key advantages for large-scale manufacturing of this intermediate?
A: The process utilizes common, inexpensive reagents like sodium hydroxide and hydrochloric acid, avoids complex transition metal catalysts, and shortens the reaction sequence, making it highly suitable for industrial scale-up.
Q: Is this intermediate suitable for synthesizing specific peptide drugs?
A: Yes, the resulting 2-acylamino-3-biphenylpropionic acid is a critical chiral building block used in the synthesis of polypeptides and amino acid-based pharmaceuticals, including anti-tumor agents like octreotide.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 2-Acylamino-3-Biphenylpropionic Acid Supplier
At NINGBO INNO PHARMCHEM, we understand the critical role that high-quality intermediates play in the successful development of life-saving medications. Our team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that your project transitions smoothly from benchtop discovery to full-scale manufacturing. We adhere to stringent purity specifications and operate rigorous QC labs to guarantee that every batch of 2-acylamino-3-biphenylpropionic acid meets the exacting standards required for peptide synthesis and pharmaceutical applications.
We invite you to collaborate with us to leverage this advanced synthesis technology for your specific needs. Please contact our technical procurement team to request a Customized Cost-Saving Analysis tailored to your volume requirements. We are ready to provide specific COA data and detailed route feasibility assessments to demonstrate how our optimized manufacturing processes can drive efficiency and reliability in your supply chain.
