Advanced One-Step Synthesis and Chiral Resolution of 2-Acylamino-3-Biphenylpropionic Acid for Commercial Scale-Up
The pharmaceutical industry continuously seeks efficient pathways for synthesizing complex amino acid derivatives, particularly those serving as critical building blocks for peptide hormones. Patent CN101774941A introduces a groundbreaking methodology for the preparation and resolution of 2-acylamino-3-biphenylpropionic acid, a pivotal intermediate in the synthesis of Luteinizing Hormone-Releasing Hormone (LHRH) analogs. This compound, characterized by its biphenyl moiety and protected amino acid structure, has historically presented significant synthetic challenges regarding yield and stereochemical purity. The disclosed innovation offers a streamlined alternative that bypasses traditional bottlenecks, providing a robust foundation for cost reduction in pharmaceutical intermediate manufacturing. By leveraging a direct thermal decarboxylation strategy coupled with an optimized chemical resolution process, this technology addresses the urgent need for reliable supply chains in the development of next-generation therapeutic agents.
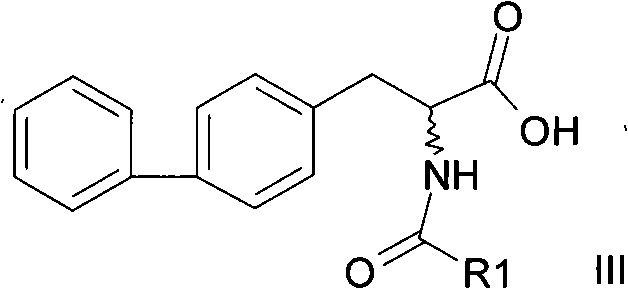
The structural versatility of this molecule allows for various acyl substitutions, yet the core challenge remains the efficient construction of the chiral center adjacent to the biphenyl system. As a reliable pharmaceutical intermediate supplier, understanding the nuances of such transformations is essential for delivering high-quality materials to global partners. The patent details a comprehensive approach that not only simplifies the synthetic route but also enhances the overall economic viability of producing these high-value compounds. This report delves into the technical specifics, comparing legacy methods with this novel approach to highlight the tangible benefits for R&D and procurement teams aiming to optimize their production pipelines.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Prior art, specifically the methods described in literature such as Yabe et al. (Chemical & Pharmaceutical Bulletin, 1976), relied on a convoluted synthetic sequence that severely impacted efficiency and cost. The traditional route involved the synthesis of 2-acetylamino-2-(4-phenylbenzyl)malonic acid followed by a problematic conversion to the target propionic acid derivative. A critical flaw in this legacy process was the lack of reagent selectivity during the initial transformation steps. To achieve the desired product, manufacturers were forced to employ a protective group strategy where the acetyl group was first removed and then laboriously re-added. This "deprotection-reprotection" cycle necessitated additional reaction vessels, increased solvent consumption, and extended processing times, all of which contributed to inflated production costs and reduced overall throughput.
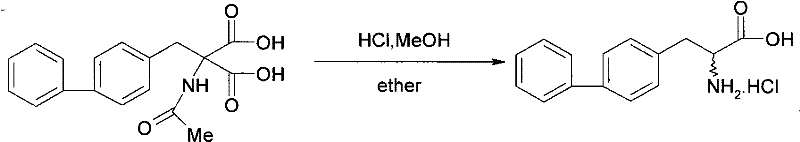
Furthermore, the conventional resolution methods often utilized enzymatic catalysts, which, while selective, introduced significant supply chain vulnerabilities. Enzymes can be expensive, sensitive to batch variations, and difficult to source in the quantities required for commercial scale-up of complex pharmaceutical intermediates. The patent documentation highlights that in these older methods, the acetylamino group was prone to hydrolysis under the harsh conditions required for resolution, leading to impurity profiles that were difficult to manage. This susceptibility to degradation meant that rigorous purification steps were mandatory, further eroding yield and increasing the environmental footprint of the manufacturing process. For procurement managers, these inefficiencies translate directly into higher raw material costs and less predictable delivery schedules.
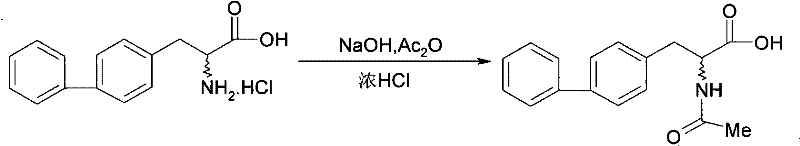
The Novel Approach
In stark contrast, the methodology outlined in CN101774941A presents a paradigm shift by utilizing a direct thermal decarboxylation pathway. This innovative approach allows for the conversion of 2-acylamino-2-(4-phenylbenzyl)malonic acid directly into 2-acylamino-3-biphenylpropionic acid in a single step. By simply heating the precursor, potentially in the presence of benign solvents like water or xylene, the reaction proceeds with high selectivity and minimal byproduct formation. This elimination of the deprotection-reprotection cycle drastically shortens the synthetic route, reducing the number of unit operations and the associated labor and utility costs. The ability to perform this transformation under relatively mild conditions (80°C to 250°C) ensures that the sensitive amide bond remains intact, preserving the integrity of the molecule throughout the synthesis.
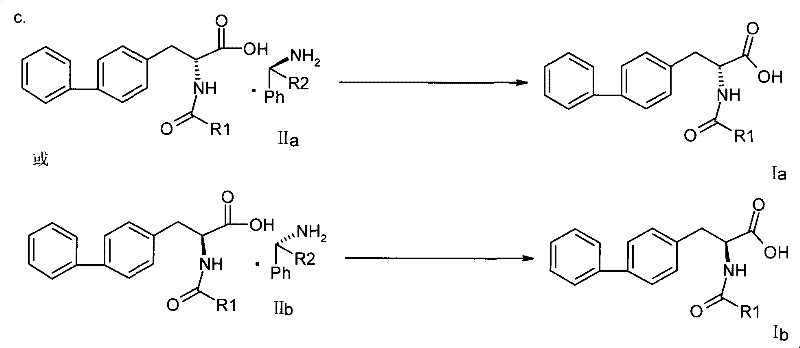
Moreover, the resolution strategy employed in this new method replaces fragile enzymatic processes with robust chemical resolution using chiral amines. By forming diastereomeric salts with resolving agents such as phenylethylamine derivatives, the process achieves high optical purity through straightforward crystallization techniques. This chemical approach is far more scalable and controllable than enzymatic methods, offering consistent results across different batch sizes. The patent demonstrates that this method can accommodate various acyl groups (R1) without compromising the reaction efficiency, making it a versatile platform technology. For supply chain heads, this translates to a more resilient manufacturing process that is less susceptible to biological variability and easier to validate under Good Manufacturing Practice (GMP) standards.
Mechanistic Insights into Thermal Decarboxylation and Chiral Resolution
The core of this technological advancement lies in the mechanistic elegance of the thermal decarboxylation step. The precursor, a malonic acid derivative, possesses a geminal dicarboxylic acid structure that is inherently unstable upon heating. When subjected to elevated temperatures, the molecule undergoes a concerted pericyclic reaction where one carboxyl group is expelled as carbon dioxide. This process is driven by the formation of a stable enol intermediate, which subsequently tautomerizes to the more thermodynamically stable keto form, yielding the target 2-acylamino-3-biphenylpropionic acid. The presence of the electron-withdrawing acylamino group at the alpha position facilitates this decarboxylation by stabilizing the transition state, allowing the reaction to proceed efficiently even in aqueous media. This mechanistic pathway avoids the need for aggressive chemical reagents that could otherwise attack the biphenyl system or the amide linkage.
Following the synthesis of the racemic acid, the resolution mechanism relies on the principle of diastereomeric salt formation. The racemic mixture interacts with a single enantiomer of a chiral amine resolving agent, such as (S)-1-phenylethylamine. Due to the spatial differences between the resulting diastereomeric salts, their physical properties, particularly solubility, differ significantly in specific solvent systems like ethanol or methanol. By carefully controlling the temperature and solvent composition, one diastereomer preferentially crystallizes out of the solution while the other remains in the mother liquor. This fractional crystallization effectively separates the enantiomers. Subsequent treatment with a mineral or organic acid protonates the amine, releasing the free chiral acid and regenerating the resolving agent salt. This chemical liberation step is crucial as it occurs under mild acidic conditions that do not hydrolyze the acyl group, ensuring the final product retains its intended structure and high purity.
How to Synthesize 2-Acylamino-3-Biphenylpropionic Acid Efficiently
Implementing this synthesis requires precise control over thermal parameters and crystallization conditions to maximize yield and optical purity. The process begins with the selection of the appropriate malonic acid precursor, followed by a controlled heating phase that can be conducted neat or in solvent depending on the specific derivative. Once the racemic acid is obtained, the focus shifts to the resolution stage, where the choice of resolving agent and solvent ratio becomes critical for achieving the desired enantiomeric excess. The following guide outlines the standardized operational framework derived from the patent examples, providing a clear roadmap for laboratory and pilot-scale execution.
- Perform thermal decarboxylation of 2-acylamino-2-(4-phenylbenzyl)malonic acid at 80°C to 250°C, optionally in solvents like water or xylene, to obtain the racemic acid.
- Conduct chiral resolution by reacting the racemic acid with a chiral amine resolving agent (e.g., phenylethylamine derivatives) in alcohol solvents to form diastereomeric salts.
- Treat the isolated chiral salt with an acidic reagent such as hydrochloric acid or acetic acid to liberate the final optically pure 2-acylamino-3-biphenylpropionic acid.
Commercial Advantages for Procurement and Supply Chain Teams
For organizations focused on the bottom line and supply security, the adoption of this novel synthesis route offers profound strategic advantages. The primary value driver is the significant simplification of the manufacturing workflow. By collapsing a multi-step sequence into a single thermal transformation, the process inherently reduces the consumption of raw materials, solvents, and energy. This reduction in material intensity directly correlates to a lower cost of goods sold (COGS), allowing for more competitive pricing in the global market. Furthermore, the elimination of enzymatic steps removes a major variable from the supply chain, mitigating the risks associated with biological reagent availability and stability. This robustness ensures a more predictable production schedule, which is vital for maintaining continuity in the supply of critical drug intermediates.
- Cost Reduction in Manufacturing: The streamlined nature of this process eliminates the need for expensive protecting group manipulations and enzymatic catalysts. By removing the deprotection and reprotection steps, manufacturers save substantially on reagent costs and waste disposal fees. The use of common, inexpensive solvents like water, ethanol, and xylene further drives down operational expenses. Additionally, the high yields reported in the patent examples indicate minimal material loss, maximizing the output from every kilogram of starting material. This efficiency creates a leaner manufacturing model that is highly resistant to fluctuations in raw material pricing, providing long-term financial stability for procurement budgets.
- Enhanced Supply Chain Reliability: The reliance on standard chemical reagents and thermal processes rather than specialized biological enzymes significantly enhances supply chain resilience. Chemical resolving agents like phenylethylamine derivatives are commodity chemicals with stable global supply networks, unlike niche enzymes that may have long lead times. The simplicity of the equipment required—essentially reactors capable of heating and filtration units—means that production can be easily transferred between facilities or scaled up without extensive capital investment. This flexibility ensures that supply can be ramped up quickly to meet surging demand, reducing lead times for high-purity pharmaceutical intermediates and preventing stockouts.
- Scalability and Environmental Compliance: From an environmental and regulatory perspective, this method is superior due to its reduced waste generation. Fewer reaction steps mean fewer workup procedures and less solvent waste to treat. The ability to use water as a solvent in the decarboxylation step aligns with green chemistry principles, reducing the volatile organic compound (VOC) emissions associated with organic solvents. The high selectivity of the reaction minimizes the formation of hazardous byproducts, simplifying effluent treatment. These factors make the process easier to permit and operate within strict environmental regulations, facilitating smoother scale-up from pilot batches to multi-ton commercial production without encountering regulatory bottlenecks.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this synthesis technology. These insights are derived directly from the experimental data and beneficial effects described in the patent documentation, providing clarity on how this method compares to existing standards. Understanding these details is crucial for technical teams evaluating the feasibility of adopting this route for their specific product portfolios.
Q: How does this new method improve upon the conventional Yabe synthesis route?
A: The conventional route requires a multi-step process involving the removal and subsequent re-addition of the acetyl protecting group due to poor selectivity. The novel method described in CN101774941A achieves the transformation in a single thermal decarboxylation step, significantly reducing reagent consumption and operational complexity.
Q: What are the advantages of the chemical resolution method over enzymatic methods?
A: Unlike enzymatic resolution which can be costly and sensitive to reaction conditions, this patent utilizes robust chemical resolution with chiral amines. This approach avoids the hydrolysis of the amide bond often seen in enzymatic processes, ensuring higher product integrity and lower production costs.
Q: Is this process suitable for large-scale industrial production?
A: Yes, the process is highly scalable. It relies on standard unit operations such as heating, reflux, filtration, and crystallization. The use of common solvents like water, ethanol, and xylene, combined with high yields (up to 95%), makes it ideal for commercial manufacturing.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 2-Acylamino-3-Biphenylpropionic Acid Supplier
The technical superiority of the synthesis method described in CN101774941A represents a significant opportunity for pharmaceutical companies to optimize their supply chains. At NINGBO INNO PHARMCHEM, we possess the technical expertise and infrastructure to leverage this advanced chemistry for your specific needs. Our team has extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the transition from laboratory discovery to industrial reality is seamless. We maintain stringent purity specifications and operate rigorous QC labs to guarantee that every batch of 2-acylamino-3-biphenylpropionic acid meets the highest international standards, supporting your regulatory filings and clinical trials with confidence.
We invite you to collaborate with us to unlock the full potential of this efficient manufacturing route. By partnering with our technical procurement team, you can request a Customized Cost-Saving Analysis tailored to your volume requirements. We encourage you to reach out today to obtain specific COA data and discuss route feasibility assessments that can demonstrate exactly how this technology can reduce your costs and secure your supply of this critical pharmaceutical intermediate.
