Advanced Purification Technology for High-Purity Dapagliflozin Intermediates
Advanced Purification Technology for High-Purity Dapagliflozin Intermediates
The global demand for SGLT2 inhibitors continues to surge as type 2 diabetes prevalence rises, placing immense pressure on supply chains to deliver high-quality active pharmaceutical ingredients efficiently. Patent CN109705075B introduces a transformative purification methodology for Dapagliflozin that bypasses the traditional bottlenecks of chemical derivatization. Unlike conventional approaches that rely on complex protection and deprotection sequences, this innovation utilizes a sophisticated physicochemical separation strategy based on selective crystallization and solvent partitioning. By leveraging specific hydrogen-bonding interactions between polar reagents and the hydroxyl groups of the target molecule, the process achieves exceptional purity levels exceeding 98.0% in a single pass, with the potential to reach over 99.5% upon repetition. This technical breakthrough represents a significant paradigm shift for manufacturers seeking to optimize their production lines for this critical antidiabetic agent.
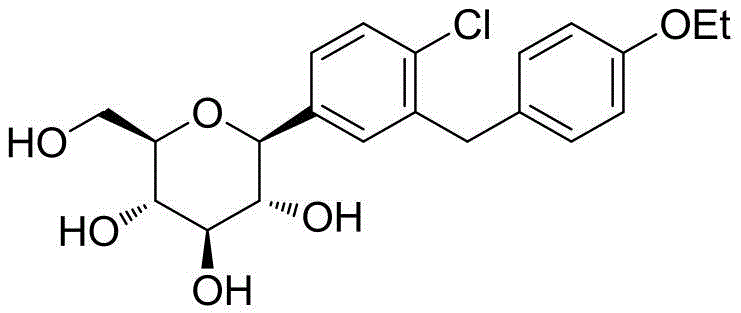
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historical purification protocols for Dapagliflozin, as referenced in prior art such as CN101092409A and CN101628905B, are plagued by inherent inefficiencies stemming from their reliance on chemical modification. These legacy methods typically involve converting the crude tetrahydroxy product into a tetra-acetyl derivative using acetic anhydride, catalyzed by reagents like DMAP and bases such as DIEPA. Following recrystallization of this protected intermediate, a harsh hydrolysis step using lithium hydroxide is required to regenerate the free hydroxyl groups. This multi-step sequence not only consumes substantial quantities of expensive reagents and solvents but also introduces significant yield losses, often resulting in overall recovery rates of less than 50%. Furthermore, the use of column chromatography in some variations imposes severe limitations on throughput and generates excessive hazardous waste, rendering these methods economically and environmentally unsustainable for modern large-scale manufacturing.
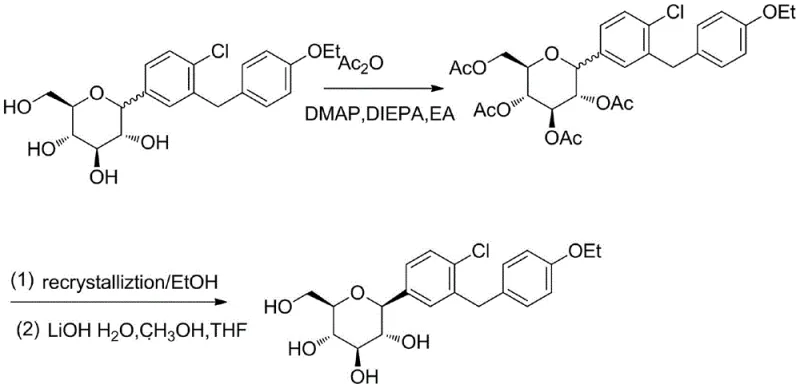
The Novel Approach
In stark contrast to the cumbersome derivatization pathways of the past, the method disclosed in CN109705075B streamlines the purification workflow into a direct, non-destructive physical process. The core innovation lies in the strategic use of water-immiscible organic solvents combined with specific polar reagents that facilitate hydrogen bonding. Instead of chemically altering the molecule to change its solubility profile, this approach manipulates the solvation environment to selectively precipitate the pure product while leaving impurities in the solution phase. This elimination of protection and deprotection steps drastically shortens the processing time and reduces the consumption of auxiliary chemicals. The result is a robust, scalable protocol that maintains the structural integrity of the sensitive glucoside moiety while delivering a refined product with superior purity profiles, effectively solving the long-standing challenge of purifying low-melting-point glycosides without resorting to chromatography.
Mechanistic Insights into Selective Crystallization and Extraction
The efficacy of this purification strategy is rooted in the precise manipulation of intermolecular forces and solubility parameters. In the first critical stage, the crude Dapagliflozin is dissolved in a water-immiscible organic solvent, such as ethyl acetate or toluene, creating a homogeneous phase. Upon the addition of a polar reagent—specifically those capable of forming strong hydrogen bonds with hydroxyl groups, such as propylene glycol or ethanol—the solubility equilibrium is disrupted. The polar reagent interacts preferentially with the hydroxyl groups of the Dapagliflozin, promoting the formation of a stable crystal lattice that excludes non-polar or structurally dissimilar impurities. These impurities, lacking the specific interaction capability or possessing different solubility characteristics, remain dissolved in the mother liquor, allowing for their removal via simple filtration. This mechanism ensures that the initial solid obtained is already significantly enriched in the target compound, setting the foundation for high final purity.
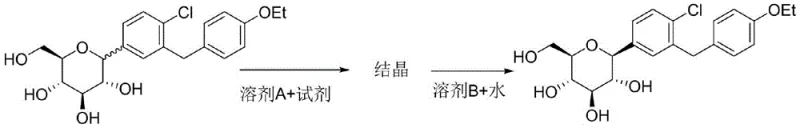
Following the initial crystallization, the process employs a secondary purification mechanism through liquid-liquid extraction to remove residual polar reagents and trace contaminants. The filtered solid is redissolved in a second water-immiscible organic solvent, and an aqueous phase containing a controlled amount of the polar reagent is introduced. This step is crucial for mass transfer; the polar reagent, now dissolved in the water, acts as a carrier to extract any remaining hydrophilic impurities or excess crystallization agents from the organic phase into the aqueous layer. Since Dapagliflozin prefers the organic phase under these specific conditions, it remains in the solvent while the unwanted polar species are washed away. The final concentration of the organic phase yields the refined product. This dual-stage mechanism—crystallization followed by partitioning—provides a synergistic effect that rigorously controls the impurity profile, ensuring the final material meets stringent pharmaceutical specifications without the need for destructive chemical reactions.
How to Synthesize Dapagliflozin Efficiently
The implementation of this purification technology requires careful control of solvent ratios, temperature, and stirring dynamics to maximize yield and purity. The patent outlines a reproducible protocol that begins with the dissolution of crude material in solvents like ethyl acetate or methyl tert-butyl ether, followed by the controlled addition of polar agents such as 1,2-propylene glycol. The detailed standardized synthesis steps, including specific volume-to-mass ratios and temperature ranges for optimal crystal growth, are provided in the technical guide below to ensure consistent results across different production batches.
- Dissolve crude dapagliflozin in a water-immiscible organic solvent (Solvent A) and add a polar reagent capable of hydrogen bonding to induce selective crystallization.
- Filter the resulting solid to separate the product from impurities that remain dissolved in the organic mother liquor.
- Redissolve the solid in a second water-immiscible solvent (Solvent B), wash with water containing the polar reagent to extract residues, and concentrate the organic phase.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain directors, the adoption of this purification method offers tangible strategic benefits that directly impact the bottom line and operational resilience. By eliminating the need for expensive acetylating agents, specialized catalysts like DMAP, and strong bases for hydrolysis, the raw material cost structure is significantly optimized. The process relies on commodity solvents such as ethyl acetate, toluene, and alcohols, which are readily available in the global market and less subject to supply volatility compared to specialized fine chemical reagents. Furthermore, the simplification of the workflow reduces the demand for complex reactor configurations and extensive downstream processing equipment, leading to substantial capital expenditure savings and lower operational overheads.
- Cost Reduction in Manufacturing: The most immediate financial advantage stems from the drastic reduction in processing steps. Traditional methods incur costs associated with multiple reaction vessels, heating and cooling cycles, and the disposal of stoichiometric amounts of acetic acid and salt byproducts generated during hydrolysis. By removing the protection-deprotection cycle entirely, this method minimizes utility consumption and waste treatment costs. The avoidance of column chromatography further eliminates the expense of silica gel and the large volumes of eluent solvents typically required, resulting in a leaner, more cost-effective manufacturing process that enhances profit margins for the final API.
- Enhanced Supply Chain Reliability: Supply continuity is bolstered by the use of widely sourced, generic solvents and reagents rather than proprietary or niche chemicals. The robustness of the crystallization process means that production is less susceptible to minor fluctuations in raw material quality, ensuring consistent output. Additionally, the shorter processing time per batch increases the overall throughput of the facility, allowing manufacturers to respond more agilely to market demand spikes. This operational flexibility is critical for maintaining reliable supply lines to downstream pharmaceutical partners who depend on just-in-time delivery of high-quality intermediates.
- Scalability and Environmental Compliance: From an environmental and scaling perspective, this method is vastly superior to legacy techniques. The absence of heavy metal catalysts and the reduction in hazardous waste generation simplify compliance with increasingly strict environmental regulations. The unit operations involved—dissolution, crystallization, filtration, and extraction—are inherently scalable from pilot plant to multi-ton commercial production without the engineering challenges associated with scaling chromatographic columns. This ease of scale-up ensures that production capacity can be expanded rapidly to meet growing global demand for diabetes medications without compromising on safety or sustainability standards.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this purification technology. These insights are derived directly from the experimental data and process descriptions found in the patent literature, providing clarity on how this method compares to existing industry standards and what stakeholders can expect regarding performance and integration.
Q: How does this purification method improve yield compared to traditional acetylation routes?
A: Traditional methods involve multi-step protection and deprotection sequences which inherently accumulate losses at each stage, often resulting in yields below 50%. This novel method eliminates chemical modification steps, relying instead on physical separation, thereby significantly preserving mass balance and improving overall recovery.
Q: What specific impurities does this process target in Dapagliflozin synthesis?
A: The process is designed to remove unreacted starting materials, side-products from the reduction step (such as silane byproducts), and colored impurities. The selective crystallization leverages solubility differences, while the subsequent aqueous wash effectively removes polar contaminants and residual reagents.
Q: Is this purification method suitable for large-scale industrial production?
A: Yes, the method is explicitly designed for scalability. It avoids column chromatography, which is difficult to scale, and utilizes standard unit operations like dissolution, crystallization, filtration, and liquid-liquid extraction, making it ideal for ton-scale manufacturing in GMP facilities.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Dapagliflozin Supplier
At NINGBO INNO PHARMCHEM, we recognize that the transition to more efficient purification technologies is essential for maintaining competitiveness in the global pharmaceutical market. Our team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that innovations like the one described in CN109705075B can be seamlessly integrated into your supply chain. We are committed to delivering high-purity intermediates that meet stringent purity specifications, supported by our rigorous QC labs and state-of-the-art analytical capabilities. Our expertise allows us to navigate the complexities of process chemistry, guaranteeing that every batch of Dapagliflozin we supply adheres to the highest quality standards required for regulatory approval.
We invite you to collaborate with us to leverage these advanced purification strategies for your specific project needs. By partnering with our technical procurement team, you can request a Customized Cost-Saving Analysis tailored to your current production volumes. We encourage you to reach out today to obtain specific COA data and comprehensive route feasibility assessments, ensuring that your next project benefits from the latest advancements in process efficiency and cost optimization.
