Advanced Synthetic Route for Temozolomide: Enhancing Safety and Scalability for Global Pharmaceutical Supply Chains
The pharmaceutical industry continuously seeks robust manufacturing processes for critical oncology agents, and Patent CN1269819C presents a transformative approach to the synthesis of temozolomide and its analogs. This intellectual property addresses long-standing safety and efficiency bottlenecks associated with the production of this potent alkylating agent. By introducing a novel sequence involving protected amino-cyanoacetamide intermediates, the invention circumvents the hazardous isolation of unstable diazo compounds and eliminates the reliance on methyl isocyanate. For R&D directors and supply chain leaders, this represents a significant leap forward in process safety and operational reliability. The methodology described herein not only enhances the purity profile of the final active pharmaceutical ingredient but also streamlines the workflow by utilizing stable, isolable solid intermediates that are far more conducive to large-scale commercial production.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
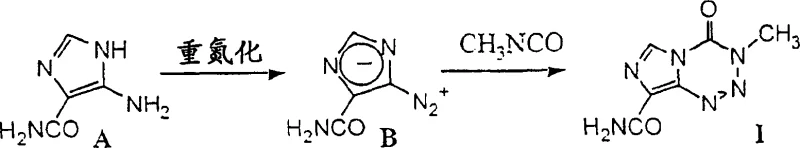
Historically, the synthesis of temozolomide has been plagued by significant safety hazards and operational inefficiencies that pose severe challenges for industrial scale-up. As illustrated in the prior art, traditional routes often rely on the direct diazotization of 5-amino-1H-imidazole-4-carboxamide to generate 5-diazo-1H-imidazole-4-carboxamide. This intermediate is notoriously unstable and potentially explosive, requiring rigorous safety protocols and specialized equipment to handle, which drastically increases capital expenditure and operational risk. Furthermore, conventional methods frequently necessitate the use of methyl isocyanate for the subsequent cycloaddition step. Methyl isocyanate is an extremely toxic reagent with a low boiling point, making its containment and transport difficult and dangerous, particularly in large-volume manufacturing settings. The reaction kinetics are also unfavorable, with literature suggesting reaction times extending up to 20 days to achieve acceptable conversion, leading to poor asset utilization and extended production cycles that strain supply chain continuity.
The Novel Approach
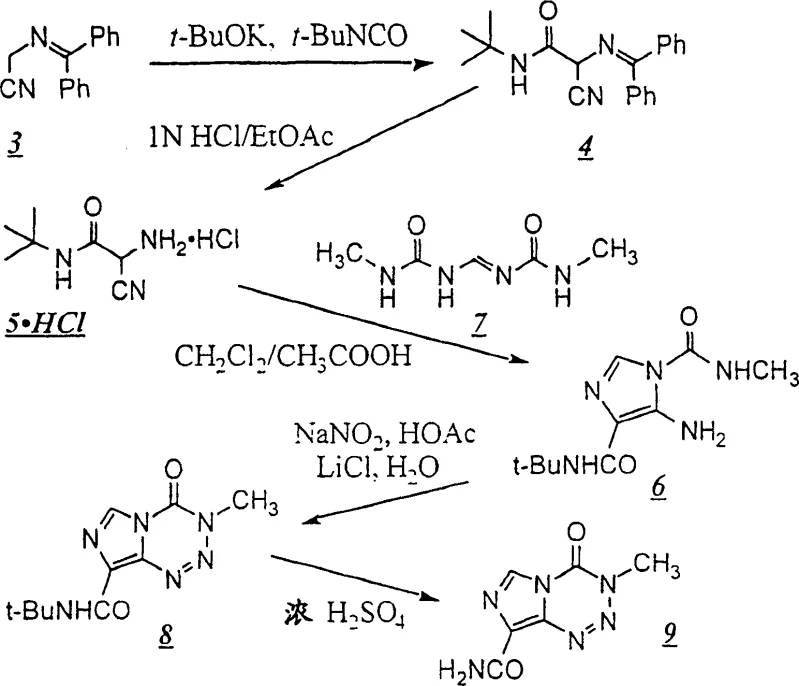
The innovative process disclosed in the patent fundamentally restructures the synthetic pathway to mitigate these risks while enhancing chemical efficiency. Instead of generating the hazardous diazo species early in the sequence, the new method employs a protected amine strategy, utilizing a bulky tert-butyl group to mask the reactive amino functionality. This protection allows for the safe construction of the imidazole core through a condensation reaction with a urea derivative under mild acidic conditions. The critical cyclization to form the tetrazine ring is achieved via an in situ diazotization in the presence of lithium chloride, which directs the reaction towards the desired regioisomer without the need to isolate the dangerous diazonium salt. This telescoped approach not only removes the most hazardous unit operations from the process flow but also significantly reduces the overall processing time. By shifting the complexity to stable, protected intermediates, the process becomes inherently safer and more robust, offering a viable solution for reliable temozolomide intermediate supplier networks aiming to secure long-term production capacity.
Mechanistic Insights into LiCl-Promoted Diazotization and Cyclization
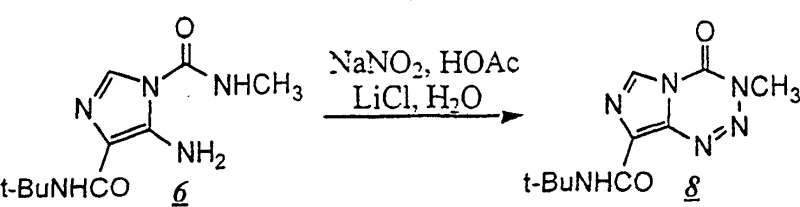
A deep understanding of the reaction mechanism reveals why this new pathway offers superior control over impurity profiles compared to legacy methods. The core innovation lies in the diazotization of the protected imidazole intermediate (Compound 6) using sodium nitrite and acetic acid. In the absence of specific additives, diazotization of such substrates can lead to a mixture of regioisomers or decomposition products. However, the presence of lithium chloride (LiCl) in the aqueous organic solvent system plays a pivotal catalytic role. It is hypothesized that the lithium cation coordinates with the nitrogen atoms of the intermediate, stabilizing the transition state and favoring the intramolecular attack required to close the tetrazine ring. This coordination effect ensures that the cyclization proceeds exclusively to form the imidazo[5,1-d]-1,2,3,5-tetrazine skeleton, minimizing the formation of structural isomers that are difficult to remove downstream. Furthermore, the steric bulk of the tert-butyl protecting group on the amide nitrogen exerts a conformational influence that further biases the reaction towards the thermodynamically stable product, effectively suppressing the formation of unwanted azahypoxanthine derivatives.
Impurity control is further enhanced by the stability of the intermediates involved in this route. Unlike the free amino-imidazole which is prone to oxidation and polymerization, the N-tert-butyl protected species is a crystalline solid that can be purified via recrystallization before entering the cyclization step. This ability to purify intermediates prior to the final ring closure is a critical quality attribute for high-purity anticancer intermediates. The final deprotection step utilizes concentrated sulfuric acid to cleave the tert-butyl group, a reaction that is highly selective and generates volatile isobutylene byproducts that are easily removed, leaving behind the clean temozolomide molecule. This mechanistic clarity provides R&D teams with the confidence that the process is scalable and capable of consistently meeting stringent pharmacopeial specifications for genotoxic impurities.
How to Synthesize Temozolomide Efficiently
The implementation of this synthetic route requires precise control over reaction parameters to maximize yield and safety. The process begins with the preparation of the protected acetamide, followed by hydrolysis to the amine salt, condensation to the imidazole, diazotization to the tetrazine, and finally acid-mediated deprotection. Each step has been optimized to utilize common industrial solvents and reagents, facilitating technology transfer. The detailed standardized synthesis steps, including specific molar ratios, temperature profiles, and workup procedures, are outlined in the guide below to ensure reproducibility across different manufacturing sites.
- React [(diphenylmethylene)amino]acetonitrile with tert-butyl isocyanate to form the protected acetamide intermediate.
- Hydrolyze the imine protecting group to obtain the amino-cyanoacetamide hydrochloride salt.
- Condense the amino-cyanoacetamide with a urea derivative to construct the imidazole ring system.
- Perform diazotization using sodium nitrite and acetic acid in the presence of LiCl to cyclize the tetrazine ring.
- Hydrolyze the tert-butyl protecting group using concentrated sulfuric acid to yield the final temozolomide product.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the adoption of this patented methodology translates into tangible strategic benefits that extend beyond simple chemical yield. By eliminating the requirement for methyl isocyanate, facilities can avoid the substantial costs associated with specialized containment systems, scrubbing units, and regulatory compliance monitoring for highly toxic gases. This removal of a high-hazard reagent significantly lowers the barrier to entry for contract manufacturing organizations and reduces the insurance premiums associated with production liability. Furthermore, the ability to isolate and store stable solid intermediates allows for decoupled production schedules, meaning that bottlenecks in one stage of the synthesis do not necessarily halt the entire plant, thereby enhancing overall equipment effectiveness and supply continuity.
- Cost Reduction in Manufacturing: The elimination of methyl isocyanate removes the need for expensive engineering controls and specialized waste treatment protocols, leading to substantial operational cost savings. Additionally, the improved regioselectivity driven by the lithium chloride additive reduces the burden on downstream purification processes, lowering solvent consumption and increasing the throughput of the final crystallization steps. The use of readily available starting materials like tert-butyl isocyanate and common acids further drives down the raw material cost base compared to specialized reagents required in older pathways.
- Enhanced Supply Chain Reliability: The stability of the key intermediates, particularly the protected imidazole and the tert-butyl-temozolomide precursor, allows for the creation of strategic inventory buffers. Unlike unstable diazo compounds that must be consumed immediately, these intermediates can be manufactured in advance and stored, providing a buffer against supply disruptions. This robustness ensures a consistent flow of high-purity anticancer intermediates to downstream API manufacturers, mitigating the risk of stockouts that can impact patient access to critical cancer therapies.
- Scalability and Environmental Compliance: The process is designed with green chemistry principles in mind, avoiding the generation of persistent organic pollutants associated with heavy metal catalysts or chlorinated solvents often found in alternative routes. The aqueous workups and the use of sulfuric acid for deprotection generate waste streams that are easier to treat and neutralize. This environmental compatibility simplifies the permitting process for new manufacturing lines and aligns with the increasingly strict sustainability mandates of global pharmaceutical buyers, ensuring long-term viability of the supply source.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this synthesis technology. These insights are derived directly from the experimental data and claims within the patent documentation, providing clarity on the practical aspects of adopting this route for commercial production.
Q: Why is the new synthesis route for temozolomide considered safer than conventional methods?
A: The novel process eliminates the need to isolate the unstable and potentially explosive 5-diazo-1H-imidazole-4-carboxamide intermediate. Additionally, it avoids the use of methyl isocyanate, a highly toxic and difficult-to-handle reagent required in older synthetic pathways.
Q: How does the use of a tert-butyl protecting group improve the reaction outcome?
A: The bulky tert-butyl group provides steric hindrance that promotes the correct orientation for cyclization, ensuring the formation of the desired imidazo[5,1-d]-1,2,3,5-tetrazine ring rather than unwanted azahypoxanthine derivatives.
Q: What role does Lithium Chloride (LiCl) play in the diazotization step?
A: LiCl acts as a crucial additive in the reaction medium that favors the correct cyclization direction during the diazotization of the imidazole intermediate, significantly improving the regioselectivity and yield of the tetrazine ring formation.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Temozolomide Intermediate Supplier
The technological advancements detailed in Patent CN1269819C underscore the complexity and sophistication required to manufacture high-value oncology intermediates efficiently. At NINGBO INNO PHARMCHEM, we possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that our clients receive a supply of material that meets the highest standards of quality and consistency. Our facility is equipped with state-of-the-art rigorous QC labs and analytical capabilities to verify stringent purity specifications, guaranteeing that every batch of temozolomide intermediate adheres to global regulatory requirements. We understand that in the pharmaceutical sector, reliability is as critical as chemistry, and our integrated supply chain is designed to deliver on both fronts.
We invite potential partners to engage with our technical procurement team to discuss how this optimized synthesis route can be tailored to your specific volume requirements. By leveraging our expertise in process development and scale-up, we can provide a Customized Cost-Saving Analysis that demonstrates the economic benefits of switching to this safer, more efficient methodology. We encourage you to request specific COA data and route feasibility assessments to validate the superiority of our manufacturing capabilities. Let us collaborate to secure a resilient and cost-effective supply chain for your critical pharmaceutical projects.
