Advanced Synthesis of Azaspiro Intermediates for Commercial Quinolone Production
The pharmaceutical industry constantly seeks robust synthetic routes for complex heterocyclic intermediates, particularly those serving as precursors for potent antibacterial agents. Patent CN1191529A introduces a groundbreaking methodology for producing nitrogen-containing heterocyclic compounds with a spiro ring structure, specifically optically active azaspiro[2,4]heptane derivatives. These compounds are critical building blocks in the synthesis of quinolone derivatives, which are expected to function as excellent antibacterial agents in clinical applications. The invention addresses significant limitations in prior art by offering a process that is inexpensive, involves fewer steps, and provides distinct advantages for industrial production scalability. By leveraging diastereoselective reactions and efficient cyclization strategies, this technology enables the manufacture of high-purity intermediates without the need for cumbersome chromatographic separations typically associated with chiral synthesis. This report analyzes the technical merits and commercial implications of this patented process for global supply chain stakeholders.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
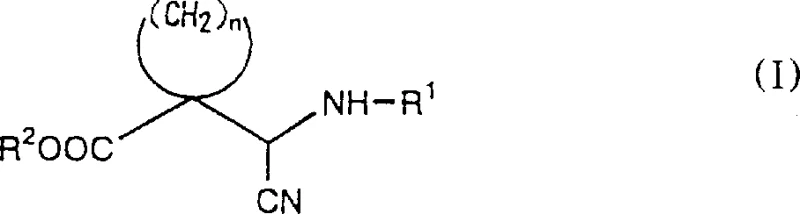
Historically, the production of compounds represented by formula (VI) relied heavily on ethyl acetoacetate as a raw material, necessitating a multi-step production process that was both time-consuming and resource-intensive. Previous methods, such as those disclosed in JP-A-2-231475, required the preparation of diastereomeric compounds possessing optically active protecting groups to achieve stereochemical control. These intermediates subsequently needed separation via preparative high-performance liquid chromatography (HPLC), followed by the removal of the protecting group to yield the final optically active compound. This reliance on preparative HPLC represents a significant bottleneck in commercial manufacturing, as it drastically increases operational costs and limits the throughput capacity of production facilities. Furthermore, the complexity of operations involved in handling and removing specific protecting groups introduces additional risks of yield loss and impurity generation, making these conventional methods less desirable for large-scale industrial applications where efficiency and cost-effectiveness are paramount.
The Novel Approach
The novel approach disclosed in the patent fundamentally shifts the synthetic strategy by utilizing a diastereoselective cyanohydrin formation reaction that bypasses the need for complex protecting group manipulations. Instead of relying on ethyl acetoacetate, the process starts with known compounds of formula (IV), which are reacted with hydrogen cyanide in the presence of an optically active single amine or its salt. This reaction directly yields compounds of formula (I) with high stereoselectivity, where one diastereomer is obtained in a predominant ratio compared to the other. The resulting intermediates can be easily transformed into the target formula (II) compounds through reduction and cyclization steps that are amenable to standard industrial equipment. By eliminating the requirement for preparative HPLC and simplifying the protection-deprotection sequence, this method significantly streamlines the production workflow. The ability to obtain optically active compounds directly through diastereoselective synthesis represents a major technological leap, offering a more direct and economically viable pathway for manufacturing complex spirocyclic intermediates.
Mechanistic Insights into Diastereoselective Cyanohydrin Formation
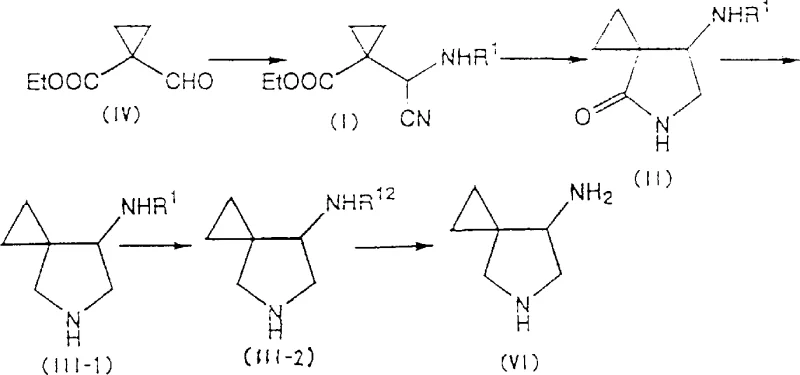
The core mechanistic advantage of this process lies in the diastereoselective addition of hydrogen cyanide to the aldehyde group of formula (IV) compounds in the presence of chiral amines. When an optically active single amine, such as (S)-1-phenylethylamine, is employed, the reaction proceeds to form a mixture of two diastereomers, but with a distinct preference for one configuration over the other. For instance, the use of specific chiral amines can result in formation ratios as favorable as 73:27 or even higher, depending on the steric and electronic properties of the amine substituent. This inherent selectivity reduces the burden on downstream purification processes, as the major diastereomer can often be isolated through simpler crystallization or extraction techniques rather than complex chromatography. Furthermore, the patent describes a unique epimerization phenomenon where the undesired diastereomer can be treated in a protic solvent, such as ethanol, to undergo configuration inversion at the cyano-bonded carbon atom. This allows for the conversion of the minor isomer into the major isomer, effectively recycling material that would otherwise be discarded and maximizing the overall yield of the desired stereoisomer.
Following the formation of the cyanohydrin intermediate, the process involves a critical reduction and cyclization sequence to construct the azaspiro ring system. The cyano group of the formula (I) compound is reduced to an aminomethyl group, typically using catalytic hydrogenation with catalysts like Raney nickel under controlled hydrogen pressure. This reduction step is carefully managed to ensure that the ester functionality remains intact while the nitrile is fully converted to the primary amine. Subsequently, the resulting amino compound undergoes thermal cyclization, often facilitated by heating the reaction mixture to temperatures ranging from 50°C to 180°C. This intramolecular condensation forms the lactam ring characteristic of the formula (II) structure, locking the stereochemistry established in the earlier steps. The use of Raney nickel is particularly advantageous due to its high activity and cost-effectiveness compared to precious metal catalysts, aligning well with the goal of developing an inexpensive industrial process. The robustness of these reaction conditions ensures consistent product quality and minimizes the formation of by-products that could complicate purification.
How to Synthesize Azaspiro[2,4]heptane Efficiently
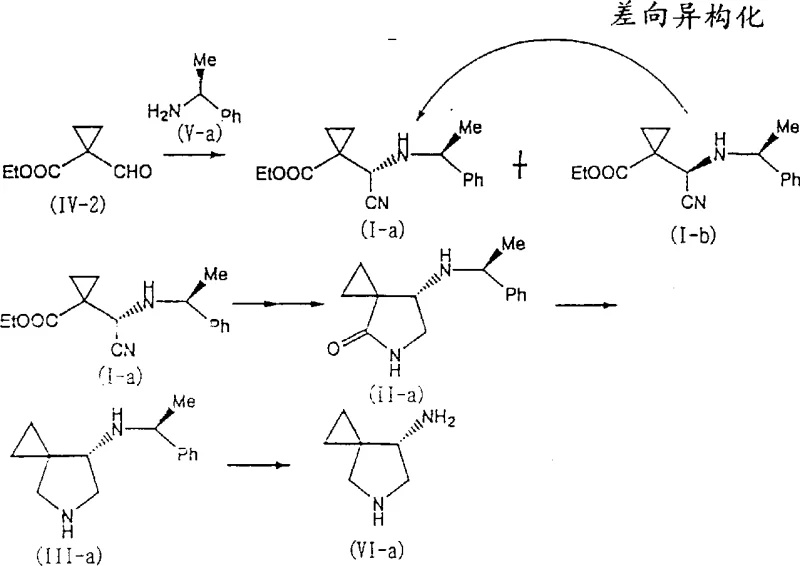
The synthesis of these high-value intermediates begins with the preparation of the cyclopropane aldehyde starting material, which can be derived from 1-cyano-cyclopropanecarboxylate using established methods. The key operational step involves the addition of hydrogen cyanide, generated in situ from alkali metal cyanides and acid, to the aldehyde in the presence of the chiral amine promoter. Reaction temperatures are typically maintained between -20°C and 100°C, with a preference for 50°C to balance reaction rate and selectivity. Following the cyanohydrin formation, the mixture is worked up using standard extraction techniques with solvents like ethyl acetate, avoiding the need for specialized chromatographic columns. The subsequent reduction and cyclization steps are performed in autoclaves capable of handling hydrogen pressure, ensuring safety and reproducibility at scale. Detailed standardized synthesis steps see the guide below.
- React cyclopropane aldehyde derivatives with hydrogen cyanide in the presence of an optically active amine to form diastereomeric cyanohydrins.
- Perform epimerization in a protic solvent to enrich the desired stereoisomer ratio before separation.
- Execute catalytic hydrogenation followed by thermal cyclization to generate the final azaspiro lactam structure.
Commercial Advantages for Procurement and Supply Chain Teams
From a procurement and supply chain perspective, this patented methodology offers substantial benefits by addressing key pain points associated with the manufacturing of complex chiral intermediates. The elimination of preparative HPLC purification significantly reduces the capital expenditure required for production facilities, as expensive chromatography columns and associated solvent recovery systems are no longer necessary. This simplification of the process flow directly translates to lower operational costs and a reduced environmental footprint due to decreased solvent consumption. Furthermore, the use of readily available starting materials and common reagents like Raney nickel enhances supply chain reliability, minimizing the risk of disruptions caused by the scarcity of specialized catalysts or reagents. The robustness of the reaction conditions also implies a higher success rate in technology transfer from laboratory to pilot and commercial scales, ensuring consistent supply continuity for downstream drug manufacturers.
- Cost Reduction in Manufacturing: The process achieves cost optimization primarily by removing the need for expensive chromatographic separation steps and reducing the number of synthetic operations required to reach the final intermediate. By utilizing diastereoselective synthesis to control stereochemistry, the method avoids the yield losses typically associated with resolving racemic mixtures or removing complex protecting groups. The use of cost-effective catalysts like Raney nickel instead of precious metals further contributes to lowering the raw material costs per kilogram of product. Additionally, the ability to epimerize unwanted isomers back into the desired product stream maximizes atom economy and reduces waste disposal costs. These factors collectively result in a significantly more economical production process compared to conventional routes relying on ethyl acetoacetate and HPLC purification.
- Enhanced Supply Chain Reliability: Supply chain stability is greatly improved through the use of commodity chemicals and standard reaction conditions that are easily sourced from multiple global suppliers. The avoidance of specialized chiral columns or rare reagents means that production is less vulnerable to single-source supply risks or geopolitical disruptions. The simplified workflow also reduces the lead time required for batch production, allowing manufacturers to respond more quickly to fluctuations in market demand. Moreover, the high yield and selectivity of the process ensure that production targets can be met consistently without the need for extensive re-processing or batch rejection. This reliability is crucial for pharmaceutical companies that require a steady supply of high-quality intermediates to maintain their own drug manufacturing schedules.
- Scalability and Environmental Compliance: The process is inherently designed for scalability, utilizing reaction conditions and equipment that are standard in the fine chemical industry, such as stirred tank reactors and hydrogenation autoclaves. The reduction in solvent usage, particularly the elimination of large volumes of mobile phase required for HPLC, aligns with increasingly stringent environmental regulations regarding volatile organic compound emissions. The ability to recycle unwanted isomers through epimerization further supports sustainability goals by minimizing chemical waste generation. Scaling this process from kilogram to tonnage levels does not require fundamental changes to the chemistry, reducing the technical risk associated with commercial scale-up. This makes it an attractive option for manufacturers looking to expand capacity while maintaining compliance with green chemistry principles and regulatory standards.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this synthesis route, based on the specific details provided in the patent documentation. These answers are derived from the experimental data and process descriptions to provide clarity on feasibility and performance. Understanding these aspects is essential for technical teams evaluating the adoption of this technology for their specific production needs.
Q: How does this patent improve upon conventional ethyl acetoacetate routes?
A: The patented method eliminates the need for complex preparative HPLC separation of protecting groups, utilizing diastereoselective cyanohydrin formation instead for superior stereocontrol.
Q: What catalysts are preferred for the reduction and cyclization steps?
A: Raney nickel is particularly preferred for the catalytic hydrogenation of the cyano group, offering high efficiency in converting intermediates to the cyclic lactam structure.
Q: Is this process suitable for large-scale commercial manufacturing?
A: Yes, the process uses inexpensive reagents and avoids chromatographic purification, making it highly advantageous for industrial production and cost reduction.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Azaspiro[2,4]heptane Supplier
The technical potential of this diastereoselective synthesis route represents a significant opportunity for optimizing the production of quinolone intermediates, and NINGBO INNO PHARMCHEM is well-positioned to facilitate this transition. As a specialized CDMO partner, we possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that complex chemistries like this are translated into robust manufacturing processes. Our facilities are equipped with the necessary hydrogenation and high-pressure reaction capabilities to execute the reduction and cyclization steps safely and efficiently. We maintain stringent purity specifications and operate rigorous QC labs to guarantee that every batch of azaspiro intermediate meets the exacting standards required for pharmaceutical applications. Our team is dedicated to delivering high-purity azaspiro[2,4]heptane derivatives that support the development of next-generation antibacterial agents.
We invite you to engage with our technical procurement team to discuss how this patented technology can be integrated into your supply chain for maximum efficiency. By requesting a Customized Cost-Saving Analysis, you can gain specific insights into the potential economic benefits of switching to this streamlined synthesis route. We encourage you to contact us to obtain specific COA data and route feasibility assessments tailored to your project requirements. Our experts are ready to collaborate on optimizing the process parameters to ensure the highest yield and quality for your commercial production needs. Partnering with us ensures access to advanced synthetic capabilities and a commitment to long-term supply reliability.
Engineering Bottleneck?
Can't scale up this synthesis? Upload your target structure or CAS, and our CDMO team will evaluate the industrial feasibility within 24 hours. Request Evaluation →
