Industrial Scale-Up of Roflumilast Intermediates: A Novel High-Yield Synthetic Route
Industrial Scale-Up of Roflumilast Intermediates: A Novel High-Yield Synthetic Route
The pharmaceutical landscape for respiratory diseases has been significantly transformed by the introduction of Roflumilast, a potent oral phosphodiesterase 4 (PDE4) inhibitor used for treating severe chronic obstructive pulmonary disease (COPD). As global demand for this critical active pharmaceutical ingredient (API) continues to rise, the efficiency of its supply chain becomes paramount for generic manufacturers and innovators alike. A pivotal advancement in this domain is detailed in patent CN102617457A, which discloses a streamlined, industrially viable method for preparing Roflumilast and its key intermediates. This technical insight report analyzes the breakthrough methodology, focusing on how it resolves long-standing bottlenecks in selectivity and purification that have historically plagued the commercial production of this vital respiratory medication.
The core innovation lies in a strategic reordering of synthetic steps that bypasses the need for complex separation technologies. Traditional routes often struggled with the selective protection of hydroxyl groups on the benzene ring, necessitating costly and time-consuming column chromatography. The novel approach presented in this patent utilizes 3-bromo-4-hydroxybenzaldehyde as a starting material, enabling a direct and highly selective difluoromethoxylation. This shift not only enhances the chemical purity of the intermediates but also drastically simplifies the downstream processing, offering a compelling value proposition for procurement teams seeking reliable roflumilast intermediate suppliers who can guarantee consistent quality without the premium price tag associated with legacy manufacturing processes.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Prior to the innovations described in CN102617457A, the industrial synthesis of Roflumilast faced significant hurdles related to reaction selectivity and purification complexity. As illustrated in the comparative reaction schemes, earlier methods such as those disclosed in WO9501338A1 and WO2005026095A1 typically commenced with 3,4-dihydroxybenzaldehyde. The fundamental flaw in this approach was the difficulty in achieving selective mono-etherification. When reacting 3,4-dihydroxybenzaldehyde with alkylating agents, chemists frequently encountered a mixture of mono-etherified, di-etherified, and unreacted starting materials. This lack of regioselectivity forced manufacturers to rely heavily on column chromatography to isolate the desired 3-cyclopropylmethoxy-4-difluoromethoxy intermediate.
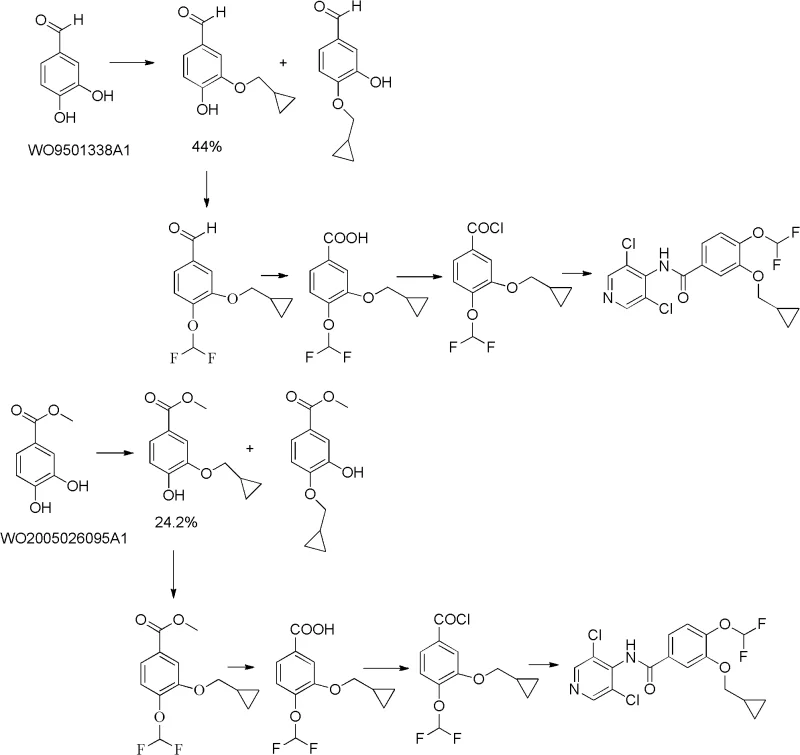
Furthermore, the reliance on column chromatography is a major impediment to cost reduction in API manufacturing. On a laboratory scale, chromatography is manageable, but on a multi-ton commercial scale, it represents a massive operational burden involving large volumes of silica gel, excessive solvent consumption, and significant waste generation. Additionally, some prior art routes utilized expensive reagents like cyclopropylmethyl bromide for the etherification step, which further inflated the raw material costs. The combination of low yields due to side reactions and the high operational expenditure of purification rendered these conventional methods suboptimal for modern, lean pharmaceutical manufacturing environments that prioritize both economic efficiency and environmental sustainability.
The Novel Approach
The methodology introduced in patent CN102617457A offers a robust solution by fundamentally altering the synthetic trajectory. Instead of starting with the symmetric 3,4-dihydroxybenzaldehyde, the process initiates with 3-bromo-4-hydroxybenzaldehyde (Compound I). This strategic choice leverages the inherent reactivity differences between the phenolic hydroxyl group and the bromine substituent. In the first critical step, the phenolic hydroxyl group is selectively etherified with chlorodifluoroacetic acid sodium or chlorodifluoromethane under basic conditions to yield 4-difluoromethoxy-3-hydroxybenzaldehyde (Compound II). This step proceeds with exceptional selectivity, effectively eliminating the formation of unwanted di-substituted byproducts that plagued previous methods.
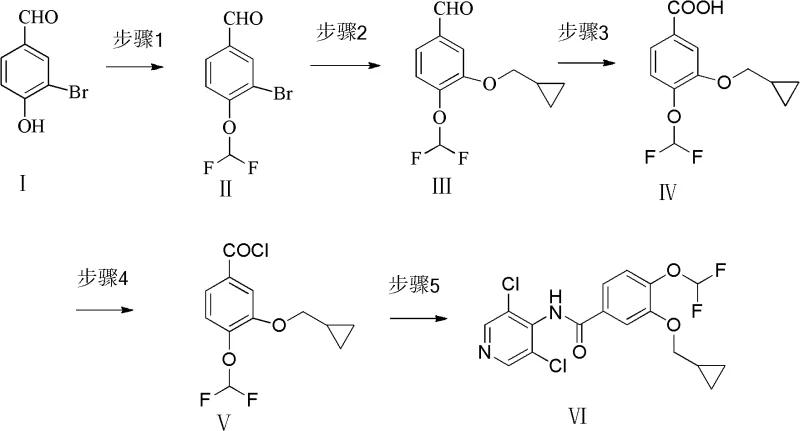
Following the initial etherification, the synthesis employs a modified Ullmann condensation reaction to introduce the cyclopropylmethoxy group. By reacting Compound II with cyclopropylcarbinol in the presence of copper catalysts and specific ligands, the process efficiently generates 3-cyclopropylmethoxy-4-difluoromethoxy-benzaldehyde (Compound III). The subsequent oxidation of the aldehyde to the carboxylic acid using sodium hypochlorite, followed by chlorination and final acylation with 3,5-dichloro-4-aminopyridine, completes the synthesis of Roflumilast (Compound VI). This route is characterized by its operational simplicity; it completely obviates the need for column chromatography, relying instead on straightforward extraction and crystallization techniques that are easily scalable and economically superior for high-purity roflumilast production.
Mechanistic Insights into Copper-Catalyzed Ullmann Condensation
The success of this novel synthetic route hinges largely on the efficiency of the Ullmann condensation step, where the cyclopropylmethoxy moiety is installed onto the aromatic ring. Mechanistically, this transformation involves the coupling of an aryl halide (in this specific variation, effectively an activated phenol derivative undergoing nucleophilic substitution facilitated by copper) or more accurately in this patent's context, a copper-catalyzed O-arylation or etherification of the phenol with the alcohol. The patent details the use of copper compounds such as cuprous iodide or cuprous oxide in conjunction with bidentate ligands like oxine (8-hydroxyquinoline), 1,10-phenanthroline, or N,N'-dimethylethylenediamine. These ligands play a crucial role in stabilizing the copper center and facilitating the oxidative addition and reductive elimination cycles necessary for the C-O bond formation.
The reaction conditions are meticulously optimized to balance reactivity and selectivity. The use of polar aprotic solvents such as DMF (dimethylformamide) or DMA (dimethylacetamide) is essential for solubilizing the inorganic bases (like potassium carbonate or cesium carbonate) and the organic substrates. Temperature control is also critical; while the reaction can proceed at temperatures ranging from 20°C to 150°C, higher temperatures generally favor faster kinetics but must be managed to prevent decomposition of the sensitive cyclopropyl group or the difluoromethoxy ether. The mechanistic pathway likely involves the formation of a copper-alkoxide species which then attacks the electron-deficient aromatic ring (activated by the adjacent aldehyde and difluoromethoxy groups), displacing the leaving group or facilitating the coupling through a radical or concerted mechanism depending on the specific copper-ligand complex formed.
Impurity control is another vital aspect of this mechanism. In traditional etherifications using strong alkylating agents like alkyl halides, over-alkylation is a common impurity. However, the Ullmann-type coupling described here, combined with the specific sequence of introducing the difluoromethoxy group first, minimizes side reactions. The steric and electronic environment created by the 4-difluoromethoxy group directs the subsequent substitution to the 3-position with high fidelity. Furthermore, the oxidation step using sodium hypochlorite is a green chemistry alternative to heavy metal oxidants like chromium or manganese salts. This choice not only reduces the toxicity of the waste stream but also simplifies the workup, as the byproducts are primarily inorganic salts that can be easily removed by aqueous washing, thereby ensuring the high purity specifications required for pharmaceutical intermediates.
How to Synthesize 3-Cyclopropylmethoxy-4-difluoromethoxy-benzoyl chloride Efficiently
The synthesis of the key acyl chloride intermediate (Compound V) is a linchpin in the production of Roflumilast, serving as the electrophile in the final amide bond formation. The patent outlines a pragmatic approach where the carboxylic acid (Compound IV), obtained from the hypochlorite oxidation of the aldehyde, is treated with thionyl chloride in the presence of a catalytic amount of DMF. This activation step converts the relatively stable carboxylic acid into the highly reactive acid chloride, which is then immediately reacted with 3,5-dichloro-4-aminopyridine. The detailed standardized synthesis steps for this transformation, including precise molar ratios, temperature profiles, and quenching procedures, are provided in the technical guide below to assist process chemists in replicating this high-yield protocol.
- Etherify 3-bromo-4-hydroxybenzaldehyde with chlorodifluoroacetic acid sodium to obtain 4-difluoromethoxy-3-hydroxybenzaldehyde.
- Perform Ullmann condensation with cyclopropylcarbinol using copper catalysts to form the cyclopropylmethoxy derivative.
- Oxidize the aldehyde to the corresponding acid using sodium hypochlorite, followed by chlorination and acylation to finalize Roflumilast.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain directors, the technical improvements in CN102617457A translate directly into tangible business benefits. The most significant advantage is the drastic simplification of the purification process. By eliminating the need for column chromatography, the manufacturing process becomes inherently more robust and scalable. Chromatography is often the bottleneck in batch processing, limiting throughput and requiring specialized equipment and large amounts of consumables. Removing this step allows for larger batch sizes and faster cycle times, which directly enhances supply chain reliability and reduces the risk of production delays. This operational efficiency ensures a more consistent supply of high-purity intermediates, mitigating the volatility often seen in the API market.
- Cost Reduction in Manufacturing: The economic implications of this new route are profound. The elimination of column chromatography removes a major cost center associated with silica gel, solvent recovery, and labor. Furthermore, the use of 3-bromo-4-hydroxybenzaldehyde as a starting material avoids the expensive and hazardous cyclopropylmethyl bromide reagent used in older methods, replacing it with the more economical cyclopropylcarbinol. The high yields reported in the patent embodiments, such as the 91.6% yield in the initial etherification step and 87.5% in the oxidation step, indicate a highly atom-economical process. These factors combine to significantly lower the cost of goods sold (COGS), allowing for more competitive pricing strategies in the generic pharmaceutical market.
- Enhanced Supply Chain Reliability: Supply continuity is critical for life-saving medications like Roflumilast. The new synthesis route relies on readily available raw materials and standard chemical transformations that are less prone to failure than complex, low-selectivity reactions. The robustness of the Ullmann condensation under the specified conditions means that batch-to-batch variability is minimized. Additionally, the simplified workup procedures reduce the dependency on specialized purification infrastructure, making it easier to qualify multiple manufacturing sites. This flexibility strengthens the supply chain against disruptions, ensuring that pharmaceutical partners can maintain uninterrupted production schedules even in fluctuating market conditions.
- Scalability and Environmental Compliance: From an environmental, health, and safety (EHS) perspective, this process represents a significant improvement. The avoidance of heavy metal oxidants and the reduction in solvent usage (due to no chromatography) align with green chemistry principles. The waste stream is simpler to treat, primarily consisting of aqueous salts and recoverable organic solvents. This ease of waste management facilitates regulatory compliance and reduces the environmental footprint of the manufacturing facility. Scalability is further enhanced because the unit operations involved—stirred tank reactions, extractions, and crystallizations—are standard in the fine chemical industry, allowing for seamless technology transfer from pilot plant to commercial scale production of complex pharmaceutical intermediates.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the synthesis of Roflumilast intermediates as described in the patent literature. These insights are derived directly from the experimental data and process descriptions provided in CN102617457A, offering clarity on the feasibility and advantages of this specific manufacturing route for industry stakeholders evaluating potential technology partnerships.
Q: What is the primary advantage of the new Roflumilast synthesis route described in CN102617457A?
A: The primary advantage is the elimination of column chromatography purification. By starting with 3-bromo-4-hydroxybenzaldehyde instead of 3,4-dihydroxybenzaldehyde, the process avoids difficult selective etherification, leading to higher purity and significantly lower production costs.
Q: How does the Ullmann condensation step impact the overall yield?
A: The optimized Ullmann condensation using ligands like oxine or 1,10-phenanthroline allows for efficient coupling of the cyclopropyl group. While early embodiments showed moderate yields, optimization of temperature and ligands can significantly improve the conversion to the key aldehyde intermediate.
Q: Is this process suitable for large-scale commercial manufacturing?
A: Yes, the process is designed for industrial suitability. It utilizes common solvents like DMF and DMA, avoids expensive reagents like cyclopropylmethyl bromide used in older methods, and simplifies post-treatment, making it highly scalable for metric-ton production.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Roflumilast Intermediate Supplier
The technological advancements detailed in patent CN102617457A underscore the potential for highly efficient, cost-effective production of Roflumilast. At NINGBO INNO PHARMCHEM, we specialize in translating such innovative laboratory protocols into robust commercial realities. Our team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the theoretical benefits of high yield and purity are fully realized in the final product. We operate stringent purity specifications and utilize rigorous QC labs to guarantee that every batch of Roflumilast intermediate meets the exacting standards required by global regulatory bodies, providing our partners with absolute confidence in the quality of their supply chain.
We invite pharmaceutical companies and contract manufacturers to collaborate with us to leverage this superior synthetic route. By partnering with NINGBO INNO PHARMCHEM, you gain access to a Customized Cost-Saving Analysis tailored to your specific volume requirements. We encourage you to contact our technical procurement team today to request specific COA data and route feasibility assessments. Let us help you optimize your Roflumilast supply chain with a solution that balances technical excellence with commercial viability.
