Scalable Crystallization Process for High-Purity Abacavir Intermediate Manufacturing
Scalable Crystallization Process for High-Purity Abacavir Intermediate Manufacturing
The global demand for antiretroviral therapies continues to drive the need for efficient, scalable synthesis routes for key pharmaceutical intermediates. A pivotal development in this sector is detailed in Chinese Patent CN107641122B, which discloses a novel purification method for a critical Abacavir intermediate, specifically the compound designated as Formula IV. This intermediate serves as the cornerstone for the synthesis of Abacavir, a potent and selective inhibitor of HIV-1 and HIV-2 reverse transcriptase. Traditionally, obtaining this intermediate in the high purity required for Good Manufacturing Practice (GMP) API production has relied on labor-intensive and costly column chromatography. However, the technology outlined in this patent introduces a robust recrystallization protocol that leverages specific solubility differentials in water-organic solvent mixtures. By shifting from chromatographic separation to thermodynamic crystallization, manufacturers can achieve purity levels exceeding 99.0% while drastically simplifying the operational workflow. This report analyzes the technical merits of this innovation, providing strategic insights for R&D directors and supply chain leaders seeking to optimize their antiviral drug supply chains.
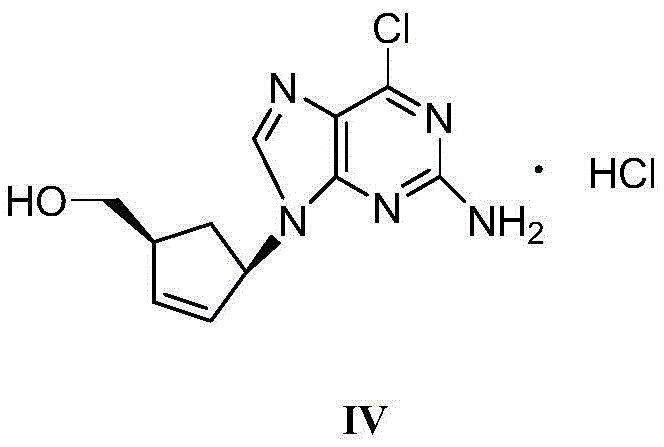
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
In the established landscape of fine chemical manufacturing, the purification of complex heterocyclic intermediates like the Abacavir precursor has historically been bottlenecked by reliance on column chromatography. While effective for laboratory-scale isolation, column chromatography presents severe disadvantages when transitioning to commercial production. The process requires massive quantities of stationary phases, such as silica gel, which not only inflates raw material costs but also generates substantial solid waste, complicating environmental compliance and disposal logistics. Furthermore, the dynamic nature of column separation makes it difficult to maintain consistent batch-to-batch reproducibility at large scales, often leading to variable impurity profiles that require re-processing. The extensive use of organic solvents to elute compounds further exacerbates the environmental footprint, increasing the E-factor of the process and necessitating complex solvent recovery systems. For procurement managers, these factors translate into unpredictable lead times and volatile pricing structures, as the throughput of chromatographic purification is inherently limited by column capacity and flow rates.
The Novel Approach
The methodology presented in Patent CN107641122B offers a paradigm shift by replacing chromatographic separation with a controlled recrystallization process. This approach capitalizes on the distinct solubility characteristics of the target intermediate versus its impurities in a binary solvent system comprising purified water and a water-soluble organic solvent. By heating the crude mixture to a specific temperature range of 40°C to 80°C, the intermediate is fully dissolved, allowing impurities to remain in solution or be removed via filtration. Subsequent cooling to 0°C to 20°C induces the selective precipitation of the target compound as high-quality rod-shaped crystals. This phase transition effectively excludes impurities from the crystal lattice, yielding a product with HPLC purity consistently above 99.0% and significantly reduced color bodies, evidenced by absorbance values ≤0.06 at 420nm. This transition from kinetic separation (chromatography) to thermodynamic equilibrium (crystallization) represents a critical optimization for industrial scalability, enabling the processing of larger batch sizes with minimal equipment modification.
Mechanistic Insights into Solubility-Driven Crystallization
To fully appreciate the efficacy of this purification strategy, one must understand the underlying physicochemical mechanisms governing the dissolution and crystallization phases. The Abacavir intermediate (Formula IV) possesses a unique amphiphilic character due to the presence of both hydrophobic aromatic/heterocyclic rings and a hydrophilic hydroxymethyl group. In the patented process, the selection of a water-soluble organic solvent (such as ethanol, methanol, or acetone) mixed with purified water creates a solvent environment where the solubility of the intermediate is highly temperature-dependent. At elevated temperatures (50°C to 60°C), the kinetic energy of the solvent molecules overcomes the lattice energy of the solid intermediate, facilitating complete solvation. Crucially, the patent specifies a mass ratio of crude intermediate to organic solvent between 1:4 and 1:20, ensuring that the solution is saturated enough to promote yield but dilute enough to prevent oiling out or amorphous precipitation. This precise control prevents the entrapment of mother liquor within the crystal matrix, which is a common source of residual impurities.
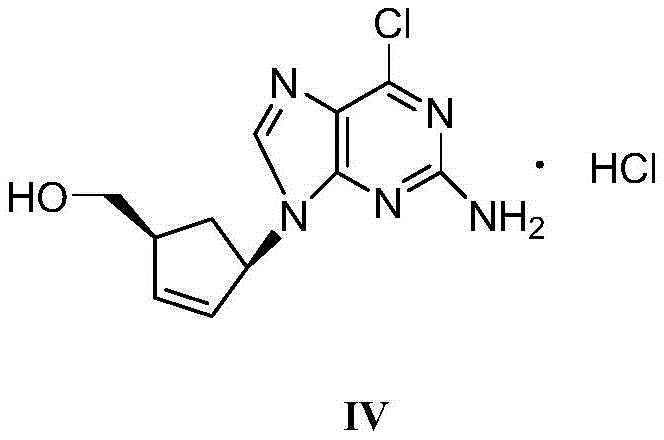
Furthermore, the synthesis pathway leading to the crude material involves the reaction of (1S-cis)-4-amino-2-cyclopentenyl-1-methanol hydrochloride (Compound I) with (2-amino-4,6-dichloro-pyrimidin-5-yl)-carboxamide (Compound II) under alkaline conditions, followed by cyclization with triethyl orthoformate. This sequence can generate colored polymeric byproducts and unreacted starting materials. The recrystallization process acts as a highly selective filter; as the solution cools, the thermodynamically stable crystal lattice of Formula IV forms preferentially. The rod-shaped morphology observed in electron microscopy images of the purified product indicates a high degree of crystallinity and order, which correlates with lower impurity inclusion. The addition of activated carbon prior to cooling further enhances purity by adsorbing high-molecular-weight colored impurities that might otherwise co-crystallize or stain the final product. This multi-mechanistic approach—combining solubility differentiation, thermal control, and adsorption—ensures that the final intermediate meets the stringent specifications required for subsequent coupling reactions in Abacavir synthesis.
How to Synthesize Abacavir Intermediate Efficiently
Implementing this purification protocol requires strict adherence to the thermal and stoichiometric parameters defined in the patent to ensure reproducibility and optimal yield. The process begins with the preparation of a homogeneous mixture of the crude intermediate, purified water, and the chosen organic solvent. Operators must monitor the heating phase carefully to ensure complete dissolution without exceeding temperatures that might degrade the sensitive heterocyclic core. Once a clear solution is obtained, the optional but recommended step of activated carbon treatment should be performed at temperature to maximize adsorption efficiency. Following hot filtration to remove the carbon and any insoluble particulates, the cooling rate becomes a critical process parameter; controlled cooling promotes the growth of larger, purer crystals rather than rapid precipitation of fines. The detailed standard operating procedures, including specific solvent ratios and temperature ramps for different solvent systems, are outlined in the technical guide below.
- Dissolve the crude abacavir intermediate in a mixture of purified water and a water-soluble organic solvent (such as ethanol or methanol) by heating to 40-80°C until completely clear.
- Optionally add activated carbon for decolorization, filter the hot solution to remove insoluble particulates, and cool the filtrate to 0-20°C to induce crystallization.
- Filter the resulting rod-shaped crystals, wash if necessary, and dry under vacuum to obtain the high-purity abacavir intermediate with absorbance values ≤0.06.
Commercial Advantages for Procurement and Supply Chain Teams
For stakeholders responsible for the economic viability and continuity of API production, the transition to this crystallization-based purification method offers profound strategic benefits. The elimination of column chromatography removes a major bottleneck in the manufacturing workflow, directly addressing concerns regarding production throughput and facility utilization. By simplifying the unit operations to basic dissolution, filtration, and drying, the process becomes amenable to continuous manufacturing techniques or large-batch reactor processing, significantly enhancing the reliability of supply. This robustness ensures that procurement managers can secure longer-term contracts with reduced risk of disruption, as the process is less susceptible to the variability inherent in manual chromatographic separations. Moreover, the reduction in complexity lowers the barrier for technology transfer between manufacturing sites, facilitating a more resilient and geographically diversified supply chain for this critical HIV medication precursor.
- Cost Reduction in Manufacturing: The economic impact of replacing chromatography with recrystallization is substantial, primarily driven by the removal of expensive stationary phases and the reduction in solvent consumption. Chromatographic processes typically require large volumes of eluent and frequent replacement of silica media, both of which represent significant operational expenditures. By contrast, the recrystallization method utilizes recoverable solvents like ethanol or methanol, which can be distilled and reused, drastically lowering raw material costs. Additionally, the simplified workflow reduces labor hours and energy consumption associated with pumping and fraction collection. These efficiencies translate into a lower cost of goods sold (COGS), allowing for more competitive pricing in the generic pharmaceutical market while maintaining healthy margins for the manufacturer.
- Enhanced Supply Chain Reliability: Supply chain resilience is heavily dependent on the predictability of production cycles. The conventional chromatographic approach often suffers from variable cycle times due to column packing inconsistencies or clogging issues. The new crystallization method offers a fixed, predictable cycle time determined solely by heating and cooling rates, which are easily automated and controlled. This predictability allows for more accurate inventory planning and shorter lead times for customers. Furthermore, the use of common, commodity-grade solvents (ethanol, methanol, acetone) mitigates the risk of supply shortages associated with specialized chromatographic grades of solvents or silica gel, ensuring that production can continue uninterrupted even during broader market fluctuations.
- Scalability and Environmental Compliance: From an environmental and regulatory perspective, this process aligns perfectly with Green Chemistry principles. The significant reduction in solid waste (silica gel) and liquid waste (solvent volume) lowers the environmental burden and simplifies waste treatment protocols. Scaling a crystallization process from pilot plant to commercial tonnage is a well-understood engineering challenge, whereas scaling chromatography often requires disproportionate increases in equipment footprint and complexity. This ease of scale-up means that manufacturers can rapidly respond to surges in demand for Abacavir without requiring massive capital investment in new purification infrastructure. The resulting high-purity product also reduces the burden on downstream QC labs, as fewer impurities need to be monitored and cleared before the material can be released for the next synthetic step.
Frequently Asked Questions (FAQ)
The following questions address common technical and operational inquiries regarding the implementation of this purification technology. These insights are derived directly from the experimental data and embodiments described in Patent CN107641122B, providing clarity on solvent selection, purity specifications, and process robustness. Understanding these details is essential for technical teams evaluating the feasibility of adopting this method for their own production lines or for procurement specialists verifying the quality standards of potential suppliers.
Q: What is the primary advantage of this purification method over column chromatography?
A: The primary advantage is industrial scalability and cost efficiency. Unlike column chromatography, which consumes vast amounts of silica gel and solvents and is difficult to scale beyond kilogram quantities, this recrystallization method utilizes simple heating and cooling cycles suitable for ton-scale production.
Q: What purity levels can be achieved using this crystallization technique?
A: Experimental data from the patent indicates that this method consistently achieves HPLC purity levels exceeding 99.0%, with specific examples reaching up to 99.82%. Additionally, the process significantly reduces colored impurities, resulting in absorbance values at 420nm and 450nm of ≤0.06.
Q: Which solvents are compatible with this purification process?
A: The process is robust across various water-soluble organic solvents. Compatible solvents include C1-C4 alcohols (methanol, ethanol), acetonitrile, acetone, tetrahydrofuran (THF), dimethyl sulfoxide (DMSO), and dioxane, allowing flexibility based on procurement availability and cost.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Abacavir Intermediate Supplier
As the pharmaceutical industry continues to evolve towards more efficient and sustainable manufacturing practices, the adoption of advanced purification technologies like the one described in Patent CN107641122B becomes a key differentiator for suppliers. NINGBO INNO PHARMCHEM stands at the forefront of this evolution, leveraging deep technical expertise to deliver high-quality pharmaceutical intermediates. Our commitment to excellence is backed by extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that we can meet the volumetric demands of global API manufacturers. We operate with stringent purity specifications and utilize rigorous QC labs equipped with state-of-the-art analytical instrumentation to verify that every batch of Abacavir intermediate meets or exceeds the 99.0% HPLC purity benchmark, with tight control over absorbance and residual solvent levels.
We invite international partners to collaborate with us to optimize their supply chains for antiretroviral medications. Our technical team is prepared to provide a Customized Cost-Saving Analysis demonstrating how our optimized crystallization processes can reduce your overall procurement costs without compromising quality. We encourage you to contact our technical procurement team to request specific COA data, route feasibility assessments, and samples for your validation studies. By partnering with NINGBO INNO PHARMCHEM, you secure a reliable source of critical intermediates that supports both your commercial goals and your commitment to delivering life-saving therapies to patients worldwide.
