Revolutionizing Muscone Production: High-Yield Phase Transfer Catalysis for Commercial Scale-Up
The global demand for high-purity synthetic musks, particularly 3-methylcyclopentadecanone (Muscone), continues to surge within the luxury fragrance and fine chemical sectors, driven by the scarcity and ethical concerns surrounding natural musk sources. Patent CN101298413A introduces a transformative manufacturing methodology that addresses the historical bottlenecks of low yield and complex purification inherent in traditional synthesis routes. This technical breakthrough leverages a sophisticated phase transfer catalytic system combined with a titanium-mediated cyclization strategy to achieve unprecedented efficiency. For R&D directors and procurement specialists seeking a reliable fragrance intermediate supplier, this patent represents a pivotal shift towards economically viable and scalable production. The core innovation lies in the precise selection of quaternary ammonium salts with a total carbon count of C12 to C25, which optimizes the interfacial reaction kinetics between organic and aqueous phases. By integrating this advanced catalytic approach, manufacturers can secure a consistent supply of high-purity muscone while drastically mitigating the operational costs associated with legacy processes.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
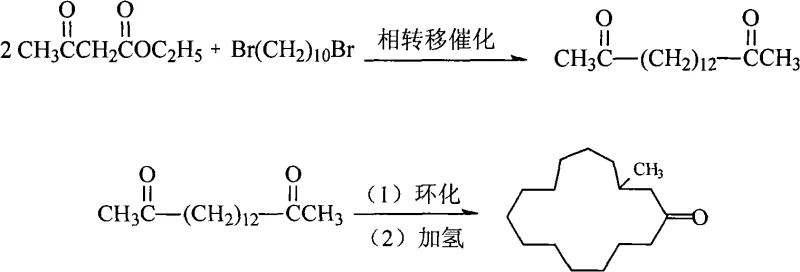
Historically, the synthesis of muscone has been plagued by inefficient methodologies that struggle to balance yield with operational simplicity. The classic Stoll method, which involves the reaction of ethyl acetoacetate with 1,10-dibromodecane followed by aldol condensation and hydrogenation, has long been the benchmark but suffers from severe drawbacks. Experimental data indicates that the Stoll method typically achieves a yield of only around 53% for the critical intermediate, hexadecanedione-(2,15), due to competing side reactions such as C-alkylation, O-alkylation, and self-condensation of the ester. Furthermore, the reliance on strong alkoxide bases often leads to uncontrolled molecular orbital pathways, generating a complex impurity profile that necessitates rigorous and costly purification steps. Even improved variations reported in literature, such as those utilizing benzyltriethylammonium chloride, have only managed to push yields to approximately 78.6%, leaving significant room for optimization in terms of raw material utilization and waste generation. These inefficiencies translate directly into higher production costs and extended lead times, creating supply chain vulnerabilities for downstream perfume manufacturers who require large volumes of consistent quality material.
The Novel Approach
The methodology disclosed in patent CN101298413A fundamentally reengineers the synthesis pathway by introducing a highly selective phase transfer catalyst system tailored for macrocycle formation. Instead of conventional alkoxides, the process employs inorganic bases like carbonates or hydroxides in conjunction with specific long-chain quaternary ammonium salts. This combination facilitates the efficient transport of anionic species into the organic phase, thereby accelerating the nucleophilic substitution reaction between 1,10-dibromodecane and ethyl acetoacetate. The result is a dramatic increase in the yield of hexadecanedione-(2,15) to over 85%, with purity levels exceeding 99.0%. Subsequent steps utilize a titanium-mediated cyclization system involving TiCl4 and organic amines, which promotes the formation of the fifteen-membered ring with a yield surpassing 90%. This novel approach not only simplifies the operational workflow by reducing the number of purification cycles but also enhances the overall atom economy of the process. For stakeholders focused on cost reduction in synthetic musk manufacturing, this route offers a compelling alternative that maximizes output from every kilogram of starting material.
Mechanistic Insights into Phase Transfer Catalytic Condensation and Ti-Mediated Cyclization
The success of this synthesis hinges on the intricate interplay between the phase transfer catalyst structure and the reaction medium. The patent specifies the use of quaternary ammonium salts where the sum of carbon atoms in the four substituent groups ranges from 12 to 25. This specific carbon chain length is critical; it ensures sufficient lipophilicity for the catalyst to reside effectively at the interface of the organic solvent and the aqueous base layer, facilitating the transfer of carbonate or hydroxide ions. When the concentration of these anions in the organic phase is optimized, the rate of the double alkylation reaction increases significantly, favoring the formation of the desired 1,10-diacetyldecane derivative over mono-alkylated byproducts. The use of aprotic polar solvents such as toluene, chloroform, or tetrahydrofuran further stabilizes the transition state, preventing premature hydrolysis of the ester groups before the dialkylation is complete. This mechanistic precision allows for the suppression of oligomerization and other parasitic reactions that typically degrade yield in macrocyclization chemistry.
Following the formation of the linear dione, the cyclization step employs a titanium-mediated mechanism that is distinct from traditional base-catalyzed aldol condensations. The system utilizes TiCl4 in the presence of organic amines like diisobutylamine or triethylamine within a halogenated hydrocarbon solvent. The titanium species likely acts as a Lewis acid, coordinating with the carbonyl oxygens to activate the alpha-protons and direct the intramolecular attack necessary for ring closure. This coordination lowers the activation energy for the formation of the fifteen-membered ring, which is entropically disfavored compared to intermolecular polymerization. The high selectivity of this Ti-mediated system results in a cyclization yield of over 90%, producing dehydromuscone with minimal impurities. Finally, the hydrogenation step uses standard heterogeneous catalysts like Pd/C or Raney Nickel, which can be recovered and reused over 50 times, ensuring that the final reduction of the double bond is both economically and environmentally sustainable. This comprehensive mechanistic control ensures that the final product meets stringent purity specifications required for high-end fragrance applications.
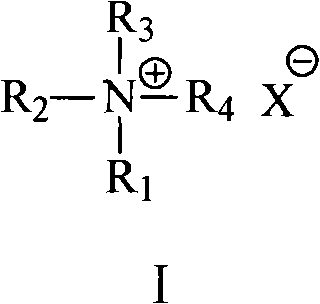
How to Synthesize 3-Methylcyclopentadecanone Efficiently
The synthesis of 3-methylcyclopentadecanone via this patented route involves a carefully orchestrated sequence of condensation, cyclization, and reduction steps that maximize yield while minimizing waste. The process begins with the phase transfer catalytic reaction of 1,10-dibromodecane and ethyl acetoacetate, followed by hydrolysis to obtain the key intermediate hexadecanedione-(2,15). This intermediate is then subjected to titanium-mediated cyclization to form the macrocyclic ring, and finally hydrogenated to saturate the double bond. The detailed standardized synthesis steps, including specific molar ratios, temperature profiles, and workup procedures, are outlined in the guide below to ensure reproducibility and safety in a commercial setting.
- Condense 1,10-dibromodecane with ethyl acetoacetate using a C12-C25 quaternary ammonium phase transfer catalyst and inorganic base to form hexadecanedione-(2,15).
- Perform cyclization of hexadecanedione-(2,15) in a halogenated hydrocarbon solvent using a TiCl4-organic amine system to obtain dehydromuscone.
- Hydrogenate dehydromuscone in a lower aliphatic alcohol solvent using a metal catalyst (Pd/C, Pt/C, or Raney Nickel) to yield final muscone product.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the adoption of this advanced synthesis route offers tangible strategic benefits that extend beyond mere chemical yield. The primary advantage lies in the substantial optimization of raw material consumption; by boosting the yield of the critical intermediate from roughly 53% to over 85%, the process drastically reduces the volume of starting materials required per unit of final product. This efficiency gain translates directly into lower variable costs and a reduced environmental footprint, as less waste solvent and byproduct require disposal. Furthermore, the ability to recover and reuse the hydrogenation catalyst for more than 50 cycles eliminates the recurring expense of precious metal catalysts, providing a significant long-term cost reduction in fragrance intermediate manufacturing. The simplified purification requirements, driven by the high selectivity of the phase transfer and titanium-mediated steps, also shorten the overall production cycle time, allowing for faster turnaround on large-scale orders.
- Cost Reduction in Manufacturing: The implementation of the C12-C25 quaternary ammonium phase transfer catalyst system eliminates the need for expensive and hazardous strong alkoxide bases, replacing them with cheaper inorganic carbonates or hydroxides. This substitution not only lowers the direct cost of reagents but also reduces the safety infrastructure required for handling pyrophoric materials. Additionally, the high yield of the cyclization step (>90%) minimizes the loss of valuable intermediate material, ensuring that the majority of input mass is converted into saleable product. The cumulative effect of these efficiencies is a significantly leaner cost structure that enhances competitiveness in the global fragrance market without compromising on quality.
- Enhanced Supply Chain Reliability: The robustness of this synthetic route contributes to greater supply chain stability by reducing the risk of batch failures and off-spec production. The use of readily available industrial solvents like toluene and dichloromethane, combined with stable inorganic bases, ensures that raw material sourcing is not subject to the volatility associated with specialized reagents. Moreover, the high purity of the intermediate (>99.0%) reduces the burden on downstream quality control laboratories, accelerating the release of finished goods. For supply chain planners, this means more predictable lead times and the ability to scale production volumes rapidly in response to market demand fluctuations, securing a steady flow of high-purity fragrance ingredients.
- Scalability and Environmental Compliance: The process is inherently designed for commercial scale-up, utilizing standard unit operations such as reflux, filtration, and distillation that are easily adaptable from pilot plant to multi-ton production facilities. The reduction in side products and the ability to recycle catalysts align with modern green chemistry principles, facilitating compliance with increasingly stringent environmental regulations. By minimizing the generation of hazardous waste and optimizing energy usage through efficient reaction conditions (e.g., moderate temperatures of 60-90°C), manufacturers can achieve sustainable production goals. This environmental stewardship not only mitigates regulatory risk but also enhances the brand value of the final fragrance products in an eco-conscious consumer market.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the patented synthesis of muscone, derived directly from the experimental data and process descriptions provided in the intellectual property documentation. These insights are intended to clarify the operational parameters and benefits for potential partners evaluating this technology for integration into their supply chains. Understanding these specifics is crucial for assessing the feasibility of adopting this high-yield route for large-scale manufacturing.
Q: What is the key advantage of the new phase transfer catalyst system over the traditional Stoll method?
A: The new method utilizes a specific C12-C25 alkyl quaternary ammonium salt phase transfer catalyst which significantly increases the yield of the intermediate hexadecanedione-(2,15) to over 85%, compared to the traditional Stoll method which often yields less than 53% due to side reactions and poor selectivity.
Q: How does the titanium-mediated cyclization step improve process efficiency?
A: By employing a TiCl4-organic amine system in a halogenated hydrocarbon solvent, the cyclization step achieves a yield of over 90% for dehydromuscone. This high efficiency minimizes waste and simplifies the purification process compared to older ring-closing methods that suffered from low conversion rates.
Q: Is the hydrogenation catalyst reusable in this manufacturing process?
A: Yes, the patent specifies that the hydrogenation catalyst, such as Pd/C or Raney Nickel, can be recovered by filtration after the reaction and reused generally more than 50 times without significant loss of activity, contributing to substantial long-term cost reductions.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 3-Methylcyclopentadecanone Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical importance of efficient and scalable synthesis routes in the competitive landscape of fine chemicals and fragrances. Our team of expert chemists possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the theoretical advantages of patent CN101298413A are fully realized in practical manufacturing environments. We are committed to delivering high-purity 3-methylcyclopentadecanone that meets stringent purity specifications, supported by our rigorous QC labs equipped with state-of-the-art analytical instrumentation. Our capability to implement complex phase transfer catalytic systems and titanium-mediated reactions allows us to offer a superior product profile that outperforms traditional market offerings in both consistency and cost-effectiveness.
We invite global fragrance houses and chemical distributors to collaborate with us to leverage this advanced technology for their supply needs. By partnering with NINGBO INNO PHARMCHEM, you gain access to a Customized Cost-Saving Analysis tailored to your specific volume requirements and quality standards. We encourage you to contact our technical procurement team today to request specific COA data and route feasibility assessments, ensuring that your next project is built on a foundation of chemical excellence and supply chain reliability.
