Revolutionizing Muscone Production: High-Yield Enolate Trapping Technology for Commercial Scale-Up
The global demand for high-quality macrocyclic ketones, particularly muscone, has driven significant innovation in synthetic organic chemistry, aiming to replicate the olfactory profile of natural musk without the ethical and supply chain constraints of animal-derived sources. Patent CN101056843A introduces a transformative methodology for the production of muscone and its intermediates, specifically addressing the longstanding inefficiencies in the 1,4-conjugate methyl addition reaction of 2-cyclopentadecenone. This technical breakthrough shifts the paradigm from energy-intensive, low-concentration processes to a robust, high-yield manufacturing route suitable for industrial application. By employing a novel enolate anion-trapping strategy, the invention successfully mitigates the formation of high molecular weight by-products that have historically plagued this synthesis, thereby enabling reactions to proceed at practical concentrations and temperatures. This advancement is critical for manufacturers seeking to optimize their production lines for fine chemical intermediates while maintaining stringent purity standards required by the flavors and fragrances industry.
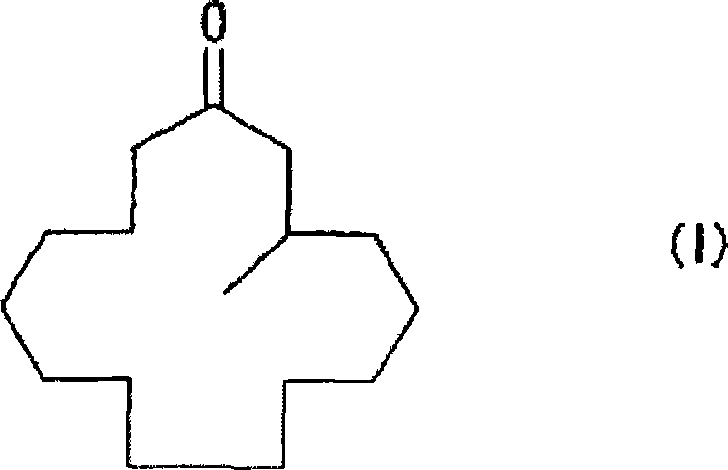
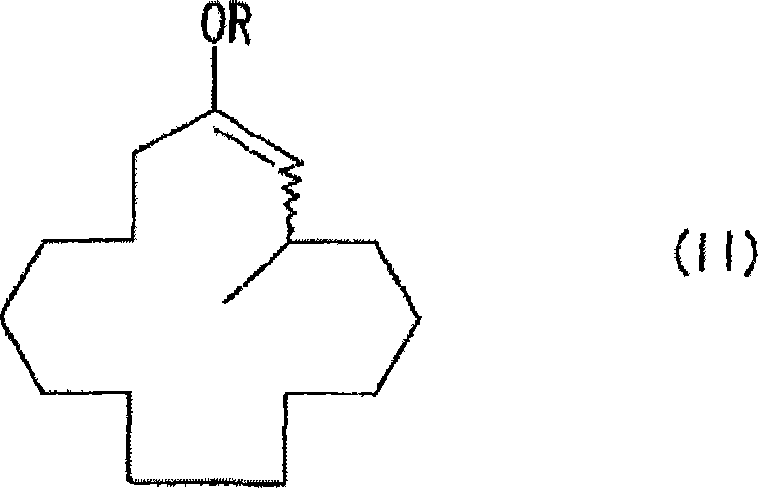
The core of this innovation lies in the stabilization of the reactive intermediate formed during the methylation step. In traditional synthetic routes, the enolate anion generated after the 1,4-addition is highly reactive and prone to undesirable side reactions, such as self-condensation or polymerization, especially when reaction concentrations are increased to improve throughput. The patented process overcomes this by introducing a specific enolate anion-trapping agent immediately following the addition of the organometallic methylating reagent. This trapping agent converts the unstable enolate into a stable 3-methyl-1-cyclopentadecene derivative, represented generally by Formula (II). This intermediate can be isolated or processed in situ, providing a controlled pathway to the final ketone. Subsequent solvolysis of this protected intermediate releases the target muscone molecule with exceptional efficiency. This two-step sequence—addition followed by trapping and then deprotection—represents a significant departure from direct ketone formation methods that often struggle with selectivity issues.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Prior art methodologies for synthesizing muscone via 1,4-conjugate addition have been severely constrained by thermodynamic and kinetic limitations that hinder commercial viability. Historical data indicates that achieving acceptable yields often necessitates operating under extremely low temperatures, frequently as low as -78°C, which imposes a heavy burden on energy consumption and requires specialized cryogenic equipment. Furthermore, to suppress the formation of polymeric by-products, these conventional reactions must be conducted at very low concentrations, often requiring solvent-to-substrate ratios as high as 50:1 or greater. This excessive dilution drastically reduces the volumetric productivity of the reactor, meaning that massive amounts of solvent must be handled, recovered, and recycled for every unit of product generated. Additionally, even under these苛刻 conditions, reported yields have often remained suboptimal, hovering around 53% in some documented cases, which translates to significant raw material waste and increased cost of goods sold. The reliance on stoichiometric amounts of chiral auxiliaries in some asymmetric variants further exacerbates the cost and complexity, making large-scale production economically challenging.
The Novel Approach
The novel approach detailed in the patent data fundamentally alters the reaction landscape by decoupling the addition step from the instability of the resulting enolate. By introducing an enolate trapping agent, such as acid anhydrides, chloroformates, or silyl halides, the process effectively 'freezes' the intermediate in a stable form before side reactions can occur. This stabilization allows the reaction to be performed at significantly higher concentrations, potentially reducing solvent usage by a substantial margin compared to traditional dilute methods. Moreover, the operating temperature window is widened considerably, with effective reactions occurring between -30°C and 30°C, eliminating the need for extreme cryogenic cooling. This shift not only lowers energy costs but also simplifies the engineering requirements for the reaction vessel. The ability to isolate the stable intermediate, such as the 3-methyl-1-cyclopentadecenyl ester or ether shown in the structural diagrams, provides flexibility in manufacturing, allowing for quality control checks before the final solvolysis step. This results in a more robust process capable of delivering high yields, often exceeding 90% in optimized examples, thereby maximizing atom economy and minimizing waste generation.
Mechanistic Insights into Copper-Catalyzed 1,4-Conjugate Addition
The mechanistic foundation of this synthesis relies on the precise coordination of a copper or nickel catalyst with an organometallic methylating reagent, such as dimethylzinc, to facilitate the 1,4-conjugate addition to the alpha,beta-unsaturated ketone substrate. The catalyst activates the organometallic species, enabling the nucleophilic attack at the beta-position of the 2-cyclopentadecenone ring. In the absence of a trapping agent, the resulting copper-enolate species is susceptible to protonation or further reaction, leading to impurities. However, in this patented system, the presence of the trapping agent intercepts the enolate oxygen, forming a covalent bond that generates the stable Formula (II) derivative. This step is kinetically favored over the decomposition pathways, ensuring that the majority of the substrate is converted into the desired intermediate. For the production of optically active (R)-muscone, which possesses three times the fragrance intensity of its (S)-enantiomer, the system incorporates chiral phosphorus ligands. These ligands create a chiral environment around the metal center, inducing asymmetry during the carbon-carbon bond formation. The stereochemical outcome is dictated by the specific geometry of the ligand, such as the binaphthyl-based phosphoramidites described in the patent, which transfer their chirality to the newly formed stereocenter at the 3-position of the macrocycle.
Controlling the impurity profile is paramount in the production of fragrance ingredients, where even trace contaminants can alter the olfactory character. The enolate trapping mechanism serves as a powerful tool for impurity control by preventing the oligomerization of the macrocyclic ring, a common side reaction in large-ring chemistry. By converting the reactive enolate into a stable ester, carbonate, or silyl ether, the process effectively shuts down pathways that lead to high molecular weight tars or polymers. This results in a cleaner crude reaction mixture, which simplifies downstream purification steps such as distillation or chromatography. Furthermore, the subsequent solvolysis step, which removes the protecting group to reveal the ketone, is highly selective. Whether using basic hydrolysis for esters or acidic conditions for ethers, this final transformation proceeds with minimal racemization, preserving the optical purity established in the initial addition step. This high fidelity in stereochemical retention ensures that the final muscone product meets the rigorous enantiomeric excess specifications demanded by high-end perfumery applications.
How to Synthesize Muscone Efficiently
The synthesis of muscone via this patented route involves a carefully orchestrated sequence of catalytic addition, intermediate stabilization, and final deprotection. The process begins with the preparation of the reaction vessel under an inert atmosphere, followed by the introduction of the copper catalyst and the chiral ligand if optical activity is desired. The organometallic reagent is then added to generate the active catalytic species, after which the 2-cyclopentadecenone substrate and the trapping agent are introduced, often in a controlled manner to manage exotherms. The reaction is allowed to proceed until conversion is complete, monitored by techniques such as gas chromatography. Once the stable intermediate is formed, the reaction mixture is quenched and worked up to isolate the 3-methyl-1-cyclopentadecene derivative. In the final stage, this intermediate is subjected to solvolysis conditions tailored to the specific protecting group used, yielding the target muscone. Detailed standard operating procedures for scaling this pathway from laboratory to production are outlined in the technical guide below.
- Perform 1,4-conjugate methyl addition on 2-cyclopentadecenone using a copper catalyst and organometallic reagent.
- Trap the generated enolate anion immediately with a scavenger to form a stable 3-methyl-1-cyclopentadecene derivative.
- Execute solvolysis on the derivative intermediate to release the final muscone product with high optical purity.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain directors, the adoption of this enolate trapping technology offers compelling advantages that directly impact the bottom line and operational resilience. The primary benefit stems from the drastic improvement in process efficiency, which translates into tangible cost reductions across the manufacturing value chain. By enabling reactions to run at higher concentrations, the volume of solvent required per kilogram of product is significantly decreased. This reduction not only lowers the direct cost of purchasing solvents but also diminishes the energy load associated with heating, cooling, and recovering these vast quantities of liquid waste. Furthermore, the elimination of extreme cryogenic conditions reduces the dependency on specialized low-temperature infrastructure, allowing production to occur in standard stainless steel reactors equipped with conventional cooling systems. This flexibility enhances asset utilization and reduces capital expenditure requirements for new production lines.
- Cost Reduction in Manufacturing: The economic impact of this technology is driven by the substantial increase in yield and the reduction in processing time. Traditional methods often suffer from yields that leave nearly half of the valuable starting material unconverted or lost to by-products. In contrast, this high-yield pathway ensures that a much larger proportion of raw materials are converted into saleable product, effectively lowering the raw material cost per unit. Additionally, the simplified workup and purification processes, resulting from a cleaner reaction profile, reduce the consumption of auxiliary chemicals and the labor hours required for separation. The avoidance of expensive chiral auxiliaries in favor of catalytic chiral ligands further drives down costs, as the ligand can be used in sub-stoichiometric amounts and potentially recovered. These factors combine to create a leaner, more cost-effective manufacturing model that improves margin potential in a competitive market.
- Enhanced Supply Chain Reliability: Supply chain continuity is often threatened by complex processes that are sensitive to minor variations in conditions. The robustness of this new method, with its wider operating temperature window and tolerance for higher concentrations, makes it less prone to batch failures and deviations. This reliability ensures a more consistent output of high-purity muscone, reducing the risk of stockouts and delivery delays. Moreover, the intermediates generated in this process are stable and can be stored, offering a strategic buffer in the production schedule. Manufacturers can produce the intermediate in bulk during periods of low demand and convert it to the final product as needed, providing greater agility in responding to market fluctuations. This decoupling of synthesis steps adds a layer of security to the supply chain, ensuring that downstream customers receive their orders on time.
- Scalability and Environmental Compliance: Scaling chemical processes from the bench to the plant floor is fraught with challenges, particularly when dealing with exothermic reactions and hazardous reagents. This technology is inherently scalable because the heat management is more manageable at higher concentrations compared to the massive solvent volumes of dilute methods. The reduced solvent usage also aligns with modern environmental, health, and safety (EHS) goals by minimizing the generation of volatile organic compounds (VOCs) and hazardous waste streams. The process avoids the use of stoichiometric heavy metal reagents where possible, favoring catalytic systems that are easier to manage from a waste disposal perspective. This environmental compatibility facilitates regulatory compliance and supports sustainability initiatives, which are increasingly important criteria for procurement decisions in the global fine chemical industry.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this muscone synthesis technology. These answers are derived from the specific embodiments and comparative data presented in the patent literature, providing a clear understanding of the process capabilities and limitations. Understanding these details is essential for technical teams evaluating the feasibility of integrating this route into their existing manufacturing portfolios.
Q: How does the enolate trapping method improve muscone yield compared to traditional methods?
A: Traditional methods often suffer from low yields due to polymerization side reactions at high concentrations. The enolate trapping method stabilizes the reactive intermediate, preventing these side reactions and allowing for higher concentration processing, which significantly boosts overall yield.
Q: What are the temperature requirements for this new muscone synthesis process?
A: Unlike conventional methods requiring cryogenic conditions around -78°C, this patented process operates effectively at much milder temperatures, typically between -30°C and 30°C, reducing energy consumption and equipment complexity.
Q: Can this process produce optically active (R)-muscone?
A: Yes, by utilizing specific chiral phosphorus ligands during the copper-catalyzed addition step, the process achieves high enantiomeric excess, producing optically active (R)-muscone which possesses superior fragrance intensity.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Muscone Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical importance of efficient and scalable synthesis routes for high-value fragrance ingredients like muscone. Our team of expert chemists has thoroughly analyzed the potential of this enolate trapping technology and is well-equipped to translate these patent insights into commercial reality. We possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the transition from laboratory success to industrial output is seamless. Our facilities are designed to handle the specific requirements of organometallic chemistry, including strict moisture control and inert atmosphere operations, guaranteeing the stringent purity specifications required for premium fragrance applications. With our rigorous QC labs and state-of-the-art analytical capabilities, we ensure that every batch of muscone meets the highest standards of quality and consistency.
We invite you to collaborate with us to optimize your supply chain for muscone and related macrocyclic ketones. By leveraging our technical expertise and manufacturing capacity, you can achieve a Customized Cost-Saving Analysis tailored to your specific volume requirements. We encourage you to contact our technical procurement team to request specific COA data and route feasibility assessments. Let us help you secure a stable, high-quality supply of this essential ingredient, driving value and innovation in your product formulations.
Engineering Bottleneck?
Can't scale up this synthesis? Upload your target structure or CAS, and our CDMO team will evaluate the industrial feasibility within 24 hours. Request Evaluation →
