Advanced Synthesis of Sitagliptin Intermediates via Optimized Hydrogenation for Commercial Scale-up
Advanced Synthesis of Sitagliptin Intermediates via Optimized Hydrogenation for Commercial Scale-up
The pharmaceutical landscape for Type 2 diabetes treatment has been profoundly shaped by the introduction of Dipeptidyl Peptidase-IV (DPP-IV) inhibitors, with Sitagliptin standing as a cornerstone therapy since its FDA approval. As detailed in patent CN102126976A, the demand for efficient, cost-effective, and high-purity synthetic routes for Sitagliptin and its key intermediates has never been more critical for global supply chains. This technical insight report analyzes a novel preparation method that addresses the longstanding bottlenecks in producing the chiral beta-amino acid precursors essential for this blockbuster drug. By leveraging a refined condensation and hydrogenation strategy, manufacturers can now access a pathway that drastically simplifies the synthetic sequence while enhancing the overall stereochemical integrity of the final product.
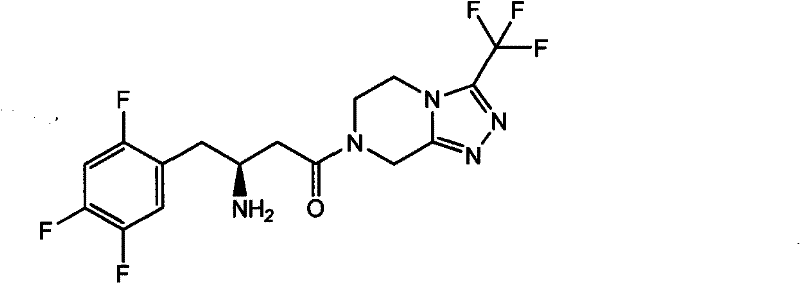
The core innovation lies in the strategic deployment of specific chiral amines to construct the carbon backbone, bypassing the need for excessive chiral resolution steps that plague earlier generations of synthesis. For R&D directors and process chemists, this represents a shift towards more atom-economical and operationally simple protocols. The patent discloses intermediates of Formula II and Formula III, which serve as pivotal junction points in the synthesis. These structures are not merely theoretical; they are validated through rigorous experimental examples demonstrating yields exceeding 90% in the condensation step and greater than 97% in the subsequent reduction, setting a new benchmark for efficiency in API intermediate manufacturing.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Prior art methodologies, such as those disclosed in WO2004085661 and related patents, have historically relied on complex multi-step sequences that often necessitate the use of substantial quantities of high-cost chiral catalysts and noble metal ligand complexes. A significant drawback of these conventional routes is the poor recovery rate and limited reusability of these expensive catalytic systems, which directly inflates the cost of goods sold (COGS) for the final active pharmaceutical ingredient. Furthermore, traditional pathways frequently involve harsh reaction conditions or difficult purification scenarios, such as extensive column chromatography, which are inherently problematic when transitioning from laboratory benchtop to kiloton-scale industrial reactors. The reliance on amide-substituted aniline derivatives in some older schemes also limits the flexibility of the process, often resulting in intermediates with suboptimal physical properties for downstream processing.
The Novel Approach
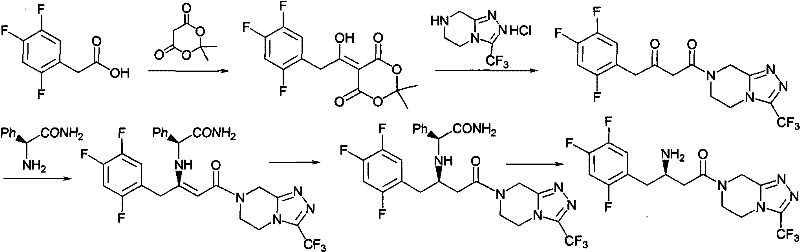
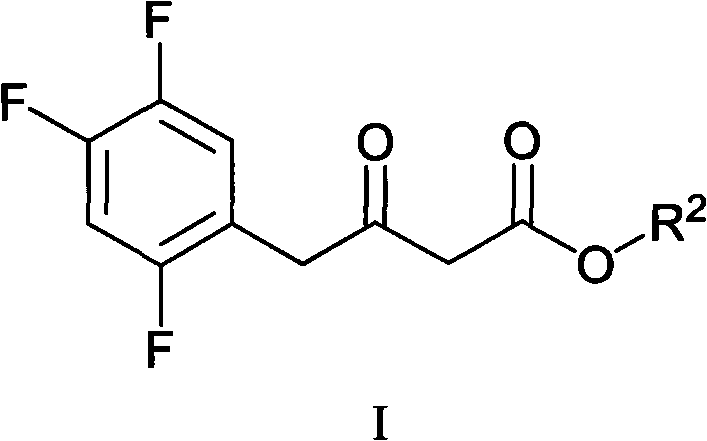
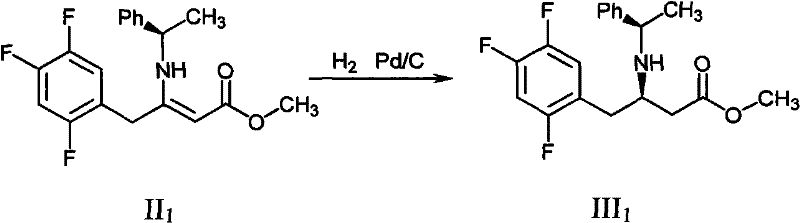
In stark contrast, the novel approach presented in CN102126976A introduces a streamlined two-stage protocol that begins with the condensation of a beta-keto ester (Formula I) with a chiral phenylethylamine derivative. This reaction proceeds under mild thermal conditions in common alcoholic solvents, yielding the enamine intermediate (Formula II) with exceptional conversion rates. The subsequent transformation involves a catalytic hydrogenation step that is remarkably robust, utilizing standard heterogeneous catalysts like Pd/C or Pt/Al2O3 under moderate hydrogen pressure (15-20 atm). This methodology effectively circumvents the need for homogeneous chiral catalysts that are difficult to remove and recycle. The result is a process that not only reduces the difficulty of synthesis but also significantly improves the purity profile of the product, making it ideally suited for reliable sitagliptin intermediate supplier operations aiming for GMP compliance.
Mechanistic Insights into Asymmetric Hydrogenation and Chiral Transfer
The mechanistic elegance of this synthesis rests on the concept of chiral pool utilization combined with diastereoselective hydrogenation. In the first stage, the reaction between the achiral beta-keto ester and the chiral (R)-1-phenylethylamine generates an enamine intermediate where the chirality of the amine auxiliary exerts a powerful stereochemical influence on the adjacent double bond. This pre-organization of the molecular geometry is crucial, as it sets the stage for the subsequent reduction. When the enamine is subjected to hydrogenation over a palladium or platinum surface, the bulky chiral auxiliary shields one face of the double bond, forcing the addition of hydrogen to occur predominantly from the less hindered face. This diastereoselective addition ensures that the newly formed stereocenter at the beta-position is established with high fidelity, effectively transferring the chirality from the inexpensive amine auxiliary to the carbon backbone of the drug molecule.
Furthermore, the choice of catalyst and reaction parameters plays a pivotal role in impurity control. The patent data indicates that using 5% Pt/Al2O3 or 10% Pd/C at temperatures around 35°C allows for precise control over the reduction kinetics. This minimizes side reactions such as over-reduction or racemization, which are common pitfalls in beta-amino acid synthesis. The high enantiomeric excess (ee > 98%) reported in the examples underscores the effectiveness of this mechanistic design. For quality assurance teams, this implies a much cleaner impurity profile, reducing the burden on downstream purification units and ensuring that the final API meets the rigorous specifications required by regulatory bodies like the FDA and EMA for diabetes medications.
How to Synthesize Sitagliptin Intermediates Efficiently
The practical execution of this synthesis involves dissolving the beta-keto ester precursor in a lower alcohol such as methanol or ethanol, followed by the addition of the chiral amine and a catalytic amount of acid to facilitate enamine formation. The mixture is heated gently to promote condensation, after which the intermediate is isolated via crystallization using heptane as an anti-solvent to maximize recovery. The detailed standardized synthesis steps see the guide below for specific molar ratios and aging times that ensure optimal crystal habit and purity.
- React the beta-keto ester precursor (Formula I) with (R)-1-phenylethylamine in an alcoholic solvent (methanol or ethanol) at 40-48°C to form the enamine intermediate (Formula II).
- Isolate the Formula II intermediate via crystallization, utilizing heptane as an anti-solvent to ensure high purity before the reduction step.
- Perform asymmetric hydrogenation of Formula II using a palladium or platinum catalyst (e.g., Pd/C or Pt/Al2O3) under 16-18 atm hydrogen pressure at 35°C to yield the chiral amine intermediate (Formula III).
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the adoption of this novel synthetic route offers transformative benefits that extend far beyond simple yield improvements. The primary advantage lies in the drastic simplification of the material input list; by eliminating the need for exotic, proprietary chiral ligands and replacing them with commodity chemicals like (R)-1-phenylethylamine and standard heterogeneous catalysts, the supply chain becomes significantly more resilient. This reduction in specialized raw material dependency mitigates the risk of supply disruptions and price volatility, ensuring a steady flow of intermediates necessary for continuous API manufacturing. Moreover, the operational simplicity of the process—utilizing standard autoclaves and filtration equipment—means that existing manufacturing infrastructure can be easily adapted without requiring massive capital expenditure on new reactor types or containment systems.
- Cost Reduction in Manufacturing: The most immediate financial impact is derived from the substantial decrease in catalyst consumption. Traditional routes often require stoichiometric or near-stoichiometric amounts of expensive chiral reagents, whereas this method utilizes catalytic quantities of recoverable metals like palladium or platinum. This shift from consumable chiral reagents to recyclable heterogeneous catalysts fundamentally alters the cost structure, leading to significant savings in raw material costs. Additionally, the high yields reported (>90% for condensation, >97% for hydrogenation) minimize waste generation and solvent usage, further driving down the variable costs associated with large-scale production runs.
- Enhanced Supply Chain Reliability: The reliance on widely available starting materials enhances the robustness of the supply chain. Beta-keto esters and phenylethylamines are produced by numerous global chemical suppliers, reducing the risk of single-source bottlenecks. This diversification of the supplier base allows procurement teams to negotiate better terms and secure long-term contracts with greater confidence. Furthermore, the stability of the intermediates (Formula II and III) allows for flexible inventory management; they can be produced in bulk and stored safely, decoupling the production of the intermediate from the immediate demand for the final API, thus smoothing out production schedules and improving overall logistical efficiency.
- Scalability and Environmental Compliance: From an environmental and regulatory standpoint, this process aligns perfectly with green chemistry principles. The use of alcoholic solvents and the avoidance of chlorinated solvents or heavy metal contaminants simplifies waste treatment and disposal. The heterogeneous nature of the hydrogenation catalyst facilitates easy separation via filtration, preventing metal leaching into the product stream and reducing the load on wastewater treatment facilities. This environmental compatibility not only reduces compliance costs but also future-proofs the manufacturing process against increasingly stringent environmental regulations, ensuring long-term operational continuity for the facility.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this synthesis route. These answers are derived directly from the experimental data and technical disclosures within the patent, providing a factual basis for decision-making regarding technology transfer and process validation. Understanding these nuances is essential for project managers evaluating the feasibility of integrating this chemistry into their existing portfolios.
Q: How does this new route reduce production costs compared to prior art?
A: The process significantly lowers the loading of expensive noble metal catalysts (Pd/Pt) required for the hydrogenation step. Additionally, the use of readily available chiral auxiliaries and simplified purification steps eliminates the need for complex chromatographic separations often found in older methods.
Q: What is the stereochemical purity (ee) achievable with this method?
A: The optimized hydrogenation conditions, specifically utilizing 5% Pt/Al2O3 or 10% Pd/C under controlled pressure (16-18 atm), consistently deliver intermediates with an enantiomeric excess (ee) greater than 98%, meeting stringent pharmaceutical specifications.
Q: Is this synthesis route scalable for industrial manufacturing?
A: Yes, the method is explicitly designed for industrial application. It utilizes standard unit operations such as autoclave hydrogenation and crystallization, avoiding exotic reagents or extreme conditions, thereby ensuring robust scalability from pilot plant to multi-ton commercial production.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Sitagliptin Intermediate Supplier
At NINGBO INNO PHARMCHEM, we recognize that the transition from patent literature to commercial reality requires a partner with deep technical expertise and robust manufacturing capabilities. Our team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the promising metrics seen in the lab are faithfully reproduced on the plant floor. We maintain stringent purity specifications and operate rigorous QC labs equipped with state-of-the-art analytical instrumentation to guarantee that every batch of sitagliptin intermediate meets the highest global standards for safety and efficacy.
We invite you to engage with our technical procurement team to discuss how this optimized synthesis route can be tailored to your specific volume requirements. By requesting a Customized Cost-Saving Analysis, you can gain a clear understanding of the economic benefits specific to your supply chain. We encourage potential partners to contact us directly to obtain specific COA data and route feasibility assessments, allowing you to make informed decisions that drive value and efficiency in your diabetes care portfolio.
