Advanced Manufacturing of 4-Aminoindan Derivatives for High-Purity Fungicide Intermediates
The chemical landscape for producing bioactive heterocycles is constantly evolving, driven by the need for more sustainable and cost-effective manufacturing routes. Patent CN109071439B introduces a groundbreaking methodology for the preparation of 4-aminoindan derivatives of formula (I) and their subsequent conversion into aminoindanamides of formula (II), which possess significant fungicidal activity. This technology addresses long-standing inefficiencies in the synthesis of these critical scaffolds by fundamentally reordering the reaction sequence. Unlike traditional approaches that struggle with the acylation of sterically hindered secondary amines, this invention utilizes a reversed hydrogenation and condensation strategy. This strategic pivot not only streamlines the synthetic pathway but also facilitates the use of environmentally friendlier solvents, positioning it as a superior choice for industrial scale-up. For stakeholders seeking a reliable agrochemical intermediate supplier, understanding this mechanistic shift is crucial for securing long-term supply chain stability.
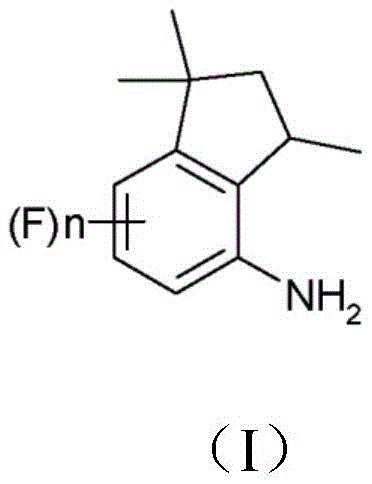
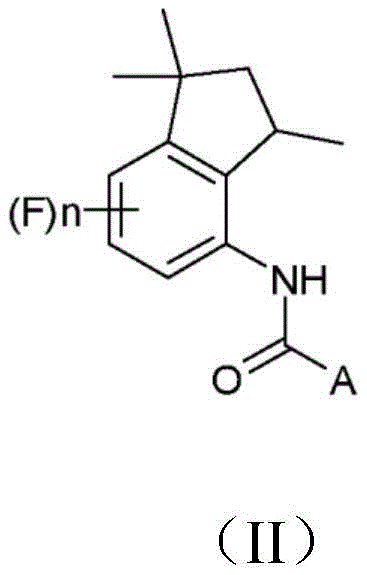
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Prior art methodologies, such as those disclosed in WO2013/186325, typically rely on a sequence where a substituted dihydroquinoline is hydrogenated to a tetrahydroquinoline, which is then acylated before undergoing acid rearrangement. A critical bottleneck in this conventional workflow is the chemical difficulty associated with acylating the secondary amine within the tetrahydroquinoline ring. From a process chemistry perspective, this transformation is sluggish and often requires forcing conditions, including the addition of excessive amounts of base and the use of chlorinated organic solvents to drive the reaction to completion. Furthermore, these harsh conditions frequently lead to relatively low overall yields and significant degradation of expensive reagents, such as pyrazole acid chloride derivatives. The necessity for multiple solvent systems—often involving tetrahydrofuran, methanol, and aqueous acids in different steps—complicates the workup procedures and generates substantial chemical waste, making cost reduction in fungicide manufacturing challenging under these legacy protocols.
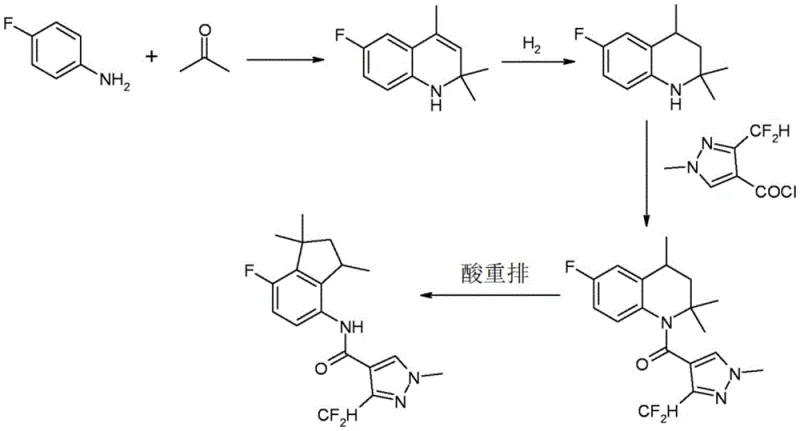
The Novel Approach
The inventive process described in CN109071439B overcomes these hurdles by surprisingly reversing the order of the hydrogenation and condensation (acylation) steps. By first subjecting the 1,2-dihydroquinoline of formula (IV) to catalytic hydrogenation to yield the tetrahydroquinoline of formula (V), and subsequently performing the acylation, the method avoids the difficult acylation of the free secondary amine under basic conditions. Instead, the acylation can be conducted more efficiently, often without the need for additional solvents or with the use of simple aliphatic hydrocarbons. This reversal allows the entire production sequence, from hydrogenation through to the final rearrangement, to potentially utilize a single type of organic solvent, such as heptane. This uniformity drastically simplifies the isolation and purification stages, as intermediates do not require rigorous drying or solvent swapping. Consequently, this approach offers a more cost-effective route to high-purity aminoindanamide, enhancing the commercial viability of large-scale production.
Mechanistic Insights into Acid-Catalyzed Rearrangement and Hydrogenation
The core of this synthetic innovation lies in the precise control of the catalytic hydrogenation and the subsequent acid-catalyzed rearrangement. In the initial step, the 1,2-dihydroquinoline is reduced using a heterogeneous metal catalyst, preferably palladium on charcoal, under mild hydrogen pressure. This step is critical for establishing the saturated nitrogen-containing ring required for the downstream rearrangement. The choice of solvent here is pivotal; aliphatic hydrocarbons like heptane are preferred not only for their safety profile but because they maintain the solubility of the intermediates while allowing for easy catalyst filtration. Following acylation, the resulting acyl-tetrahydroquinoline undergoes a skeletal rearrangement in the presence of a strong mineral acid, such as sulfuric acid or orthophosphoric acid. This acid-mediated transformation involves the migration of the acyl group and the contraction of the six-membered nitrogen heterocycle into the five-membered carbocyclic indan system. The mechanism proceeds through protonation of the amide nitrogen, facilitating the cleavage of the carbon-nitrogen bond and the formation of a new carbon-carbon bond, ultimately yielding the acyl indan compound of formula (VII).
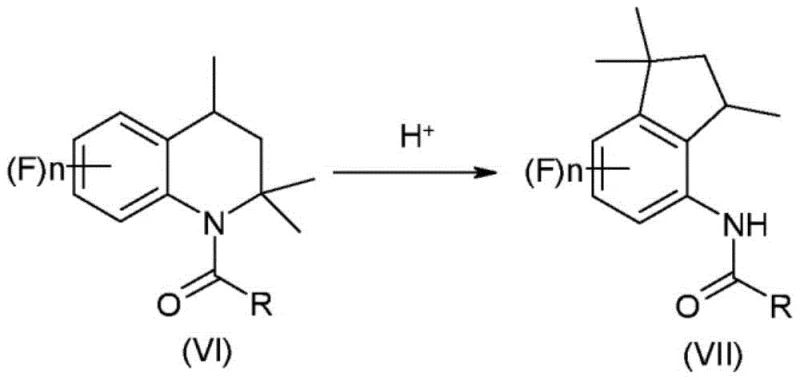
Controlling impurities during this rearrangement is essential for achieving the stringent purity specifications required for agrochemical applications. The use of concentrated sulfuric acid (80% to 98%) at controlled temperatures between 10°C and 60°C ensures substantially complete conversion while minimizing side reactions such as polymerization or over-sulfonation. The process further benefits from the ability to telescope steps; for instance, the acyl indan compound (VII) does not need to be isolated before hydrolysis. By simply diluting the acidic reaction mixture with water and heating, the acyl group is hydrolyzed to reveal the free amine as an acid addition salt. This salt can then be neutralized with a base like sodium hydroxide to liberate the final 4-aminoindan derivative. This telescoped approach minimizes material handling and exposure to air, thereby reducing the formation of oxidative impurities and ensuring a cleaner final product profile suitable for sensitive biological applications.
How to Synthesize 4-Aminoindan Derivatives Efficiently
The synthesis of these valuable intermediates begins with the condensation of an aniline derivative with acetone to form the dihydroquinoline precursor, followed by the key hydrogenation and rearrangement steps detailed in the patent. The process is designed to be robust and scalable, utilizing common industrial reagents and equipment. Operators should pay close attention to temperature control during the exothermic acid dissolution and rearrangement phases to ensure safety and reproducibility. The detailed standardized synthesis steps, including specific molar ratios, reaction times, and workup procedures, are outlined in the guide below to assist technical teams in replicating this high-efficiency route.
- Hydrogenate 1,2-dihydroquinoline (IV) using a palladium catalyst in a hydrocarbon solvent to form tetrahydroquinoline (V).
- Acylate the tetrahydroquinoline (V) with a carboxylic acid derivative such as acetic anhydride to yield acyl derivative (VI).
- Subject the acyl derivative (VI) to acid-catalyzed rearrangement using sulfuric or phosphoric acid to form the acyl indan compound (VII).
- Hydrolyze the acyl group of compound (VII) under acidic then basic conditions to isolate the final 4-aminoindan derivative (I).
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the adoption of this novel synthetic route translates into tangible operational improvements and risk mitigation. The primary advantage lies in the drastic simplification of the solvent management system. By enabling the use of a single solvent type, such as heptane, throughout the majority of the synthesis, the process eliminates the logistical burden and cost associated with sourcing, storing, and disposing of multiple diverse solvent classes like chlorinated hydrocarbons and polar aprotic solvents. This uniformity also streamlines the recovery and recycling of solvents, leading to substantial cost savings in raw material consumption. Furthermore, the ability to recover and reuse the heterogeneous hydrogenation catalyst and unreacted starting anilines directly contributes to a lower cost of goods sold (COGS), making the final API intermediate more price-competitive in the global market.
- Cost Reduction in Manufacturing: The elimination of complex solvent switching and the ability to telescope multiple reaction steps without intermediate isolation significantly reduces processing time and energy consumption. By avoiding the need for expensive chlorinated solvents and excessive bases required in conventional acylation methods, the overall chemical cost per kilogram is optimized. Additionally, the high efficiency of the rearrangement step minimizes the loss of valuable acylating agents, ensuring that expensive reagents are utilized effectively rather than being degraded in side reactions.
- Enhanced Supply Chain Reliability: The reliance on readily available commodity chemicals such as acetone, aniline derivatives, and common mineral acids ensures a stable supply of raw materials, reducing the risk of production delays caused by specialty reagent shortages. The robustness of the catalytic hydrogenation step, which can be performed at atmospheric or low pressure, allows for flexibility in manufacturing site selection and equipment requirements. This adaptability ensures consistent delivery schedules and strengthens the resilience of the supply chain against external disruptions.
- Scalability and Environmental Compliance: The process is inherently designed for commercial scale-up, with reaction conditions that are easily manageable in large-scale reactors. The reduction in hazardous waste generation, particularly the avoidance of chlorinated solvent waste streams, simplifies environmental compliance and waste treatment protocols. This greener manufacturing footprint not only aligns with increasingly strict global environmental regulations but also enhances the corporate sustainability profile of the manufacturing partner, appealing to eco-conscious end-users.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this patented technology. These answers are derived directly from the experimental data and beneficial effects described in the patent documentation, providing clarity on the process capabilities and advantages. Understanding these details is vital for technical procurement teams evaluating the feasibility of integrating this route into their existing supply networks.
Q: How does this new process improve upon conventional synthesis methods for aminoindanamides?
A: Conventional methods typically require acylating a secondary amine in a tetrahydroquinoline ring, which is chemically difficult and necessitates harsh conditions like excess base and chlorinated solvents. This novel approach reverses the sequence by hydrogenating first, then acylating, which significantly simplifies the reaction conditions and allows for the use of a single solvent type throughout the process.
Q: What are the key advantages regarding solvent usage and waste reduction?
A: The process enables the use of a single type of organic solvent, such as aliphatic hydrocarbons like heptane, across multiple steps including hydrogenation, acylation, and rearrangement. This eliminates the need for solvent switching and complex purification between steps, drastically reducing chemical waste and operational complexity compared to prior art methods that require THF, methanol, and sulfuric acid mixtures.
Q: Can the catalysts and starting materials be recycled in this manufacturing route?
A: Yes, the process is designed for high efficiency and sustainability. The heterogeneous metal catalyst used in the hydrogenation step can be recovered and reused in subsequent batches. Additionally, unreacted aniline derivatives from the initial condensation step can be recovered via fractional distillation and recycled, maximizing raw material utilization and lowering overall production costs.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 4-Aminoindan Derivatives Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical role that high-quality intermediates play in the development of next-generation agrochemicals and pharmaceuticals. Our team of expert chemists has thoroughly analyzed the process described in CN109071439B and is fully equipped to translate this laboratory-scale innovation into commercial reality. We possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the benefits of this streamlined synthesis are realized at an industrial level. Our state-of-the-art facilities are supported by rigorous QC labs capable of meeting stringent purity specifications, guaranteeing that every batch of 4-aminoindan derivatives meets the exacting standards required for fungicide manufacturing.
We invite you to collaborate with us to leverage this advanced technology for your specific project needs. Our technical procurement team is ready to provide a Customized Cost-Saving Analysis tailored to your volume requirements, demonstrating how this efficient route can optimize your budget. Please contact us today to request specific COA data and route feasibility assessments, and let us help you secure a reliable supply of these essential building blocks for your future success.
