Advanced Synthesis of Substituted 4-Aminoindan Derivatives for High-Performance Agrochemical Intermediates
Introduction to Novel 4-Aminoindan Synthesis Technology
The development of efficient synthetic routes for biologically active compounds remains a cornerstone of modern agrochemical innovation. Patent CN110799491A introduces a groundbreaking methodology for the preparation of substituted 4-aminoindan derivatives, which serve as critical scaffolds in the synthesis of potent fungicides and crop protection agents. This technology addresses long-standing challenges in the industry by utilizing anhydrous hydrogen fluoride (HF) to mediate a highly selective rearrangement of tetrahydroquinoline precursors. Unlike traditional methods that rely on harsh mineral acids or expensive transition metals, this approach operates under remarkably mild conditions, typically between 0°C and 30°C, ensuring high selectivity and minimizing side reactions. For R&D directors and procurement specialists seeking reliable agrochemical intermediate suppliers, this patent represents a significant leap forward in process chemistry, offering a pathway to high-purity materials with a substantially reduced environmental footprint.
The strategic importance of 4-aminoindan derivatives cannot be overstated, as they form the core structure of various pyrazoleindanylcarboxamides and pyridindanylcarboxamides known for their exceptional fungicidal activity. The ability to synthesize these intermediates with diverse substitution patterns allows for the fine-tuning of biological properties, making this technology invaluable for the development of next-generation crop protection solutions. By leveraging the unique reactivity of anhydrous HF, the process described in CN110799491A bypasses the limitations of prior art, such as the restricted substitution patterns achievable through acetone condensation methods. This flexibility ensures that manufacturers can access a broader library of analogues, facilitating rapid optimization of agrochemical candidates while maintaining rigorous quality standards required for commercial scale-up.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of 4-aminoindan derivatives has been plagued by significant inefficiencies and environmental concerns associated with conventional acid-catalyzed rearrangements. Prior art methods, such as those disclosed in EP 0654464 and WO 2014/103811, typically employ concentrated sulfuric acid (H2SO4) to effect the necessary ring transformation. While chemically feasible, these processes generate enormous quantities of liquid waste, specifically aqueous ammonium sulfate solutions, resulting from the mandatory neutralization step required to isolate the basic aminoindan product. For every gram of desired product, legacy methods can produce upwards of 13 grams of hazardous liquid waste, creating a substantial burden on waste treatment facilities and driving up operational costs. Furthermore, the exothermic nature of mixing organic substrates with concentrated sulfuric acid necessitates strict temperature control and specialized equipment to manage safety risks.
Alternative approaches utilizing transition metal catalysis, such as the Buchwald-Hartwig amination described in WO 2013/167545, present a different set of challenges that hinder widespread industrial adoption. These methods require the use of expensive palladium catalysts and halogenated indane precursors, which are often difficult and costly to synthesize in large quantities. Additionally, the presence of heavy metals in the final product necessitates rigorous purification protocols to meet stringent regulatory limits for agrochemical residues, adding further complexity and expense to the manufacturing process. The reliance on protected ammonia derivatives rather than free ammonia also limits the atom economy of the reaction. Consequently, there is a critical need for a simplified, economically viable method that eliminates transition metals and minimizes waste generation without compromising yield or purity.
The Novel Approach
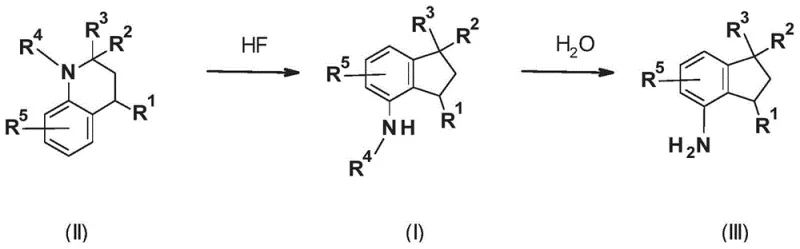
The innovative process detailed in CN110799491A offers a transformative solution by utilizing anhydrous hydrogen fluoride (HF) as both the solvent and the rearrangement catalyst. This method enables the direct conversion of 1-acyl-trialkyl-3,4-dihydro-1H-quinolines (Formula II) into the corresponding 4-aminoindan derivatives (Formula I) with exceptional efficiency. The use of anhydrous HF is particularly advantageous because it acts as a weak acid compared to sulfuric acid, allowing the reaction to proceed under very mild thermal conditions, typically ranging from -10°C to 80°C, with a preferred window of 0°C to 30°C. This gentle environment preserves sensitive functional groups and prevents the formation of degradation byproducts, resulting in a cleaner reaction profile. Moreover, the physical properties of HF allow for its easy removal from the reaction mixture via simple distillation at atmospheric pressure, facilitating its recovery and reuse in subsequent batches.
This novel approach fundamentally alters the economic and environmental calculus of 4-aminoindan production. By eliminating the need for neutralization with ammonia or sodium hydroxide, the process avoids the generation of massive salt waste streams, aligning perfectly with modern green chemistry principles. The ability to recycle the HF reagent significantly reduces raw material consumption, offering a compelling cost reduction in agrochemical intermediate manufacturing. Furthermore, the high selectivity of the HF-mediated rearrangement means that the crude product often requires minimal purification, potentially bypassing energy-intensive steps like column chromatography. For supply chain leaders, this translates to a more robust and scalable process capable of delivering high-purity intermediates consistently, thereby securing the supply chain for downstream active ingredient production.
Mechanistic Insights into HF-Catalyzed Rearrangement
The core of this technology lies in the unique ability of anhydrous HF to promote the skeletal rearrangement of the tetrahydroquinoline ring system into the indan framework. Mechanistically, the reaction likely proceeds through protonation of the nitrogen atom or the carbonyl oxygen of the acyl group, increasing the electrophilicity of the adjacent carbon centers. This activation facilitates a concerted or stepwise migration of the alkyl substituents and the closure of the five-membered indan ring, driven by the relief of steric strain and the formation of a stable aromatic system. The mild acidity of HF compared to sulfuric acid is crucial here; it provides sufficient catalytic activity to drive the rearrangement without inducing excessive polymerization or charring of the organic substrate, which are common pitfalls in strong acid media. This delicate balance ensures that the reaction proceeds with high fidelity, preserving the integrity of the substituents R1 through R5 as defined in the patent claims.
Impurity control is inherently superior in this HF-mediated system due to the homogeneity of the reaction medium and the absence of competing side reactions typical of metal-catalyzed pathways. In transition metal catalysis, issues such as dehalogenation, homocoupling, or incomplete amination can lead to complex impurity profiles that are difficult to separate. In contrast, the acid-catalyzed rearrangement described here generates a much simpler byproduct spectrum, primarily consisting of unreacted starting material which can be easily separated or recycled. The subsequent hydrolysis step to convert the acetylated intermediate (Formula I) into the free amine (Formula III) is also highly controlled, occurring cleanly upon the addition of water and heating to 50°C to 100°C. This two-stage process allows for the isolation of either the protected or deprotected amine depending on the specific needs of the downstream synthesis, providing remarkable flexibility for process chemists optimizing multi-step routes.
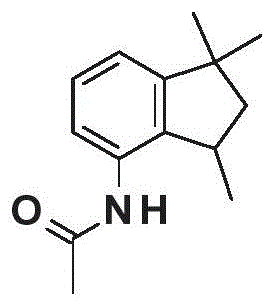
How to Synthesize N-(1,1,3-trimethylindan-4-yl)acetamide Efficiently
Implementing this synthesis route requires careful attention to the handling of anhydrous HF, which necessitates the use of corrosion-resistant equipment such as polytetrafluoroethylene (PTFE) reactors. The process begins by charging the reactor with anhydrous HF and cooling it to approximately 0°C before the slow addition of the tetrahydroquinoline precursor. Maintaining the temperature within the optimal range of 0°C to 30°C during the addition and subsequent stirring period of 10 to 22 hours is critical for maximizing yield and minimizing side products. Once the rearrangement is complete, the excess HF is distilled off and collected for recycling, leaving behind the solid acetylated indan derivative. This solid can then be washed with cold water to remove trace acids and dried to obtain the high-purity intermediate. For the production of the free amine, the intermediate is subjected to hydrolysis in water at elevated temperatures, followed by pH adjustment and extraction.
- Mix the tetrahydroquinoline precursor (Formula II) with anhydrous hydrogen fluoride (HF) in a PTFE reactor, maintaining temperatures between 0°C and 30°C.
- Stir the reaction mixture for 10 to 22 hours to ensure complete rearrangement into the protected 4-aminoindan intermediate (Formula I).
- Remove excess HF via distillation for recycling, then hydrolyze the acetyl group with water at elevated temperatures to yield the free amine (Formula III).
Commercial Advantages for Procurement and Supply Chain Teams
From a commercial perspective, the adoption of this HF-mediated rearrangement technology offers profound advantages for procurement managers and supply chain heads focused on cost efficiency and sustainability. The most significant benefit is the drastic simplification of the workup procedure, which eliminates the need for large-scale neutralization steps that generate tons of saline wastewater. This reduction in waste volume directly translates to lower disposal costs and reduced regulatory compliance burdens, making the process economically superior to traditional sulfuric acid methods. Furthermore, the recyclability of the anhydrous HF solvent creates a closed-loop system that minimizes raw material expenditure. By recovering and reusing the HF, manufacturers can achieve substantial cost savings in agrochemical intermediate manufacturing, insulating themselves from volatility in raw material pricing and ensuring long-term process viability.
- Cost Reduction in Manufacturing: The elimination of expensive transition metal catalysts such as palladium removes a major cost driver from the bill of materials. Additionally, the avoidance of complex purification steps like column chromatography reduces solvent consumption and labor hours. The high yields reported in the patent examples, reaching up to 97% for the intermediate and 90% for the free amine, ensure that raw material utilization is maximized, further driving down the cost per kilogram of the final product. This economic efficiency makes the technology highly attractive for large-scale production where margin optimization is critical.
- Enhanced Supply Chain Reliability: The robustness of this chemical process enhances supply chain reliability by reducing the risk of batch failures associated with sensitive catalytic systems. The reagents involved, including anhydrous HF and the quinoline precursors, are readily available from established chemical suppliers, ensuring a stable supply of inputs. The simplicity of the operation, which can be conducted at atmospheric pressure and moderate temperatures, allows for execution in standard chemical manufacturing facilities without the need for specialized high-pressure autoclaves. This accessibility facilitates faster technology transfer and scale-up, reducing lead time for high-purity agrochemical intermediates and ensuring consistent delivery to downstream customers.
- Scalability and Environmental Compliance: Scalability is inherently supported by the continuous or semi-continuous nature of the distillation and hydrolysis steps. The process generates significantly less hazardous waste compared to prior art, aligning with increasingly stringent global environmental regulations. The ability to operate without generating ammonium sulfate waste streams simplifies effluent treatment and reduces the environmental footprint of the manufacturing site. This commitment to green chemistry not only mitigates regulatory risk but also enhances the brand reputation of the manufacturer as a sustainable partner in the agrochemical value chain, appealing to end-users who prioritize environmentally responsible sourcing.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this HF-mediated synthesis route. These answers are derived directly from the experimental data and process descriptions provided in patent CN110799491A, ensuring accuracy and relevance for technical decision-makers. Understanding these details is essential for evaluating the feasibility of integrating this technology into existing manufacturing workflows.
Q: What are the primary advantages of using anhydrous HF over sulfuric acid for this rearrangement?
A: Unlike sulfuric acid methods which generate massive amounts of aqueous ammonium sulfate waste requiring neutralization, anhydrous HF can be easily removed by distillation and recycled, drastically reducing liquid waste and environmental impact.
Q: Does this process require expensive transition metal catalysts?
A: No, this method relies on acid-catalyzed rearrangement using HF, eliminating the need for costly palladium catalysts and avoiding the complex purification steps required to remove trace heavy metals from the final product.
Q: What is the typical purity and yield achievable with this HF-mediated route?
A: Experimental data from the patent indicates high efficiency, with yields reaching up to 97% for the acetylated intermediate and 90% for the free amine, achieving high purity without the need for column chromatography.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 4-Aminoindan Derivatives Supplier
At NINGBO INNO PHARMCHEM, we recognize the transformative potential of the HF-mediated rearrangement technology for producing high-value agrochemical intermediates. As a leading CDMO partner, we possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that your transition from laboratory discovery to industrial reality is seamless. Our state-of-the-art facilities are equipped to handle corrosive reagents like anhydrous HF safely and efficiently, adhering to the highest safety and quality standards. We are committed to delivering products with stringent purity specifications, supported by our rigorous QC labs that utilize advanced analytical techniques to verify identity and potency. Our team of expert chemists is ready to optimize this specific route to meet your unique volume and quality requirements.
We invite you to collaborate with us to leverage this advanced synthesis method for your next-generation crop protection programs. By partnering with NINGBO INNO PHARMCHEM, you gain access to a Customized Cost-Saving Analysis that quantifies the economic benefits of switching to this waste-reduced process. We encourage you to contact our technical procurement team today to request specific COA data and route feasibility assessments tailored to your target molecules. Let us help you secure a sustainable and cost-effective supply of critical 4-aminoindan building blocks, empowering your R&D efforts with reliable, high-quality materials.
Engineering Bottleneck?
Can't scale up this synthesis? Upload your target structure or CAS, and our CDMO team will evaluate the industrial feasibility within 24 hours. Request Evaluation →
