Advanced Manufacturing of Halogen-Substituted Phenylenedimethanols for Global Supply Chains
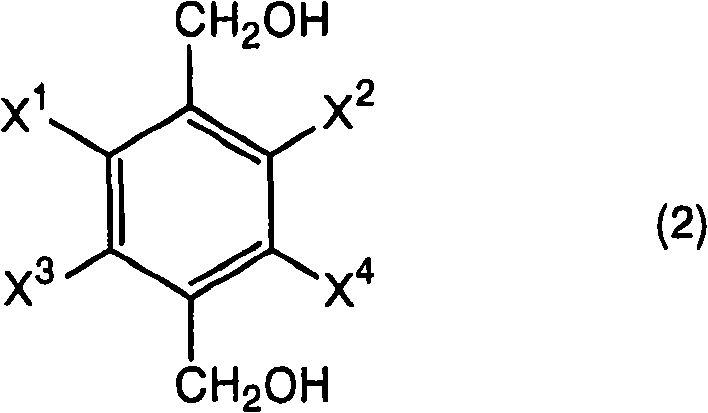
The chemical landscape for producing high-value intermediates is constantly evolving, driven by the need for higher purity and more sustainable manufacturing processes. Patent CN101356146B represents a significant leap forward in the synthesis of halogen-substituted phenylenedimethanols, which are critical building blocks for next-generation agrochemicals and pharmaceutical agents. This technology addresses a long-standing challenge in organic synthesis: the efficient reduction of halogenated aromatic diesters without compromising the integrity of the sensitive halogen substituents. By introducing a precisely controlled amount of water into a borohydride reduction system, the inventors have unlocked a pathway that delivers superior yields and simplified downstream processing. For global procurement teams and R&D directors, this patent offers a blueprint for cost reduction in agrochemical intermediate manufacturing while ensuring the consistent quality required for regulatory compliance. The ability to produce compounds like 2,3,5,6-tetrafluorobenzenedimethanol with an 86% yield marks a distinct advantage over legacy methods, positioning this technology as a cornerstone for reliable supply chain strategies in the fine chemicals sector.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
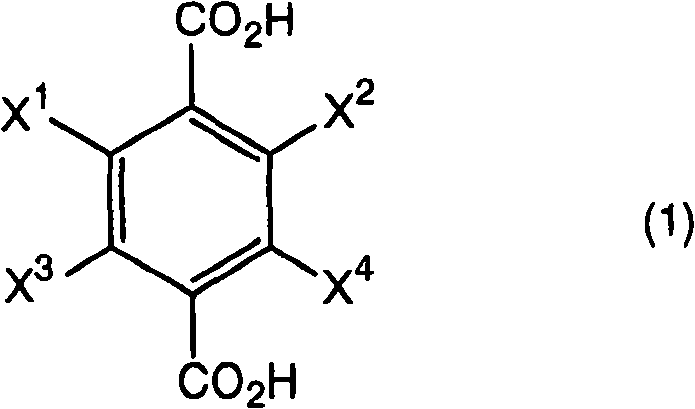
Traditional synthetic routes for preparing halogen-substituted benzenedimethanols often rely on standard reduction protocols that fail to account for the unique electronic effects of multiple halogen atoms on the aromatic ring. In conventional practices, such as those referenced in prior art like CN1458137A, the reduction of diesters using borohydride compounds is typically conducted under strictly anhydrous conditions to prevent the premature decomposition of the reducing agent. However, this approach frequently results in incomplete conversion and the formation of difficult-to-remove byproducts, leading to suboptimal yields that hover around 57% as demonstrated in comparative studies. The inefficiency of these older methods necessitates larger reactor volumes and increased consumption of raw materials to achieve the same output, thereby inflating the cost of goods sold (COGS). Furthermore, the rigorous requirement for absolute dryness adds complexity to the operational workflow, requiring extensive drying of solvents and reagents, which increases energy consumption and extends batch cycle times. For supply chain managers, these inefficiencies translate into longer lead times and reduced flexibility when responding to market demand fluctuations for high-purity pharmaceutical intermediates.
The Novel Approach
The methodology disclosed in CN101356146B fundamentally disrupts the conventional wisdom regarding borohydride reductions by intentionally introducing water into the reaction matrix. This novel approach utilizes a specific molar ratio of 0.5 to 10 moles of water per mole of boron hydride compound, which surprisingly enhances the reduction efficiency rather than inhibiting it. By carefully modulating the water content, the reaction kinetics are optimized to favor the complete reduction of the ester groups to hydroxymethyl groups while preserving the halogen substituents. This strategic modification allows the process to achieve yields as high as 86%, representing a dramatic improvement over the 57% benchmark of traditional anhydrous methods. The robustness of this new protocol means that manufacturers can utilize commercially available solvents and reagents without the need for extreme drying procedures, significantly simplifying the operational overhead. This shift not only improves the economic viability of producing complex halogenated intermediates but also enhances the commercial scale-up of complex polymer additives and agrochemical precursors by providing a more forgiving and scalable reaction window.
Mechanistic Insights into Controlled Hydration Borohydride Reduction
The mechanistic underpinning of this invention lies in the delicate balance between the reducing power of the borohydride species and the hydrolytic activity introduced by the controlled water addition. In a standard reduction, the borohydride anion attacks the carbonyl carbon of the ester to form a tetrahedral intermediate, which subsequently collapses to release an alkoxide. However, in the presence of multiple electron-withdrawing halogen atoms, the reactivity of the aromatic ring can be altered, sometimes leading to side reactions or incomplete reduction. The introduction of water in the specific range of 0.9 to 4 moles per mole of borohydride appears to facilitate the hydrolysis of intermediate borate esters, driving the equilibrium towards the formation of the free alcohol product. This hydration effect likely prevents the accumulation of stable borate complexes that might otherwise trap the product or require harsh acidic workups to liberate the diol. Consequently, the reaction proceeds with greater fidelity, minimizing the formation of mono-alcohol impurities or dehalogenated byproducts that often plague the synthesis of poly-halogenated aromatics.
From an impurity control perspective, this mechanism offers a cleaner profile that is highly attractive for reducing lead time for high-purity pharmaceutical intermediates. The suppression of side reactions means that the crude reaction mixture contains fewer structurally related impurities, simplifying the purification process. Instead of requiring multiple recrystallizations or complex chromatographic separations, the product can often be isolated through standard aqueous workup and extraction techniques. The patent specifies that after the reaction, the mixture can be quenched with aqueous mineral acids like hydrochloric acid, followed by extraction with water-insoluble solvents such as ethyl acetate or toluene. This streamlined isolation process not only reduces solvent waste but also ensures that the final high-purity OLED material or agrochemical precursor meets stringent specifications with minimal additional processing. The ability to maintain the integrity of sensitive halogen bonds, particularly fluorine and chlorine, during such a vigorous reduction highlights the precision of this catalytic system.
How to Synthesize Halogen-Substituted Phenylenedimethanols Efficiently
Implementing this synthesis route requires careful attention to the stoichiometry of water and the temperature profile to maximize the benefits observed in the patent examples. The process begins with the preparation of a slurry containing the halogen-substituted terephthalic acid diester and the borohydride reducing agent in an inert organic solvent such as tetrahydrofuran. It is critical to maintain the reaction temperature within the optimal range of 40°C to 100°C, with 65°C being particularly effective for substrates like 2,3,5,6-tetrafluoroterephthalic acid dimethyl ester. The key operational step involves the slow, dropwise addition of the water-solvent mixture over a period of several hours, typically around 3 to 5 hours, to ensure that the local concentration of water does not exceed the threshold that would cause rapid gas evolution or reagent destruction. Following the addition, the reaction is allowed to stir for an additional period to ensure complete conversion before being quenched with dilute acid. The detailed standardized synthesis steps see the guide below.
- Prepare the reaction mixture by combining the halogen-substituted terephthalic acid diester, sodium borohydride, and an ether solvent such as tetrahydrofuran in a reactor.
- Heat the mixture to a reaction temperature between 40°C and 100°C, preferably around 65°C, to initiate the reduction environment.
- Slowly add a mixture of water and solvent over several hours, maintaining a molar ratio of 0.5 to 10 moles of water per mole of borohydride, then quench with acid and extract the product.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the adoption of this patented process translates into tangible strategic advantages that extend beyond simple yield metrics. The primary benefit is the substantial optimization of raw material utilization, as the improved conversion rates mean that less starting material is required to produce the same quantity of finished goods. This efficiency directly impacts the bottom line by lowering the variable costs associated with production, making the final product more competitive in the global marketplace. Additionally, the elimination of the need for rigorously anhydrous conditions reduces the energy burden on the manufacturing facility, as extensive solvent drying systems are no longer a prerequisite for successful batch execution. This relaxation of process constraints allows for faster batch turnover and increased throughput capacity, enabling suppliers to respond more agilely to sudden spikes in demand from downstream formulators.
- Cost Reduction in Manufacturing: The economic impact of this technology is driven by the dual factors of increased yield and simplified reagent requirements. By achieving yields of 86% compared to the historical baseline of roughly 57%, the process effectively reduces the cost per kilogram of the active intermediate by nearly half in terms of raw material consumption. Furthermore, the use of sodium borohydride, a commodity chemical, instead of specialized or expensive reducing agents ensures that the input costs remain stable and predictable. The reduction in waste generation also lowers the costs associated with waste disposal and environmental compliance, contributing to a leaner and more sustainable manufacturing model that aligns with modern corporate responsibility goals.
- Enhanced Supply Chain Reliability: The robustness of the reaction conditions significantly de-risks the supply chain by minimizing the potential for batch failures due to moisture sensitivity. In traditional anhydrous processes, even minor leaks or humidity fluctuations can compromise an entire batch, leading to delays and shortages. The tolerance for water in this new method creates a more resilient production environment where consistent quality can be maintained across different manufacturing sites and equipment setups. This reliability is crucial for long-term contracts with major agrochemical and pharmaceutical companies that require guaranteed continuity of supply for their critical production lines, ensuring that downstream manufacturing schedules are never disrupted by upstream intermediate shortages.
- Scalability and Environmental Compliance: Scaling this process from laboratory to industrial production is straightforward due to the use of common solvents and the absence of hazardous catalysts. The workup procedure, which involves simple acid quenching and phase separation, is easily adaptable to large-scale reactors without the need for complex filtration or distillation trains. From an environmental perspective, the process generates fewer organic byproducts and utilizes less energy for solvent conditioning, resulting in a lower overall carbon footprint. This alignment with green chemistry principles facilitates easier regulatory approval in jurisdictions with strict environmental standards, allowing for smoother market entry and expanded geographic distribution of the manufactured intermediates.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this synthesis technology. These answers are derived directly from the experimental data and claims presented in patent CN101356146B, providing a factual basis for evaluating the feasibility of adopting this route. Understanding these details is essential for technical teams assessing the compatibility of this process with existing infrastructure and for commercial teams negotiating supply agreements. The clarity provided here aims to remove ambiguity regarding the scope and limitations of the patented method.
Q: What is the primary technical breakthrough in patent CN101356146B?
A: The core innovation lies in the controlled addition of water (0.5 to 10 moles per mole of borohydride) during the reduction of halogenated terephthalic acid diesters. Traditionally, water is avoided in borohydride reductions to prevent reagent decomposition, but this patent demonstrates that specific stoichiometric amounts significantly enhance yield, improving it from roughly 57% in conventional methods to 86%.
Q: Which specific halogenated substrates are compatible with this process?
A: The process is highly versatile and supports a wide range of halogen substitutions including fluorine, chlorine, and bromine. Specific examples validated in the patent include 2,3,5,6-tetrafluoroterephthalic acid dimethyl ester and various chloro-substituted analogs, making it suitable for synthesizing diverse intermediates for both pesticide and pharmaceutical applications.
Q: How does this method impact production costs compared to traditional routes?
A: By achieving yields as high as 86% compared to 57% in comparative examples without water control, the process drastically reduces raw material waste. Furthermore, the use of commercially available sodium borohydride and simple ether solvents like THF eliminates the need for expensive or exotic catalysts, leading to substantial operational cost savings.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Halogen-Substituted Phenylenedimethanols Supplier
At NINGBO INNO PHARMCHEM, we recognize that the transition from patent literature to commercial reality requires a partner with deep technical expertise and proven manufacturing capabilities. Our team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the promising yields demonstrated in the lab can be consistently replicated on an industrial scale. We are committed to maintaining stringent purity specifications through our rigorous QC labs, utilizing advanced analytical techniques to verify that every batch of halogen-substituted phenylenedimethanols meets the exacting standards required for pharmaceutical and agrochemical applications. Our infrastructure is designed to handle the specific nuances of halogenated chemistry, providing a secure and compliant environment for the manufacture of these high-value intermediates.
We invite you to collaborate with us to leverage this advanced synthesis technology for your specific product needs. By engaging with our technical procurement team, you can request a Customized Cost-Saving Analysis that quantifies the potential economic benefits of switching to this high-yield route for your supply chain. We encourage you to reach out today to obtain specific COA data and route feasibility assessments tailored to your project requirements, ensuring that you have all the necessary information to make informed sourcing decisions that drive value and efficiency for your organization.