Advanced Manufacturing of Halogen-Substituted Phenylenedimethanols for Global Pharma and Agrochemical Supply Chains
The global demand for high-performance intermediates in the pharmaceutical and agrochemical sectors continues to drive innovation in synthetic methodologies. Patent CN101001823A represents a significant technological advancement in the manufacture of halogen-substituted phenylenedimethanols, which serve as critical building blocks for various bioactive compounds. This patent discloses a robust process for producing compounds represented by formula (2) through the reduction of halogen-substituted terephthalic acid diesters, denoted as formula (1). Unlike traditional methods that often rely on hazardous reagents or complex multi-step sequences, this invention utilizes a borohydride compound in the presence of an alcohol under controlled conditions. The core innovation lies in the specific order of addition, where the alcohol is added to a pre-mixed suspension of the diester, the borohydride, and a solvent. This strategic modification not only enhances reaction safety by managing exotherms but also significantly improves the yield and purity profile of the final product, addressing key pain points for R&D directors seeking reliable synthetic routes for complex fluorinated or chlorinated intermediates.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Prior art methods for synthesizing halogen-substituted benzenedimethanols have historically suffered from significant operational and economic drawbacks. For instance, earlier techniques described in patents such as US4927852 involved the bromination of tetrafluoro-p-xylene followed by acetoxylation and hydrolysis, a sequence that introduces multiple unit operations and generates substantial halogenated waste streams. Other approaches, like those found in GB2127013A, utilized 2,3,5,6-tetrafluoroterephthaloyl chloride, a highly moisture-sensitive and corrosive intermediate that poses severe handling challenges and requires stringent anhydrous conditions throughout the process. Furthermore, methods employing borane-tetrahydrofuran complexes, as seen in US5583131, involve expensive reagents and potential safety risks associated with borane stability. These conventional pathways often result in lower overall yields due to cumulative losses across multiple steps and complicate the supply chain with the need for specialized storage and transport of unstable intermediates, thereby increasing the total cost of ownership for manufacturers.
The Novel Approach
The process disclosed in CN101001823A offers a streamlined alternative by directly reducing stable halogen-substituted terephthalic acid diesters. By reacting the diester with a borohydride compound, preferably an alkali metal borohydride like sodium borohydride, in a solvent system, the method eliminates the need for hazardous acyl chlorides or expensive borane complexes. The critical procedural improvement involves adding the alcohol, such as methanol, to the mixture of the diester, borohydride, and solvent rather than the reverse. This specific addition protocol moderates the reaction rate, preventing runaway exotherms that could lead to dehalogenation or other degradation pathways. As illustrated in the reaction scheme below, this approach converts the diester functionality directly to the dimethanol with high efficiency.
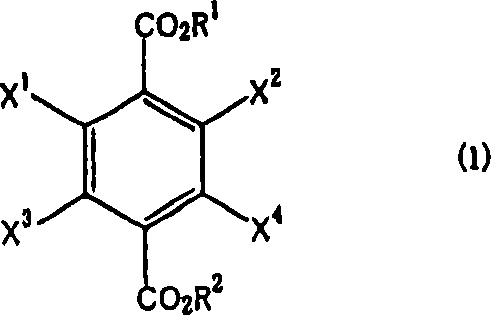
Experimental data from the patent demonstrates the efficacy of this approach, with Example 2 achieving an 87% yield and 95.5% purity for 2,3,5,6-tetrafluorobenzenedimethanol, while Example 3 showed a 90% yield for the tetrachloro analog with purity reaching 99.6%. This direct reduction strategy simplifies the workflow, reduces raw material costs, and enhances the safety profile of the manufacturing process, making it an attractive option for cost reduction in fine chemical intermediates manufacturing.
Mechanistic Insights into Borohydride-Mediated Ester Reduction
The mechanistic pathway of this transformation relies on the nucleophilic attack of the borohydride species on the carbonyl carbon of the ester group. In the presence of an alcohol, the borohydride generates alkoxyborohydride species which are active reducing agents. The patent highlights that the order of addition is paramount; by introducing the alcohol to the solid-liquid mixture of ester and borohydride, the concentration of the active reducing species is generated in situ at a controlled rate. This prevents the localized high concentrations of heat and reactive intermediates that typically occur when mixing bulk reagents rapidly. For R&D professionals, understanding this kinetic control is essential for scaling up the reaction, as it ensures that the reduction proceeds selectively at the ester moiety without affecting the sensitive halogen substituents on the aromatic ring, which are prone to nucleophilic substitution or reduction under harsher conditions.
Impurity control is another critical aspect addressed by this mechanism. Traditional reduction methods using strong hydride sources can sometimes lead to over-reduction or cleavage of carbon-halogen bonds, particularly with activated aryl halides. The moderated reactivity in this patented process minimizes such side reactions. The use of ether solvents like tetrahydrofuran or methyl tert-butyl ether provides a stable medium that solubilizes the intermediates while maintaining the stability of the borohydride. Post-reaction workup involving acid quenching followed by neutralization allows for the efficient separation of boron salts, facilitating the isolation of the target phenylenedimethanol through simple extraction and crystallization. This results in a product with a clean impurity profile, reducing the burden on downstream purification processes and ensuring the material meets the stringent specifications required for high-purity pharmaceutical intermediates.
How to Synthesize Halogen-Substituted Phenylenedimethanols Efficiently
To implement this synthesis effectively, operators must adhere to strict parameter controls regarding temperature and addition rates. The process begins by charging a reactor with the halogen-substituted terephthalic acid diester, the borohydride compound, and the chosen organic solvent. The mixture is stirred to form a homogeneous suspension before the controlled addition of the alcohol commences. Maintaining the internal temperature, typically around 55°C during addition, is crucial for optimizing reaction kinetics without compromising safety. Following the addition, the reaction is allowed to stir for a prolonged period to ensure complete conversion, after which careful quenching and workup procedures are executed to isolate the product. The detailed standardized synthesis steps see the guide below.
- Prepare a mixture of halogen-substituted terephthalic acid diester, a borohydride compound such as sodium borohydride, and an organic solvent like tetrahydrofuran.
- Controlledly add an alcohol, preferably methanol, to the reaction mixture at a maintained internal temperature, typically around 55°C, to initiate reduction.
- Upon completion, quench the reaction with aqueous acid, neutralize, extract with organic solvents, and purify the resulting crystals via recrystallization.
Commercial Advantages for Procurement and Supply Chain Teams
From a procurement and supply chain perspective, the adoption of this manufacturing process offers substantial strategic benefits. The shift from using acyl chlorides or borane complexes to stable diesters and sodium borohydride drastically simplifies the raw material sourcing landscape. Sodium borohydride is a commodity chemical with a robust global supply chain, ensuring consistent availability and price stability compared to specialized reagents. Furthermore, the elimination of corrosive acyl chlorides reduces the requirements for specialized corrosion-resistant equipment and lowers maintenance costs associated with reactor degradation. This transition supports significant cost reduction in manufacturing by streamlining the bill of materials and reducing the complexity of waste treatment protocols, as the process generates less hazardous byproducts.
- Cost Reduction in Manufacturing: The utilization of sodium borohydride instead of expensive borane-tetrahydrofuran complexes or multi-step bromination sequences leads to a direct decrease in raw material expenditures. Additionally, the simplified one-pot reduction process reduces energy consumption and labor hours associated with intermediate isolations and transfers. By avoiding the need for cryogenic conditions or high-pressure hydrogenation equipment often required in alternative routes, capital expenditure for plant setup is also minimized, allowing for more flexible and economical production scheduling.
- Enhanced Supply Chain Reliability: The starting materials, specifically halogen-substituted terephthalic acid diesters, are chemically stable and can be stored for extended periods without degradation, unlike moisture-sensitive acyl chlorides. This stability enhances inventory management capabilities and reduces the risk of supply disruptions due to material spoilage. The reliance on common solvents like methanol and THF further secures the supply chain, as these are widely produced and less susceptible to logistical bottlenecks compared to niche reagents, ensuring reducing lead time for high-purity intermediates.
- Scalability and Environmental Compliance: The process operates under atmospheric pressure and moderate temperatures, making it inherently safer and easier to scale from pilot plants to commercial production volumes. The waste stream primarily consists of boron salts and organic solvents, which are well-understood and manageable within standard wastewater treatment frameworks. This aligns with increasingly stringent environmental regulations, reducing the compliance burden and potential fines associated with hazardous waste disposal, thereby supporting sustainable commercial scale-up of complex polymer additives and pharmaceutical precursors.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this patented technology. These insights are derived directly from the experimental data and claims within CN101001823A, providing clarity on process feasibility and product quality. Understanding these details is vital for stakeholders evaluating the integration of this route into their existing manufacturing portfolios.
Q: What are the primary advantages of using diesters over acyl chlorides in this synthesis?
A: Using halogen-substituted terephthalic acid diesters avoids the handling hazards and moisture sensitivity associated with acyl chlorides, leading to a safer and more robust manufacturing process with easier waste treatment.
Q: How does the controlled addition of alcohol impact product purity?
A: Adding the alcohol to the mixture of ester and borohydride allows for precise control over the reaction exotherm and the generation of active reducing species, minimizing side reactions like dehalogenation and ensuring high chemical purity.
Q: Is this process scalable for industrial production of agrochemical intermediates?
A: Yes, the process utilizes common solvents like THF and methanol and standard reagents like sodium borohydride, making it highly suitable for commercial scale-up from kilogram to multi-ton quantities without requiring specialized high-pressure equipment.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Halogen-Substituted Phenylenedimethanols Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical role that high-quality intermediates play in the development of next-generation pharmaceuticals and agrochemicals. Our technical team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the innovative processes described in patents like CN101001823A can be translated into reliable industrial reality. We maintain stringent purity specifications and operate rigorous QC labs to guarantee that every batch of halogen-substituted phenylenedimethanols meets the exacting standards required by global regulatory bodies. Our commitment to quality assurance ensures that our clients receive materials with consistent impurity profiles, facilitating smoother downstream synthesis and faster time-to-market for final products.
We invite potential partners to engage with our technical procurement team to discuss how this advanced synthesis route can optimize your supply chain. By requesting a Customized Cost-Saving Analysis, you can gain a deeper understanding of the economic benefits specific to your volume requirements. We encourage you to contact us to obtain specific COA data and route feasibility assessments tailored to your project needs, ensuring a seamless transition from laboratory concept to commercial success with a trusted partner dedicated to excellence in fine chemical manufacturing.
