Scalable Production of High-Purity Cyclopropyl Formaldehyde via Optimized Corey-Chaykovsky Reaction
The pharmaceutical industry continuously seeks robust and environmentally sustainable pathways for constructing strained ring systems, particularly the cyclopropane moiety, which is a privileged structure in numerous bioactive molecules. A significant technological breakthrough in this domain is detailed in Chinese patent CN115636740A, which discloses a novel and highly efficient synthesis process for cyclopropyl formaldehyde. This critical building block serves as a versatile pharmaceutical intermediate, enabling the introduction of cyclopropyl groups into complex drug candidates through subsequent functionalization. The disclosed methodology leverages the classic yet powerful Corey-Chaykovsky reaction, utilizing acrolein and a trimethyl sulfoxide sulfonium salt to construct the three-membered ring with exceptional precision. By shifting away from traditional oxidative routes that rely on hazardous heavy metals, this innovation offers a compelling value proposition for reliable pharmaceutical intermediate suppliers aiming to enhance their green chemistry credentials while maintaining rigorous quality standards.
The strategic importance of this patent lies not only in its chemical elegance but also in its direct address of long-standing supply chain vulnerabilities associated with older synthetic technologies. For R&D directors and procurement specialists alike, the ability to source cyclopropyl formaldehyde produced via this metal-free pathway represents a significant de-risking opportunity. The process ensures a consistent supply of high-purity material without the regulatory baggage of residual chromium or other transition metals, thereby streamlining the drug substance registration process. As we delve deeper into the technical specifics, it becomes evident that this methodology is not merely a laboratory curiosity but a fully scalable industrial solution capable of supporting the demanding timelines of modern drug development pipelines.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of cyclopropyl formaldehyde has often relied on the oxidation of cyclopropyl methanol, a route fraught with significant chemical and operational challenges that hinder efficient cost reduction in pharmaceutical intermediate manufacturing. Traditional oxidation protocols frequently employ stoichiometric amounts of toxic oxidants such as sodium dichromate or catalytic systems involving TEMPO coupled with sodium hypochlorite. These reagents introduce severe complications, including the generation of hazardous heavy metal waste streams that require expensive and energy-intensive remediation processes before disposal. Furthermore, the presence of transition metal residues in the final product necessitates additional purification steps, such as treatment with scavenger resins or complex extraction sequences, to meet the stringent ICH Q3D guidelines for elemental impurities. These extra processing stages not only extend the overall production lead time but also inevitably erode the overall process yield, leading to higher raw material consumption and increased manufacturing costs.
Beyond the environmental and regulatory burdens, conventional oxidative methods often suffer from issues related to selectivity and safety. The use of strong oxidants on sensitive substrates can lead to over-oxidation, resulting in the formation of carboxylic acid by-products that are difficult to separate from the desired aldehyde. Additionally, the handling of large quantities of hypochlorite or chromate salts poses significant safety risks in a plant environment, requiring specialized containment and handling procedures that further inflate capital and operational expenditures. For supply chain managers, these complexities translate into fragile production schedules where minor deviations in reagent quality or reaction conditions can result in batch failures, jeopardizing the continuity of supply for downstream API synthesis. Consequently, there is a pressing industry need for a cleaner, safer, and more atom-economical alternative that bypasses these inherent limitations of oxidation chemistry.
The Novel Approach
The innovative process outlined in patent CN115636740A fundamentally reimagines the construction of the cyclopropane ring by employing a constructive rather than destructive strategy. Instead of oxidizing a pre-formed ring, this method builds the cyclopropane motif directly from acrolein through a cyclopropanation reaction with an oxysulfur ylide generated in situ. This Corey-Chaykovsky transformation proceeds under remarkably mild conditions, typically ranging from minus 10 to 80 degrees Celsius, which significantly reduces the thermal stress on the reactants and minimizes the formation of thermal degradation by-products. The avoidance of harsh oxidizing agents means that the reaction mixture remains chemically benign, allowing for a much simpler workup procedure that involves merely adjusting the pH to weak acidity and performing a standard phase separation. This simplicity is a game-changer for commercial scale-up of complex pharmaceutical intermediates, as it allows manufacturers to utilize standard glass-lined or stainless steel reactors without the need for exotic corrosion-resistant materials.
Moreover, the new approach delivers superior performance metrics that directly impact the bottom line. The patent data indicates that this route consistently achieves yields exceeding 90 percent, with product purity surpassing 99 percent after a straightforward atmospheric distillation. This high level of efficiency means that less raw material is wasted, and the throughput per batch is maximized, effectively lowering the cost of goods sold. The elimination of heavy metal catalysts also removes the bottleneck of metal scavenging, drastically shortening the cycle time from reaction completion to finished product release. For procurement teams, this translates into a more predictable and cost-effective supply source, while R&D teams benefit from a cleaner impurity profile that simplifies analytical method development. The combination of high yield, exceptional purity, and operational simplicity positions this novel synthesis route as the preferred choice for next-generation manufacturing of cyclopropyl formaldehyde.
Mechanistic Insights into Corey-Chaykovsky Cyclopropanation
The core of this technological advancement lies in the precise generation and reactivity of the dimethylsulfoxonium methylide, commonly referred to as the Corey-Chaykovsky reagent. The mechanism initiates with the deprotonation of a trimethyl sulfoxide sulfonium salt by a strong base, such as sodium methoxide, sodium hydride, or potassium hydroxide, to form the reactive sulfur ylide species. This ylide possesses a unique electronic structure where the negative charge on the carbon is stabilized by the adjacent positively charged sulfur atom, rendering it a potent nucleophile capable of attacking electron-deficient olefins. In this specific application, the ylide targets the beta-carbon of acrolein, an alpha,beta-unsaturated aldehyde. The initial nucleophilic attack forms a betaine intermediate, which subsequently undergoes an intramolecular displacement of the dimethyl sulfoxide leaving group to close the three-membered ring. This concerted cyclization step is highly stereoselective and energetically favorable, driving the reaction to completion with minimal formation of open-chain by-products.
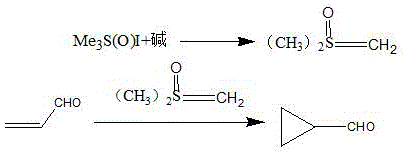
Understanding the nuances of this mechanism is crucial for controlling the impurity profile, a key concern for any high-purity pharmaceutical intermediate supplier. The choice of base and solvent plays a pivotal role in stabilizing the ylide and modulating its reactivity. For instance, the patent highlights the effectiveness of using mixed solvent systems containing dimethyl sulfoxide and tetrahydrofuran or toluene, which optimize the solubility of the ionic ylide species while maintaining a homogeneous reaction medium. Furthermore, the controlled addition of acrolein is critical; adding the aldehyde too rapidly can lead to local excesses that might promote polymerization of the acrolein or side reactions with the ylide. By maintaining the reaction temperature within the specified window of minus 10 to 80 degrees Celsius and carefully managing the addition rate, the process ensures that the cyclopropanation proceeds cleanly. The resulting crude product contains minimal impurities, primarily consisting of unreacted starting materials or dimethyl sulfoxide, both of which are easily removed during the subsequent aqueous workup and distillation steps, ensuring the final product meets the rigorous specifications required for GMP manufacturing.
How to Synthesize Cyclopropyl Formaldehyde Efficiently
Implementing this synthesis route in a production environment requires strict adherence to the optimized parameters defined in the patent to ensure reproducibility and safety. The process is divided into two distinct operational phases: the generation of the active ylide species and the subsequent cyclopropanation followed by isolation. Operators must first ensure that the reaction vessel is properly dried and inerted, as moisture can prematurely quench the ylide or degrade the sulfonium salt. The selection of the base, typically sodium methoxide for its balance of reactivity and cost, must be matched with the appropriate solvent system to facilitate efficient mixing and heat transfer. Once the ylide is formed, the dropwise addition of acrolein must be monitored closely to manage the exotherm and maintain the reaction within the optimal temperature range. Following the reaction period, the quenching step with dilute hydrochloric acid serves a dual purpose: it neutralizes the excess base and protonates any remaining ylide, while also facilitating the partitioning of the organic product into the organic phase for easy separation.
- Generate the oxysulfur ylide by reacting trimethyl sulfoxide sulfonium salt with a strong base such as sodium methoxide in a solvent like THF or toluene.
- Add acrolein dropwise to the ylide solution at controlled temperatures between minus 10 and 80 degrees Celsius to effect the cyclopropanation.
- Quench the reaction with dilute acid, separate the organic phase, dry, and purify via atmospheric distillation to achieve over 99 percent purity.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain directors, the adoption of this patented synthesis route offers substantial strategic advantages that extend far beyond simple unit cost calculations. The primary economic driver is the drastic simplification of the downstream processing train. By eliminating the need for heavy metal catalysts and toxic oxidants, the process removes the requirement for expensive metal scavenger resins, specialized filtration equipment, and complex wastewater treatment protocols associated with chromium or TEMPO waste. This reduction in auxiliary material consumption and waste disposal fees leads to significant operational expenditure savings. Furthermore, the ability to purify the final product via atmospheric distillation, rather than requiring high-vacuum systems, reduces energy consumption and equipment maintenance costs. These efficiencies collectively contribute to a more competitive pricing structure, allowing buyers to secure cost reduction in pharmaceutical intermediate manufacturing without compromising on quality or compliance.
- Cost Reduction in Manufacturing: The economic benefits of this process are rooted in its atom economy and simplified workflow. Unlike oxidative methods that generate stoichiometric amounts of inorganic salt waste, the Corey-Chaykovsky reaction produces dimethyl sulfoxide as a by-product, which is easier to recover and recycle or dispose of safely. The absence of transition metals means that there is no need for costly analytical testing for residual metals at every stage, nor is there a risk of batch rejection due to failed metal limits. Additionally, the high yield of over 90 percent ensures that raw material utilization is maximized, reducing the effective cost per kilogram of the active intermediate. The use of commodity chemicals like acrolein and sodium methoxide further insulates the supply chain from price volatility associated with specialized reagents, providing long-term cost stability.
- Enhanced Supply Chain Reliability: Supply continuity is paramount in the pharmaceutical sector, and this process enhances reliability through the use of robust and readily available starting materials. Acrolein and trimethyl sulfoxide sulfonium salts are produced on a large industrial scale globally, minimizing the risk of raw material shortages that can plague niche reagent-dependent syntheses. The mild reaction conditions also reduce the likelihood of equipment failure or safety incidents that could halt production. Moreover, the process tolerance to a range of solvents, including toluene, THF, and ethyl acetate, provides flexibility for manufacturers to adapt to local solvent availability or pricing fluctuations. This flexibility ensures that production schedules remain uninterrupted, allowing suppliers to meet tight delivery windows and maintain consistent inventory levels for their clients.
- Scalability and Environmental Compliance: As regulatory pressures regarding environmental sustainability intensify, this synthesis route positions manufacturers favorably for future compliance. The process is inherently greener, generating significantly less hazardous waste and avoiding the use of substances of very high concern (SVHC). This aligns with the growing demand from multinational pharma companies for sustainable supply chains and facilitates easier regulatory filings in jurisdictions with strict environmental laws. The scalability of the process is proven by its suitability for atmospheric distillation and standard batch reactors, meaning it can be scaled from pilot plant to multi-ton production without fundamental changes to the chemistry. This ease of scale-up reduces the time and capital required to bring new capacity online, ensuring that supply can grow in tandem with market demand.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the production and application of cyclopropyl formaldehyde synthesized via this novel pathway. These insights are derived directly from the experimental data and beneficial effects reported in the underlying patent documentation, providing a transparent view of the technology's capabilities. Understanding these details helps stakeholders make informed decisions about integrating this intermediate into their own synthesis workflows and supply strategies.
Q: Why is the Corey-Chaykovsky route preferred over oxidation methods for cyclopropyl formaldehyde?
A: Traditional oxidation methods often rely on toxic reagents like sodium dichromate or TEMPO/sodium hypochlorite, which generate hazardous heavy metal waste and require complex removal steps. The patented Corey-Chaykovsky approach utilizes acrolein and sulfonium salts, resulting in a cleaner process with significantly reduced environmental impact and simpler wastewater treatment.
Q: What purity levels can be achieved with this synthesis method?
A: The process described in patent CN115636740A demonstrates the capability to produce cyclopropyl formaldehyde with a content exceeding 99 percent after simple atmospheric distillation. Specific examples in the patent report purities of 99.5 percent and 99.7 percent, meeting the stringent requirements for pharmaceutical intermediate applications.
Q: Is this process suitable for large-scale industrial production?
A: Yes, the synthesis is explicitly designed for industrial scale-up. It operates under mild conditions ranging from minus 10 to 80 degrees Celsius and avoids the need for high-vacuum equipment by utilizing atmospheric distillation. The use of common solvents like toluene and THF further enhances its feasibility for commercial manufacturing.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Cyclopropyl Formaldehyde Supplier
At NINGBO INNO PHARMCHEM, we recognize that the successful commercialization of novel therapeutics depends heavily on the reliability and quality of the supply chain. As a leading CDMO and manufacturer, we have closely evaluated the technology described in patent CN115636740A and possess the technical expertise to implement this advanced Corey-Chaykovsky route at scale. Our facilities are equipped with the necessary infrastructure to handle the specific solvent systems and reaction conditions required for this synthesis, ensuring that we can deliver high-purity cyclopropyl formaldehyde that meets the most exacting specifications. We pride ourselves on our extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, leveraging our rigorous QC labs to guarantee batch-to-batch consistency. Our commitment to stringent purity specifications means that every shipment is accompanied by comprehensive analytical data, giving our partners the confidence to proceed with their drug development programs without delay.
We invite potential partners to engage with our technical team to explore how this optimized synthesis route can benefit your specific project requirements. Whether you are in the early stages of process development or looking to secure a long-term commercial supply, we are prepared to offer a Customized Cost-Saving Analysis tailored to your volume needs. We encourage you to contact our technical procurement team today to request specific COA data, discuss route feasibility assessments, and learn more about how our commitment to innovation and quality can support your supply chain goals. Let us be your trusted partner in bringing high-value pharmaceutical intermediates to the global market efficiently and sustainably.
