Strategic Scale-Up of L-Norvaline: A Robust Chemical Route for Perindopril Intermediates
The pharmaceutical industry constantly seeks robust, scalable pathways for critical amino acid intermediates, particularly those serving as building blocks for major cardiovascular drugs. Patent CN100352801C introduces a significant advancement in the manufacturing of L-norvaline, a pivotal chiral precursor for the synthesis of Perindopril, a widely prescribed ACE inhibitor. Unlike traditional biotechnological approaches that often struggle with low titers and complex purification, this invention details a purely chemical synthesis route starting from n-butyraldehyde and acetone cyanohydrin. The methodology outlines a streamlined sequence involving cyanation, ammonification, amidation, chiral resolution, and final hydrolysis. This shift from biological fermentation to chemical catalysis represents a strategic pivot for manufacturers aiming to secure a more reliable pharmaceutical intermediate supplier network capable of meeting global demand without the volatility associated with biological systems.
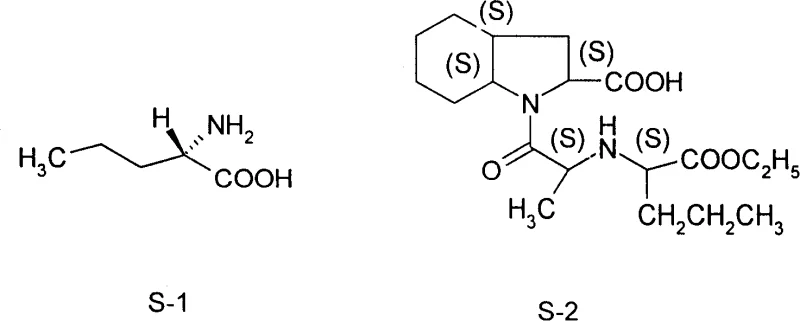
The structural relationship between the intermediate and the final drug is critical for understanding the value proposition of this technology. As illustrated in the provided chemical diagram, L-norvaline (labeled S-1) serves as the foundational chiral scaffold for Perindopril (labeled S-2). The precise stereochemistry at the alpha-carbon is non-negotiable for the biological activity of the final API. The patent addresses the historical bottleneck where fermentation methods yielded merely 3.7g/L, a figure far too low for industrial viability. By establishing a chemical route that leverages commodity feedstocks, this technology enables cost reduction in API manufacturing by decoupling production from the limitations of microbial growth rates and substrate conversion efficiencies inherent in fermentation processes.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the production of optically pure amino acids like L-norvaline has relied heavily on fermentation or enzymatic resolution, both of which present distinct scalability challenges for large-scale commercial scale-up of complex pharmaceutical intermediates. Fermentation processes are notoriously sensitive to environmental variables, requiring sterile conditions, complex nutrient media, and extensive downstream processing to separate the product from biomass and metabolic byproducts. The patent explicitly notes that existing fermentation reports indicate outputs around 3.7g/L, which is economically inefficient for high-volume drug production. Furthermore, biological systems often produce a mixture of isomers or require genetically modified strains that introduce regulatory hurdles. The purification of the target amino acid from a dilute fermentation broth involves energy-intensive concentration steps and generates significant aqueous waste, complicating environmental compliance and driving up operational expenditures.
The Novel Approach
In stark contrast, the novel approach detailed in CN100352801C utilizes a deterministic chemical synthesis pathway that offers superior control over reaction parameters and throughput. By employing n-butyraldehyde and acetone cyanohydrin as starting materials, the process bypasses the biological constraints entirely. The reaction sequence is designed for high atom economy and operational simplicity, utilizing standard chemical reactors rather than specialized bioreactors. The key innovation lies in the efficient construction of the carbon skeleton followed by a highly selective resolution step using L-tartrate. This chemical route allows for reducing lead time for high-purity amino acid intermediates because the reaction times are measured in hours rather than the days required for cell culture growth. Additionally, the use of common organic solvents and inorganic acids simplifies the workup procedures, making the process inherently more adaptable to multi-ton production scales.
Mechanistic Insights into Cyanation and Chiral Resolution
The core of this synthesis lies in the initial cyanation step, where n-butyraldehyde reacts with acetone cyanohydrin under basic catalysis to form 2-hydroxyl valeronitrile. This transformation is critical as it establishes the carbon chain length and introduces the nitrile functionality required for subsequent amination. The patent specifies maintaining a pH between 7 and 9 and a temperature range of 0°C to 15°C, conditions that minimize side reactions such as aldol condensation of the aldehyde. Following this, the ammonification step converts the hydroxyl nitrile into 2-amino valeronitrile using liquid ammonia. This nucleophilic substitution is driven by the high pressure and concentration of ammonia, effectively replacing the hydroxyl group while retaining the nitrile moiety. The subsequent hydrolysis in concentrated sulfuric acid converts the nitrile to an amide, a crucial intermediate that is more stable and easier to handle than the free amino acid during the resolution phase.
Stereochemical control is achieved not through asymmetric catalysis, but through classical diastereomeric salt formation, a proven and robust technique in industrial chemistry. The racemic 2-amino valeramide is reacted with L-tartrate to form the 2-amino valeramide L-tartrate salt. Because the L-enantiomer of the amide forms a less soluble salt with L-tartrate compared to the D-enantiomer, it precipitates out of the solution, allowing for physical separation via filtration. This resolution step is further optimized by recrystallization in a water-methanol mixture, which enhances the optical purity of the intermediate. Finally, the resolved amide is hydrolyzed using a cation exchange resin. This solid-phase hydrolysis technique is particularly advantageous as it simplifies the isolation of the final zwitterionic amino acid, avoiding the formation of inorganic salts that would occur with traditional acid or base hydrolysis, thereby facilitating the production of high-purity pharmaceutical intermediates suitable for direct use in API synthesis.
How to Synthesize L-Norvaline Efficiently
The synthesis of L-norvaline via this patented route requires precise control over reaction stoichiometry and temperature profiles to maximize yield and optical purity. The process begins with the careful addition of n-butyraldehyde to a buffered solution of acetone cyanohydrin, followed by high-pressure ammoniation. The resulting racemic amide must then undergo rigorous resolution and recrystallization before the final hydrolytic cleavage of the amide bond. For R&D teams looking to implement this technology, adherence to the specific solvent ratios and ion-exchange protocols described in the patent is essential for reproducibility. The detailed standardized synthesis steps, including specific molar ratios and temperature ramps, are outlined in the technical guide below to ensure successful replication in a pilot or production environment.
- Perform cyanation of n-butyraldehyde with acetone cyanohydrin under basic catalysis at 0-15°C to form 2-hydroxyl valeronitrile.
- React the nitrile intermediate with liquid ammonia for ammonification, followed by dehydration to obtain 2-amino valeronitrile.
- Hydrolyze the amino nitrile in concentrated sulfuric acid to form racemic 2-amino valeramide, then resolve using L-tartrate.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain directors, the transition from fermentation to this chemical synthesis route offers profound strategic benefits regarding cost stability and supply security. The reliance on n-butyraldehyde and acetone cyanohydrin means that the raw material base is anchored in the petrochemical industry, which generally offers greater price stability and availability compared to the specialized nutrients and enzymes required for fermentation. This shift fundamentally alters the cost structure of the intermediate, removing the high overhead associated with maintaining sterile biological environments and managing biological waste streams. Consequently, manufacturers can achieve cost reduction in pharmaceutical intermediate manufacturing through simplified infrastructure requirements and higher volumetric productivity per batch.
- Cost Reduction in Manufacturing: The elimination of fermentation tanks and the associated sterilization utilities significantly lowers capital expenditure (CAPEX) and operating expenditure (OPEX). The chemical process utilizes standard glass-lined or stainless steel reactors, which are ubiquitous in fine chemical plants, reducing the need for specialized equipment investment. Furthermore, the higher concentration of product in the reaction mixture compared to fermentation broths drastically reduces the energy costs associated with solvent removal and product concentration. By avoiding the low yields typical of biological systems, the effective cost per kilogram of the active intermediate is substantially decreased, providing a competitive edge in pricing negotiations for downstream API production.
- Enhanced Supply Chain Reliability: Chemical synthesis is inherently more predictable than biological fermentation, which is susceptible to strain degeneration, contamination, and batch-to-batch variability. This reliability ensures a consistent supply of L-norvaline, mitigating the risk of production stoppages that could disrupt the supply of Perindopril. The use of commodity chemicals also means that raw material sourcing is not limited to a few specialized biological suppliers but can be diversified across the global chemical market. This diversification strengthens the supply chain against regional disruptions and ensures that reliable pharmaceutical intermediate supplier status can be maintained even during market fluctuations.
- Scalability and Environmental Compliance: The process is designed for straightforward scale-up, moving from laboratory glassware to industrial reactors without the complex engineering challenges of scaling oxygen transfer and mixing in viscous fermentation broths. Additionally, the use of ion exchange resin for the final hydrolysis step minimizes the generation of inorganic salt waste, a common environmental burden in amino acid production. The ability to recover and recycle the L-tartrate resolving agent further enhances the environmental profile of the process, aligning with modern green chemistry principles and reducing the costs associated with waste treatment and disposal.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this L-norvaline synthesis technology. These insights are derived directly from the patent specifications and are intended to clarify the operational advantages and chemical principles underlying the process. Understanding these details is crucial for technical teams evaluating the feasibility of adopting this route for commercial production.
Q: Why is chemical synthesis preferred over fermentation for L-norvaline?
A: Chemical synthesis offers significantly higher yields and shorter production cycles compared to fermentation methods, which typically suffer from low output (around 3.7g/L) and complex downstream processing.
Q: What are the key starting materials for this L-norvaline process?
A: The process utilizes n-butyraldehyde and acetone cyanohydrin as the primary raw materials, which are readily available commodity chemicals, ensuring supply chain stability.
Q: How is chirality controlled in this synthetic route?
A: Chirality is established through a classical resolution step where the racemic 2-amino valeramide is reacted with L-tartrate to form a diastereomeric salt, allowing for the isolation of the L-enantiomer.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable L-Norvaline Supplier
The technological potential of this chemical synthesis route for L-norvaline is immense, offering a clear path to high-volume, cost-effective production of this critical chiral building block. At NINGBO INNO PHARMCHEM, we possess the technical expertise and infrastructure to translate such patented methodologies into commercial reality. Our team has extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the transition from lab-scale optimization to full-scale manufacturing is seamless. We maintain stringent purity specifications and operate rigorous QC labs equipped to verify the optical purity and chemical identity of every batch, guaranteeing that our intermediates meet the exacting standards required for API synthesis.
We invite pharmaceutical companies and contract manufacturers to collaborate with us to leverage this advanced synthesis technology for their Perindopril supply chains. By partnering with us, you gain access to a Customized Cost-Saving Analysis tailored to your specific volume requirements and quality needs. We encourage you to contact our technical procurement team today to request specific COA data and discuss route feasibility assessments that can optimize your production costs and secure your supply of high-quality L-norvaline.
