Revolutionizing Tenofovir Manufacturing: A Deep Dive into the Novel Dimethyl Phosphonate Route
The pharmaceutical landscape for antiretroviral therapy continues to evolve, driven by the urgent need for accessible and cost-effective treatments for HIV and Hepatitis B. Central to this effort is Tenofovir, a potent nucleotide analog whose demand necessitates robust and scalable manufacturing processes. Patent CN102219805A introduces a transformative production methodology that fundamentally alters the synthetic pathway of this critical active pharmaceutical ingredient (API). By shifting from traditional diethyl phosphonate precursors to a novel dimethyl phosphonate strategy, this technology addresses long-standing bottlenecks in hydrolysis efficiency and impurity management. 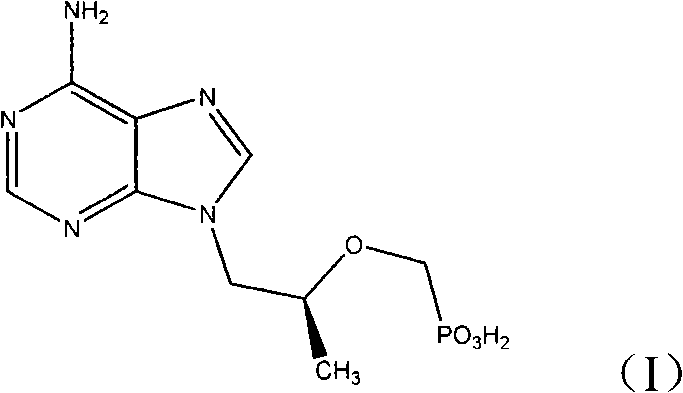 As depicted in the structural formula (I), the integrity of the phosphonate moiety is paramount, and this patent offers a refined approach to constructing it with greater operational simplicity. For global supply chain leaders and R&D directors, understanding this shift is crucial for optimizing the procurement of high-purity pharmaceutical intermediates.
As depicted in the structural formula (I), the integrity of the phosphonate moiety is paramount, and this patent offers a refined approach to constructing it with greater operational simplicity. For global supply chain leaders and R&D directors, understanding this shift is crucial for optimizing the procurement of high-purity pharmaceutical intermediates.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the industrial synthesis of Tenofovir has relied heavily on routes involving diethyl phosphonate intermediates, which present significant logistical and chemical challenges. A primary drawback lies in the deprotection phase, where the removal of ethyl groups requires harsh conditions, typically employing a trimethylchlorosilane and sodium bromide system. This reagent combination is not only costly but also extremely sensitive to moisture, necessitating rigorous drying protocols such as azeotropic distillation with cyclohexane to maintain reaction integrity. Furthermore, the phosphonylation step generates a complex mixture of magnesium salts, including magnesium p-toluenesulfonate and excess magnesium tert-butoxide. In conventional workflows, this magnesium salt sludge must be laboriously separated from the organic phase, often requiring multiple extraction and filtration cycles that trap product and reduce overall yield. 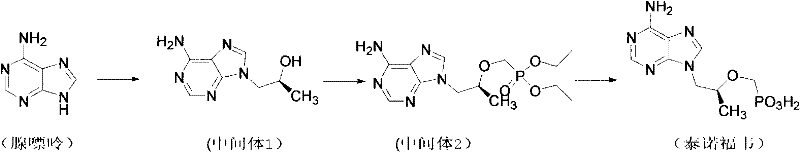 These operational complexities translate directly into higher production costs, increased safety risks due to moisture sensitivity, and a larger environmental footprint from solvent and reagent consumption.
These operational complexities translate directly into higher production costs, increased safety risks due to moisture sensitivity, and a larger environmental footprint from solvent and reagent consumption.
The Novel Approach
The innovation disclosed in CN102219805A circumvents these hurdles by utilizing tolysulfonyloxy dimethyl methyl phosphonate as the key phosphonylating agent. This strategic substitution results in the formation of Intermediate 3, a dimethyl phosphonate derivative that possesses inherently higher hydrolytic activity than its diethyl counterpart. Consequently, the deprotection step can be achieved using common, inexpensive mineral acids such as hydrochloric acid, hydrobromic acid, or sulfuric acid under reflux conditions. This eliminates the dependency on expensive silylating agents and the associated moisture control measures. Moreover, the process ingeniously integrates the removal of magnesium by-products into the hydrolysis step itself; the excess inorganic acid dissolves the magnesium salts, allowing them to be washed away during the final aqueous workup. 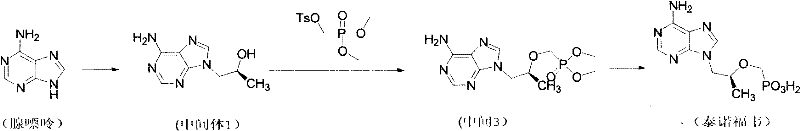 This streamlined workflow drastically simplifies the isolation of the final product, reducing unit operations and enhancing the overall robustness of the manufacturing process for commercial scale-up of complex antiviral intermediates.
This streamlined workflow drastically simplifies the isolation of the final product, reducing unit operations and enhancing the overall robustness of the manufacturing process for commercial scale-up of complex antiviral intermediates.
Mechanistic Insights into Dimethyl Phosphonate Hydrolysis
The core chemical advantage of this novel route lies in the differential reactivity of methyl versus ethyl esters in the context of phosphonic acid deprotection. While both are alkyl esters, the dimethyl phosphonate structure in Intermediate 3 demonstrates a kinetic preference for acid-catalyzed hydrolysis that the diethyl structure lacks under mild conditions. In the presence of strong mineral acids like concentrated hydrochloric acid, the methyl groups are cleaved efficiently to reveal the free phosphonic acid moiety of Tenofovir. This reactivity profile allows the reaction to proceed to completion without the need for the aggressive Lewis acid conditions (TMSCl/NaBr) required to activate the more sterically hindered or electronically stable diethyl esters. From a mechanistic standpoint, this suggests a lower activation energy barrier for the nucleophilic attack of water on the phosphorus center when methyl groups are the leaving groups, facilitating a cleaner conversion.
Furthermore, the interaction between the reaction matrix and the inorganic acid plays a pivotal role in impurity control. In traditional methods, the presence of magnesium salts can interfere with downstream processing, often acting as adsorption sites for the product or complicating filtration. In this new protocol, the addition of mineral acid serves a dual purpose: it catalyzes the ester hydrolysis and simultaneously converts insoluble magnesium alkoxides and sulfonates into soluble magnesium chloride or sulfate salts. This solubilization prevents the formation of gelatinous precipitates that often plague filtration processes in nucleotide synthesis. By maintaining a homogeneous solution during the critical hydrolysis phase, the process ensures that the product remains in solution until the precise moment of crystallization induced by pH adjustment, thereby maximizing recovery and purity.
How to Synthesize Tenofovir Efficiently
The synthesis of Tenofovir via this novel pathway is designed for operational ease and scalability, making it an attractive option for contract development and manufacturing organizations (CDMOs). The process begins with the alkylation of adenine using (R)-propylene carbonate in a polar aprotic solvent like DMF, facilitated by a base such as sodium hydroxide at elevated temperatures to yield Intermediate 1. This chiral intermediate is then subjected to phosphonylation with the specialized dimethyl phosphonate tosylate in N-methylpyrrolidone (NMP) using magnesium tert-butoxide. Unlike previous methods, the resulting reaction mixture containing Intermediate 3 does not require immediate purification. Instead, it proceeds directly to hydrolysis upon the addition of mineral acid, followed by a straightforward extractive workup and pH-controlled crystallization. For detailed standard operating procedures and specific reaction parameters, please refer to the guide below.
- Alkylation of Adenine with (R)-propylene carbonate in DMF with NaOH at 120°C to form Intermediate 1.
- Condensation of Intermediate 1 with tolysulfonyloxy dimethyl methyl phosphonate in NMP using magnesium tert-butoxide to yield Intermediate 3.
- Direct hydrolysis of Intermediate 3 using mineral acids (HCl, HBr, or H2SO4) under reflux, followed by pH adjustment and crystallization to isolate Tenofovir.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the adoption of this novel synthesis route offers compelling economic and logistical benefits that extend beyond simple yield improvements. The elimination of specialized reagents like trimethylchlorosilane and sodium bromide removes a significant cost driver from the bill of materials, as these reagents are not only expensive but also require careful handling and storage due to their reactivity. Additionally, the ability to use standard mineral acids for the final deprotection step simplifies the supply chain, as these commodities are readily available globally and do not suffer from the same supply volatility as specialty fine chemicals. This shift contributes to substantial cost savings in pharmaceutical intermediates manufacturing by reducing raw material expenses and minimizing the need for complex inventory management of hazardous substances.
- Cost Reduction in Manufacturing: The most significant financial impact arises from the simplification of the deprotection and workup stages. By removing the requirement for azeotropic drying with cyclohexane and the use of expensive silylating agents, the process drastically reduces solvent consumption and reagent costs. The ability to dissolve magnesium by-products in situ rather than filtering them out as a separate unit operation saves considerable labor time and reduces product loss associated with mechanical separations. This streamlined approach leads to a more efficient use of reactor capacity and utilities, ultimately driving down the cost per kilogram of the final API.
- Enhanced Supply Chain Reliability: Reliance on moisture-sensitive reagents often introduces variability and risk into the supply chain, as any breach in packaging or handling can compromise an entire batch. By transitioning to a moisture-tolerant system based on mineral acids, the manufacturing process becomes far more robust and forgiving. This reliability ensures consistent batch-to-batch quality and reduces the likelihood of production delays caused by reagent degradation or failed quality control tests. Furthermore, the use of common solvents and acids means that sourcing is less dependent on niche suppliers, enhancing the overall resilience of the supply network for high-purity antiviral intermediates.
- Scalability and Environmental Compliance: From an environmental, health, and safety (EHS) perspective, this process represents a significant improvement. The avoidance of silane by-products reduces the burden on waste treatment facilities and minimizes the release of volatile silicon compounds. The reduction in solvent usage, particularly the elimination of cyclohexane washes, lowers the facility's volatile organic compound (VOC) emissions. These factors make the process easier to scale from pilot plant to commercial production while maintaining strict adherence to increasingly stringent environmental regulations, ensuring long-term operational sustainability.
Frequently Asked Questions (FAQ)
The following questions address common technical inquiries regarding the implementation and benefits of this novel Tenofovir production process. These insights are derived directly from the experimental data and comparative analysis presented in the patent literature, providing clarity on how this method outperforms legacy technologies. Understanding these nuances is essential for technical teams evaluating process transfer opportunities.
Q: Why is the dimethyl phosphonate intermediate superior to the diethyl variant in Tenofovir synthesis?
A: The dimethyl phosphonate intermediate (Intermediate 3) exhibits significantly higher hydrolytic activity compared to the diethyl variant. This allows for the use of common mineral acids like hydrochloric acid for deprotection, eliminating the need for harsh and expensive reagents like trimethylchlorosilane and sodium bromide required for the diethyl ester.
Q: How does this new process handle the magnesium salt by-products generated during phosphonylation?
A: Unlike conventional methods that require complex separation of magnesium salts prior to hydrolysis, this novel process allows the magnesium salt mixture to remain in the reaction system. The subsequent addition of excess inorganic acid dissolves these salts, allowing them to be removed easily during the aqueous workup and filtration steps.
Q: What are the environmental and safety advantages of using mineral acid hydrolysis?
A: Replacing the trimethylchlorosilane/sodium bromide system with mineral acids eliminates the generation of silane by-products and reduces the reliance on moisture-sensitive reagents. This significantly lowers production risks, simplifies solvent handling (no need for azeotropic drying with cyclohexane), and results in a more environmentally friendly waste profile.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Tenofovir Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical importance of efficient and scalable synthesis routes for life-saving antiviral medications. Our team of expert chemists has extensively analyzed the technological advancements presented in CN102219805A and is well-positioned to implement this novel dimethyl phosphonate pathway. We possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the transition from laboratory optimization to full-scale manufacturing is seamless. Our state-of-the-art facilities are equipped with rigorous QC labs capable of meeting stringent purity specifications, guaranteeing that every batch of Tenofovir intermediate meets the highest global standards for pharmaceutical applications.
We invite pharmaceutical partners and generic drug manufacturers to collaborate with us to leverage these process improvements for their supply chains. By adopting this advanced synthesis route, you can achieve significant operational efficiencies and cost optimizations. Please contact our technical procurement team today to request a Customized Cost-Saving Analysis tailored to your specific volume requirements. We are ready to provide specific COA data and comprehensive route feasibility assessments to support your next project milestone.
