Advanced Low-Temperature Synthesis of High-Purity Triphenylphosphine for Industrial Scale-Up
The chemical industry constantly seeks more efficient pathways for producing essential reagents, and Patent CN113912645B represents a significant breakthrough in the synthesis of triphenylphosphine, a cornerstone reagent in organic synthesis widely used for Wittig reactions and as a ligand in transition metal catalysis. This patent discloses a novel preparation method that fundamentally shifts the reduction paradigm by utilizing sodium hydride and trimethyl borate to generate a reactive borohydride intermediate in situ, which then reduces triphenylphosphine dichloride to the target phosphine. Unlike traditional methods that rely on expensive silanes or hazardous aluminum powders, this approach operates under mild low-temperature conditions to stabilize key intermediates, thereby achieving high yields exceeding 88% and purity levels greater than 99.7%. For R&D directors and procurement managers alike, this technology offers a compelling value proposition by replacing costly raw materials with commodity chemicals while simultaneously simplifying the waste treatment profile, making it an ideal candidate for reliable triphenylphosphine supplier networks aiming for sustainable growth.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the industrial regeneration or synthesis of triphenylphosphine from its oxide or dichloride precursors has been plagued by significant economic and environmental inefficiencies. One prevalent method involves the direct reduction of triphenylphosphine oxide using silanes such as 1,3-tetramethyldisiloxane or trichlorosilane, as illustrated in the reaction scheme below. While this route is chemically direct, it suffers from severe drawbacks including the high market price of specialized silanes and the generation of substantial quantities of polydimethylsiloxane or polydichlorosilane byproducts, which require complex and costly environmental treatment protocols to meet discharge standards.
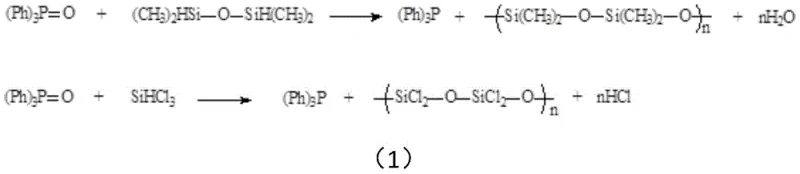
Another common industrial pathway involves converting triphenylphosphine oxide to triphenylphosphine dichloride using oxalyl chloride, followed by reduction with lithium aluminum hydride or aluminum powder. Although the byproducts like aluminum chloride are easier to handle than polysiloxanes, this method introduces its own set of critical challenges. The use of lithium aluminum hydride drives up raw material costs prohibitively for large-scale operations, while the use of aluminum powder requires a massive excess (often more than 3 times stoichiometric) to achieve high conversion. Furthermore, the post-reaction treatment of active aluminum residues involves acidification which generates explosive hydrogen gas and large volumes of aluminum salt wastewater, posing significant safety risks and environmental burdens that complicate commercial scale-up of complex pharmaceutical intermediates.
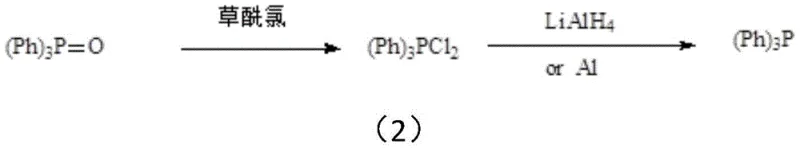
The Novel Approach
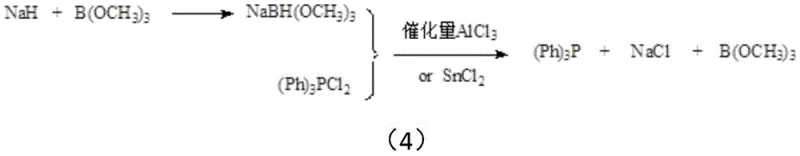
In stark contrast to these legacy technologies, the method disclosed in Patent CN113912645B introduces a streamlined, cost-effective strategy that leverages the reducing power of sodium hydride activated by trimethyl borate. This innovative process avoids the formation of difficult-to-treat polymeric silicon wastes and eliminates the safety hazards associated with active aluminum powder residues. By carefully controlling the reaction temperature below 10°C, the process stabilizes the sodium trimethoxyborohydride intermediate, allowing it to effectively reduce triphenylphosphine dichloride in the presence of a catalytic amount of aluminum trichloride or stannous chloride. The result is a cleaner reaction profile where the primary solid byproduct is merely sodium chloride, which can be easily filtered off, and the solvent system along with trimethyl borate can be recovered and recycled through azeotropic distillation. This shift not only drastically simplifies the downstream purification process but also aligns perfectly with modern green chemistry principles, offering a robust solution for cost reduction in pharmaceutical intermediates manufacturing.
Mechanistic Insights into Sodium Hydride-Borate Mediated Reduction
The core scientific innovation of this patent lies in the precise manipulation of reaction thermodynamics to stabilize a typically transient species. In conventional sodium borohydride synthesis, reaction conditions often involve temperatures exceeding 200°C and excess sodium hydride, which drives the complete conversion to NaBH4 but causes any intermediate alkoxyborohydrides to decompose rapidly. However, this patent reveals that sodium trimethoxyborohydride (NaBH(OCH3)3) can be stabilized in solution if the temperature is strictly maintained below 10°C, preferably between -50°C and 10°C. At temperatures above this threshold, the intermediate undergoes rapid disproportionation and decomposition, releasing gaseous borane and forming sodium methoxide, which renders the reducing agent ineffective for the subsequent phosphine synthesis. This delicate thermal window is critical for R&D teams to replicate, as it ensures the active hydride species remains available for the nucleophilic attack on the phosphorus center of the triphenylphosphine dichloride.
Furthermore, the inclusion of a Lewis acid catalyst, such as aluminum trichloride or stannous chloride, plays a pivotal role in facilitating the reduction step. These catalysts likely coordinate with the phosphorus-chlorine bonds, increasing the electrophilicity of the phosphorus atom and lowering the activation energy for the hydride transfer from the boron species. This catalytic enhancement allows the reaction to proceed efficiently even with the milder reducing power of the alkoxyborohydride compared to pure borohydride. From an impurity control perspective, this mechanism is highly advantageous because it avoids the introduction of transition metals that are difficult to remove from the final product. The only inorganic byproduct generated is sodium chloride, which precipitates out of the organic solvent matrix, allowing for a simple filtration step that significantly enhances the purity of the crude triphenylphosphine before the final crystallization, ensuring the material meets stringent purity specifications required for high-value applications.
How to Synthesize Triphenylphosphine Efficiently
The synthesis protocol outlined in the patent provides a clear roadmap for transitioning from laboratory discovery to pilot plant production, emphasizing strict temperature control and reagent addition rates to manage exotherms and intermediate stability. The process begins with the generation of the active reducing agent in a dedicated vessel under inert atmosphere, followed by the controlled addition to the phosphine precursor solution containing the catalyst. Detailed standardized operating procedures regarding solvent ratios, cooling capacities, and filtration parameters are essential for maintaining the high yields and purity reported in the experimental examples.
- Prepare sodium trimethoxyborohydride by reacting sodium hydride with trimethyl borate in solvent A (e.g., THF) at temperatures between -50°C and 10°C, ensuring the intermediate remains stable.
- Add the prepared sodium trimethoxyborohydride solution dropwise to a solution of triphenylphosphine dichloride containing a catalytic amount of aluminum trichloride or stannous chloride, maintaining the temperature between -20°C and 10°C.
- Filter off sodium chloride byproduct, recover solvents via distillation, hydrolyze the residue with dilute hydrochloric acid, and purify the crude product through crystallization to obtain high-purity triphenylphosphine.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the adoption of this novel synthesis route offers transformative benefits that extend far beyond simple yield improvements. The primary driver for cost optimization is the substitution of expensive, specialized reducing agents like lithium aluminum hydride or tetramethyldisiloxane with sodium hydride and trimethyl borate, which are commodity chemicals available in bulk quantities with stable pricing structures. This raw material swap fundamentally alters the cost structure of the manufacturing process, leading to substantial cost savings without compromising on product quality. Moreover, the ability to recover and recycle the solvent system and trimethyl borate through distillation creates a closed-loop material flow that minimizes raw material consumption and reduces the volume of purchased inputs required per kilogram of final product, further enhancing the economic viability of the process.
- Cost Reduction in Manufacturing: The elimination of high-cost reagents and the minimization of waste treatment expenses directly contribute to a lower cost of goods sold. By avoiding the generation of polymeric silicon wastes or hazardous aluminum sludge, the facility saves significantly on environmental compliance and waste disposal fees, which are often hidden but substantial costs in fine chemical manufacturing. The simplified workup procedure, which relies on filtration and crystallization rather than complex extraction or chromatography, also reduces labor hours and utility consumption, driving overall operational efficiency.
- Enhanced Supply Chain Reliability: Relying on commodity feedstocks like sodium hydride mitigates the risk of supply disruptions that often plague specialized reagent markets. Since the process does not depend on single-source suppliers for exotic catalysts or reducing agents, procurement teams can secure multiple sources for raw materials, ensuring continuous production even during market volatility. Additionally, the improved safety profile of the process, which avoids the handling of pyrophoric aluminum powders and the generation of hydrogen gas during workup, reduces the likelihood of unplanned shutdowns due to safety incidents, thereby guaranteeing more reliable delivery schedules for customers.
- Scalability and Environmental Compliance: The inherent safety and simplicity of this method make it exceptionally well-suited for commercial scale-up. The absence of corrosive trichlorosilane or explosive hydrogen evolution simplifies the engineering requirements for large-scale reactors, allowing for faster deployment of production capacity. From an environmental standpoint, the generation of benign sodium chloride waste and the recyclability of solvents align with increasingly strict global environmental regulations, future-proofing the manufacturing site against tightening emission standards and reducing the carbon footprint of the supply chain.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this patented technology, drawing directly from the experimental data and beneficial effects described in the documentation. Understanding these nuances is crucial for technical teams evaluating the feasibility of adopting this route for their specific production needs.
Q: Why is temperature control below 10°C critical in this synthesis?
A: According to Patent CN113912645B, the intermediate sodium trimethoxyborohydride is unstable above 10°C. At higher temperatures, it undergoes disproportionation and decomposition into sodium methoxide and gaseous borane byproducts, significantly reducing yield and creating safety hazards. Maintaining low temperatures ensures the intermediate remains stable in solution for the subsequent reduction step.
Q: What are the primary environmental advantages of this method compared to silane reduction?
A: Traditional silane reduction methods generate large amounts of polydimethylsiloxane waste, which is difficult and costly to treat. In contrast, this novel method primarily produces sodium chloride as a solid byproduct, which is easily filtered and disposed of. Additionally, the main solvents and auxiliary agents like trimethyl borate can be recovered and recycled via azeotropic distillation, drastically reducing chemical waste.
Q: How does this process improve supply chain reliability for triphenylphosphine?
A: This method utilizes sodium hydride and trimethyl borate, which are commodity chemicals with stable global supply chains, unlike expensive or specialized reducing agents like lithium aluminum hydride or specific silanes. The elimination of hazardous active aluminum powder residues also simplifies safety protocols and waste disposal, ensuring more consistent production schedules and reduced regulatory bottlenecks.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Triphenylphosphine Supplier
At NINGBO INNO PHARMCHEM, we recognize that the transition to a more efficient synthesis route requires a partner with deep technical expertise and robust manufacturing capabilities. As a leading CDMO, we possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the delicate low-temperature conditions and catalytic requirements of this patent are meticulously managed at scale. Our state-of-the-art facilities are equipped with rigorous QC labs capable of verifying stringent purity specifications, guaranteeing that every batch of triphenylphosphine meets the exacting standards required for pharmaceutical and electronic applications.
We invite you to collaborate with us to leverage this advanced technology for your supply chain. By engaging with our technical procurement team, you can request a Customized Cost-Saving Analysis tailored to your specific volume requirements, demonstrating exactly how this greener, more efficient route can impact your bottom line. We encourage potential partners to contact us immediately to discuss route feasibility assessments and to obtain specific COA data for our high-purity triphenylphosphine, securing a competitive advantage in your respective markets through superior material quality and supply reliability.
