Advanced Synthesis of Aminophenyl-Beta-Hydroxyethyl Sulfone Sulfate for Global Dye Markets
Advanced Synthesis of Aminophenyl-Beta-Hydroxyethyl Sulfone Sulfate for Global Dye Markets
The global demand for high-performance reactive dyes continues to drive innovation in the synthesis of key intermediates, specifically aminophenyl-beta-hydroxyethyl sulfone sulfate. A pivotal advancement in this sector is detailed in patent CN101525309B, which outlines a revolutionary production method that addresses the longstanding inefficiencies of traditional manufacturing. This patent introduces a streamlined synthetic pathway starting from p-nitrochlorobenzene and mercaptoethanol, bypassing the hazardous and waste-intensive steps associated with the conventional acetanilide route. By leveraging catalytic oxidation and controlled hydrogenation, this technology not only enhances the purity profile of the final intermediate but also aligns with modern green chemistry principles. For R&D directors and procurement specialists, understanding this shift is critical, as it represents a move towards more sustainable and cost-effective supply chains for textile chemicals. The following analysis dissects the technical merits and commercial implications of adopting this superior methodology.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the industrial production of p-amino phenyl-beta-hydroxyethyl sulfone sulfate has relied on a cumbersome multi-step sequence initiated by the sulfonation of acetanilide. This legacy process involves the use of excessive chlorosulfonic acid, a highly corrosive reagent that requires stringent temperature control below -10°C using energy-intensive ice-salt baths to manage the violent exothermic reaction. The generation of substantial hydrogen chloride gas during this phase necessitates complex absorption systems to recover industrial hydrochloric acid, adding to the capital expenditure and operational overhead. Furthermore, the subsequent hydrolysis and reduction steps generate massive quantities of waste, including 8-10 tons of waste acid and 6-8 tons of high-salt wastewater per ton of product. The overall yield of this archaic method stagnates around 70%, leading to significant raw material wastage and a heavy environmental burden that complicates regulatory compliance for manufacturers.
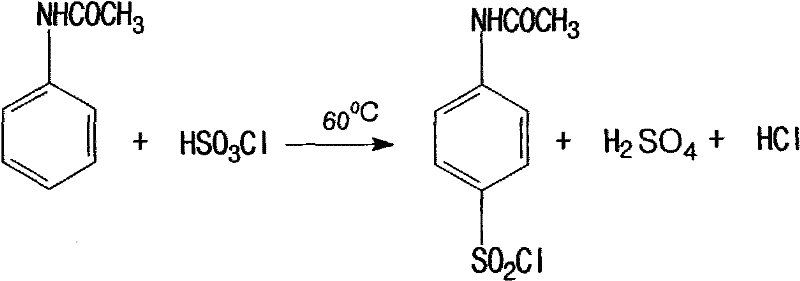
The Novel Approach
In stark contrast, the novel approach disclosed in the patent utilizes a direct nucleophilic substitution strategy that fundamentally simplifies the molecular construction. By reacting p-nitrochlorobenzene directly with mercaptoethanol in a dimethylformamide (DMF) solvent system, the process eliminates the need for dangerous chlorosulfonic acid entirely. The subsequent oxidation of the thioether intermediate is achieved using hydrogen peroxide catalyzed by tungstate, a greener oxidant that produces water as the primary by-product rather than toxic heavy metal waste. This streamlined pathway not only boosts the final product yield to approximately 85% but also reduces wastewater discharge to a mere 1-2 tons per ton of product. The elimination of high-salt waste streams and the reduction in raw material consumption mark a paradigm shift in how this critical dye intermediate is manufactured, offering a clear competitive advantage.
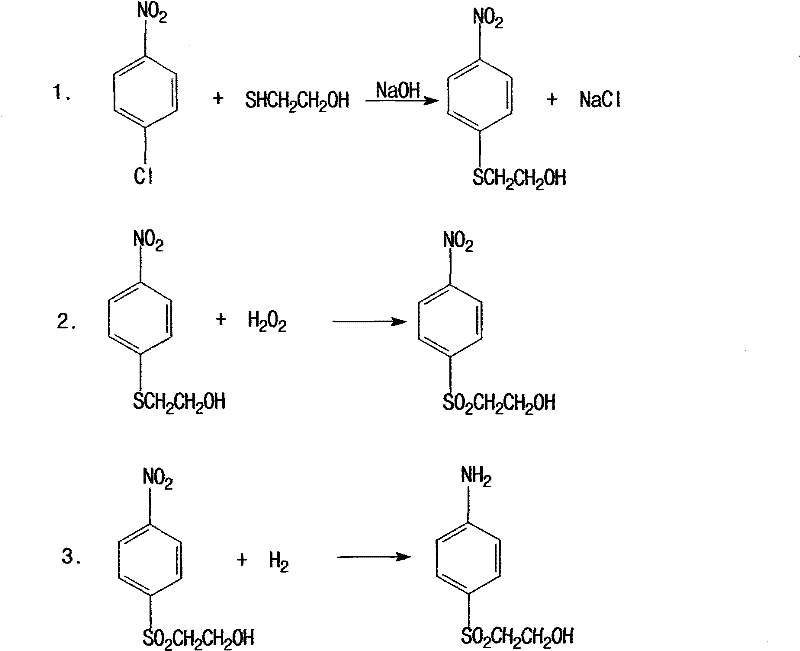
Mechanistic Insights into Tungstate-Catalyzed Oxidation and Hydrogenation
The core chemical innovation lies in the selective oxidation of the sulfide bond to a sulfone without affecting other sensitive functional groups on the aromatic ring. In the patented process, sodium tungstate acts as a phase-transfer-like catalyst that activates hydrogen peroxide, facilitating the transfer of oxygen atoms to the sulfur center. This reaction is conducted at controlled temperatures between 95-100°C, ensuring that the oxidation proceeds to the sulfone stage (SO2) without over-oxidation to sulfonic acids, which would render the molecule useless for subsequent vinyl sulfone formation. The precision of this catalytic cycle minimizes the formation of side products, thereby simplifying the downstream purification process and enhancing the overall purity of the crystallized intermediate. This level of control is unattainable in the old process where harsh acidic conditions often lead to unpredictable degradation.
Following oxidation, the nitro group is reduced to an amine using a palladium carbon catalyst under hydrogen pressure ranging from 1.5 to 2.5 MPa. This heterogeneous catalytic hydrogenation is highly efficient and allows for the easy recovery of the expensive palladium catalyst via filtration, contributing to cost stability. The mechanism involves the adsorption of hydrogen onto the palladium surface, followed by the stepwise reduction of the nitro group through nitroso and hydroxylamine intermediates to the final amine. The mild conditions of this hydrogenation step preserve the integrity of the beta-hydroxyethyl sulfone moiety, preventing the elimination reaction that could prematurely form the reactive vinyl sulfone group before the desired esterification step. This sequential control ensures that the final esterification with sulfuric acid yields the stable sulfate ester ready for dye synthesis.
How to Synthesize p-Amino Phenyl-Beta-Hydroxyethyl Sulfone Sulfate Efficiently
The implementation of this synthesis route requires precise control over stoichiometry and thermal parameters to maximize the benefits described above. The process begins with the careful addition of sodium hydroxide to the reaction mixture of p-nitrochlorobenzene and mercaptoethanol, maintaining a temperature of 50-80°C to drive the substitution to completion while minimizing side reactions. Following the isolation of the thioether, the oxidation step demands a slow addition of hydrogen peroxide to manage the exotherm and ensure complete conversion. The subsequent hydrogenation and esterification steps must be monitored closely to prevent hydrolysis of the sulfate ester group. For a detailed breakdown of the specific operational parameters, reagent ratios, and equipment setup required to replicate this high-yield process, please refer to the standardized synthesis guide below.
- React p-Nitrophenyl chloride with mercaptoethanol in DMF solvent with sodium hydroxide at 50-80°C to form p-nitrophenyl-beta-hydroxyethyl thioether.
- Oxidize the thioether intermediate using hydrogen peroxide and a tungstate catalyst at 95-100°C to obtain the sulfone derivative.
- Perform catalytic hydrogenation on the oxidation product using a palladium carbon catalyst under 1.5-2.5MPa pressure to reduce the nitro group to an amine.
- Complete the synthesis by esterifying the hydrogenation product with concentrated sulfuric acid at 100-130°C to yield the final sulfate ester.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the adoption of this patented technology translates into tangible strategic benefits beyond simple chemical efficiency. The primary advantage lies in the drastic simplification of the waste management infrastructure required for production. By eliminating the generation of tons of waste acid and high-salt wastewater per batch, manufacturers can significantly reduce their expenditure on effluent treatment facilities and hazardous waste disposal fees. This reduction in environmental liability not only lowers operational costs but also mitigates the risk of production shutdowns due to regulatory non-compliance, ensuring a more reliable supply continuity for downstream dye manufacturers. Furthermore, the higher yield means that less raw material is required to produce the same amount of finished goods, effectively stretching the purchasing budget and improving margin potential in a competitive market.
- Cost Reduction in Manufacturing: The elimination of chlorosulfonic acid removes the need for specialized corrosion-resistant equipment and the associated maintenance costs, while the recovery of the palladium catalyst further optimizes the cost structure. The process avoids the expensive and energy-intensive cryogenic cooling required by the old method, leading to substantial savings in utility consumption. Additionally, the reduction in by-product formation means less material is lost to waste streams, directly improving the atom economy and reducing the effective cost per kilogram of the active intermediate.
- Enhanced Supply Chain Reliability: The raw materials for this new process, such as p-nitrochlorobenzene and mercaptoethanol, are widely available commodity chemicals with stable supply chains, unlike some of the more specialized reagents or conditions required in the legacy route. The robustness of the reaction conditions, which do not require extreme low temperatures or high vacuum for extended periods, makes the process less susceptible to equipment failure or utility fluctuations. This operational stability ensures consistent batch-to-batch quality and on-time delivery, which is critical for maintaining the production schedules of large-scale textile dyeing operations.
- Scalability and Environmental Compliance: The simplified workflow, consisting of fewer unit operations and safer reagents, facilitates easier scale-up from pilot plants to commercial production volumes without the exponential increase in safety risks seen in the sulfonation-heavy traditional method. The significant reduction in wastewater volume and toxicity aligns perfectly with increasingly stringent global environmental regulations, future-proofing the manufacturing facility against tighter emission standards. This eco-friendly profile enhances the brand value of the supplier, appealing to end-users in the textile industry who are under pressure to source sustainable and responsibly manufactured chemicals.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the production of aminophenyl-beta-hydroxyethyl sulfone sulfate using this advanced methodology. These answers are derived directly from the experimental data and beneficial effects reported in the patent literature, providing a transparent view of the technology's capabilities. Understanding these details helps stakeholders make informed decisions about integrating this intermediate into their supply chains.
Q: How does the new synthesis route improve yield compared to traditional methods?
A: The patented process achieves a product yield of approximately 85%, which is a significant improvement over the conventional acetanilide route that typically yields only around 70%. This increase is attributed to the milder reaction conditions and the avoidance of harsh hydrolysis steps that degrade product quality.
Q: What are the environmental benefits of this production method?
A: This method drastically reduces wastewater generation, lowering effluent from 6-8 tons per ton of product in the old process to just 1-2 tons. Additionally, it eliminates the production of large quantities of waste acid and salt by-products associated with chlorosulfonic acid usage.
Q: Which catalysts are utilized in the oxidation and reduction steps?
A: The process employs sodium tungstate as a catalyst for the oxidation step using hydrogen peroxide, ensuring selective conversion to the sulfone. For the reduction of the nitro group, a palladium carbon (Pd/C) catalyst is used under hydrogen pressure, allowing for efficient recovery and reuse.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Aminophenyl-Beta-Hydroxyethyl Sulfone Sulfate Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical role that high-quality intermediates play in the performance of final reactive dyes. As a leading CDMO expert, we possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the benefits of this patented synthesis are fully realized at an industrial level. Our state-of-the-art facilities are equipped with rigorous QC labs capable of meeting stringent purity specifications, guaranteeing that every batch of aminophenyl-beta-hydroxyethyl sulfone sulfate delivers consistent reactivity and color strength for your dye formulations. We are committed to bridging the gap between innovative patent chemistry and reliable commercial supply.
We invite you to collaborate with us to optimize your sourcing strategy for this essential textile chemical. Our technical team is ready to provide a Customized Cost-Saving Analysis tailored to your specific volume requirements, demonstrating how switching to this superior grade can lower your total cost of ownership. Please contact our technical procurement team today to request specific COA data and route feasibility assessments, and let us support your journey towards more efficient and sustainable dye manufacturing.
