Revolutionizing Water-Soluble Honokiol Synthesis: A Scalable Route for High-Purity Pharmaceutical Intermediates
Introduction to Advanced Magnolol Derivative Synthesis
The pharmaceutical industry continuously seeks efficient pathways to enhance the bioavailability of potent natural products, and magnolol and honokiol stand out as prime candidates due to their extensive pharmacological profile including anti-inflammatory and anti-tumor activities. However, their poor water solubility has historically limited their clinical application, necessitating complex derivatization strategies. Patent CN112125805A introduces a groundbreaking methodology for synthesizing water-soluble magnolol and honokiol derivatives, specifically addressing the critical bottleneck of regioselective functionalization. This technology leverages a unique activated ester system to achieve precise monohydroxy protection, thereby enabling highly selective nitration at the ortho-position of the remaining free hydroxyl group. 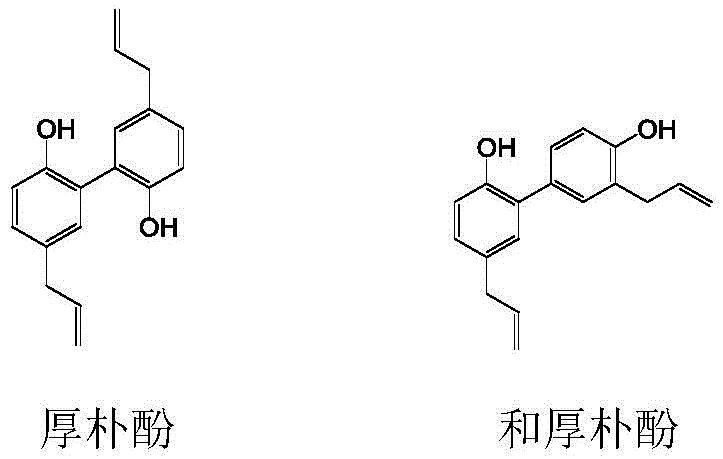 This innovation represents a significant leap forward for reliable pharmaceutical intermediate supplier networks, as it transforms a laboratory curiosity into a commercially viable process capable of meeting stringent purity specifications without the burden of expensive purification techniques.
This innovation represents a significant leap forward for reliable pharmaceutical intermediate supplier networks, as it transforms a laboratory curiosity into a commercially viable process capable of meeting stringent purity specifications without the burden of expensive purification techniques.
The core of this technological advancement lies in the meticulous control of reaction conditions and reagent selection, which collectively overcome the inherent symmetry and similar reactivity of the dihydroxy groups present in the magnolol and honokiol scaffolds. By shifting away from traditional acylation methods that often result in mixtures of isomers requiring tedious separation, this patent outlines a streamlined approach that prioritizes atomic economy and process safety. The ability to produce high-purity OLED material precursors or pharmaceutical intermediates with such precision is rare, and this specific pathway offers a robust solution for cost reduction in API manufacturing. As we delve deeper into the mechanistic nuances and operational parameters, it becomes evident that this process is not merely an incremental improvement but a fundamental rethinking of how complex phenolic compounds can be functionalized for industrial applications.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the derivatization of magnolol and honokiol has been plagued by poor selectivity and cumbersome purification requirements, creating significant barriers to large-scale production. Traditional methods often employ fatty acid chlorides, such as lauroyl chloride, to protect hydroxyl groups; however, these reagents possess flexible alkyl chains that fail to differentiate effectively between the two hydroxyl positions based on steric hindrance alone. As demonstrated in the comparative examples within the patent data, the use of lauroyl chloride results in a nitration step yield as low as 18%, necessitating multiple rounds of column chromatography to isolate the desired isomer. 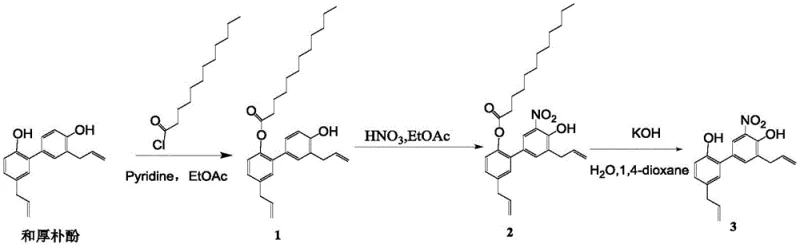 This reliance on chromatographic purification is economically prohibitive for commercial scale-up of complex polymer additives or pharmaceutical ingredients, as it drastically increases solvent consumption, processing time, and waste generation. Furthermore, the formation of isomeric byproducts complicates the regulatory landscape, as impurity profiles must be strictly controlled to meet international pharmacopoeia standards.
This reliance on chromatographic purification is economically prohibitive for commercial scale-up of complex polymer additives or pharmaceutical ingredients, as it drastically increases solvent consumption, processing time, and waste generation. Furthermore, the formation of isomeric byproducts complicates the regulatory landscape, as impurity profiles must be strictly controlled to meet international pharmacopoeia standards.
The Novel Approach
In stark contrast, the novel approach detailed in CN112125805A utilizes an activated ester generated in situ from p-toluenesulfonyl chloride (TsCl) and 1-hydroxybenzotriazole (HOBt) to achieve exceptional regioselectivity. This system creates a bulky, rigid aromatic protecting group that preferentially reacts with the less sterically hindered hydroxyl group, effectively directing subsequent electrophilic substitution to the desired position. 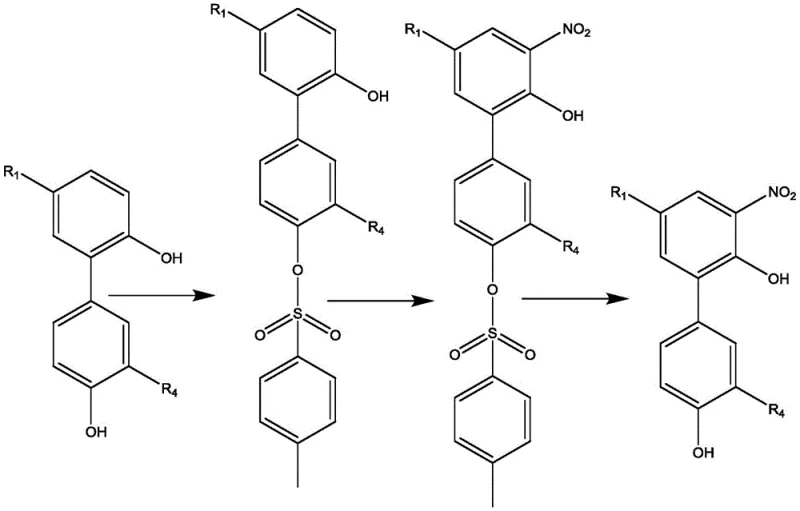 The result is a dramatic improvement in process efficiency, where the nitration step proceeds with high fidelity to the target ortho-position, yielding intermediates with purities exceeding 95% after simple crystallization. This methodological shift eliminates the need for column chromatography entirely, simplifying the workflow to a series of manageable liquid-liquid extractions and precipitations. For procurement managers, this translates to a significantly simplified supply chain and reduced dependency on specialized purification resins, while for R&D teams, it offers a reproducible protocol that can be reliably transferred from gram-scale optimization to multi-ton manufacturing campaigns.
The result is a dramatic improvement in process efficiency, where the nitration step proceeds with high fidelity to the target ortho-position, yielding intermediates with purities exceeding 95% after simple crystallization. This methodological shift eliminates the need for column chromatography entirely, simplifying the workflow to a series of manageable liquid-liquid extractions and precipitations. For procurement managers, this translates to a significantly simplified supply chain and reduced dependency on specialized purification resins, while for R&D teams, it offers a reproducible protocol that can be reliably transferred from gram-scale optimization to multi-ton manufacturing campaigns.
Mechanistic Insights into Selective Monohydroxy Protection
The success of this synthesis hinges on the sophisticated interplay between steric hindrance and electronic effects governed by the TsCl/HOBt activated ester system. Unlike flexible aliphatic acylating agents, the tosyl-HOBt ester presents a planar, rigid aromatic structure that cannot easily navigate the steric environment surrounding the more hindered hydroxyl group of the magnolol or honokiol molecule. Consequently, the reaction kinetics favor the attack of the more accessible hydroxyl group, establishing a kinetic control that dictates the regiochemistry of the entire synthetic sequence. This steric differentiation is further enhanced by operating at low temperatures, typically between 0°C and 10°C, which suppresses non-selective background reactions and ensures that the thermodynamic product distribution does not erode the kinetic selectivity achieved during the initial protection phase.
Following the selective protection, the nitration reaction is directed exclusively to the ortho-position of the remaining free phenol, driven by the strong activating nature of the hydroxyl group. The electron-donating capability of the free hydroxyl facilitates the electrophilic aromatic substitution with nitric acid, while the protected phenol remains inert under these conditions. Subsequent deprotection using aqueous alkali metal hydroxides, such as potassium hydroxide in 1,4-dioxane, cleanly removes the tosyl group without affecting the newly installed nitro group or the allyl side chains. This orthogonal stability is crucial for maintaining the integrity of the molecular scaffold, ensuring that the final nitrated intermediate retains the structural features necessary for downstream reduction and conjugation with amino acids or peptides to enhance water solubility.
How to Synthesize Nitrated Honokiol Intermediate Efficiently
The practical implementation of this synthesis involves a carefully orchestrated three-step sequence that balances reaction rate with selectivity to maximize overall yield. The process begins with the generation of the activated ester in a hydrophobic solvent like dichloromethane, followed by the addition of the substrate at controlled low temperatures to enforce selectivity. The subsequent nitration is performed using concentrated nitric acid in a solvent system that stabilizes the intermediate, and the final deprotection utilizes mild alkaline conditions to reveal the target dihydroxy-nitro structure. Detailed operational parameters, including specific molar ratios and solvent choices, are critical for replicating the high purity and yield reported in the patent examples. 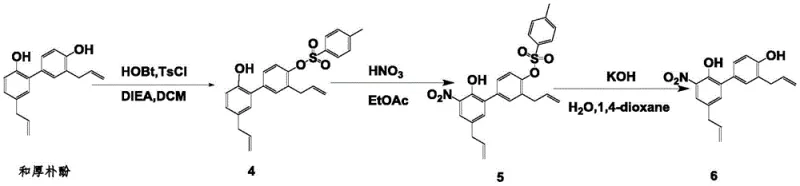 The following guide outlines the standardized procedure derived from the patent data, serving as a foundational reference for process chemists aiming to implement this technology in a GMP environment.
The following guide outlines the standardized procedure derived from the patent data, serving as a foundational reference for process chemists aiming to implement this technology in a GMP environment.
- Perform selective monohydroxy protection on honokiol using an activated ester formed from p-toluenesulfonyl chloride and 1-hydroxybenzotriazole in dichloromethane at 0-10°C.
- Conduct nitration of the protected intermediate using 60-70% nitric acid in a non-reactive solvent like ethyl acetate or dichloromethane at 0-25°C.
- Execute deprotection by treating the nitrated product with aqueous potassium hydroxide in 1,4-dioxane at 85°C, followed by acidification and crystallization.
Commercial Advantages for Procurement and Supply Chain Teams
The transition from academic synthesis to industrial manufacturing is often hindered by processes that are difficult to scale or rely on hazardous purification methods, but this patented technology directly addresses these supply chain vulnerabilities. By eliminating the need for column chromatography, the process drastically reduces the volume of organic solvents required for purification, leading to substantial cost savings in waste disposal and solvent recovery operations. This simplification also shortens the batch cycle time, allowing manufacturing facilities to increase throughput without requiring additional capital investment in reactor capacity. For supply chain heads, this means a more resilient production schedule with reduced lead time for high-purity pharmaceutical intermediates, ensuring consistent availability for downstream drug formulation.
- Cost Reduction in Manufacturing: The elimination of chromatographic purification represents a major economic advantage, as column chromatography is notoriously expensive and difficult to scale beyond kilogram quantities. By relying on crystallization and extraction, the process utilizes standard unit operations found in any multipurpose chemical plant, significantly lowering the barrier to entry for contract manufacturing organizations. Furthermore, the high selectivity of the reaction minimizes the formation of hard-to-remove isomers, reducing the loss of valuable starting materials and improving the overall mass balance of the process.
- Enhanced Supply Chain Reliability: The reagents employed in this synthesis, such as p-toluenesulfonyl chloride, HOBt, and nitric acid, are commodity chemicals with robust global supply chains, mitigating the risk of raw material shortages. The process does not rely on exotic catalysts or sensitive organometallic reagents that might require special handling or storage conditions, thereby simplifying logistics and inventory management. This accessibility ensures that production can be maintained continuously, supporting long-term supply agreements with pharmaceutical partners who require guaranteed continuity of supply for their clinical and commercial programs.
- Scalability and Environmental Compliance: The use of common solvents like dichloromethane, ethyl acetate, and 1,4-dioxane allows for established recycling protocols, aligning with modern green chemistry initiatives and environmental regulations. The absence of heavy metal catalysts in the nitration and deprotection steps simplifies the wastewater treatment process, reducing the environmental footprint of the manufacturing site. This compliance with environmental standards is increasingly critical for maintaining operating licenses and meeting the sustainability goals of multinational corporations seeking responsible suppliers for their active pharmaceutical ingredient supply chains.
Frequently Asked Questions (FAQ)
Understanding the technical specifics of this synthesis is vital for stakeholders evaluating its potential for integration into existing production lines. The following questions address common inquiries regarding the selectivity mechanisms, purification strategies, and scalability of the process described in patent CN112125805A. These answers are derived directly from the experimental data and technical disclosures within the patent, providing a factual basis for decision-making regarding technology adoption and process validation.
Q: How does the TsCl/HOBt system improve selectivity compared to traditional acylation?
A: The activated ester formed by TsCl and HOBt possesses a rigid aromatic structure that imposes significant steric hindrance. Unlike flexible fatty acid chlorides which react based on electronic activity, this system selectively protects the less sterically hindered hydroxyl group, drastically improving regioselectivity for subsequent ortho-nitration.
Q: Does this process require column chromatography for purification?
A: No, one of the primary advantages of this patented method is the elimination of column chromatography. The high selectivity of the protection and nitration steps allows for purification via simple crystallization and liquid-liquid extraction, making it suitable for industrial ton-scale production.
Q: What are the typical yields for the nitrated intermediate synthesis?
A: In the provided examples, the overall process demonstrates robust efficiency. For instance, the conversion of honokiol to the nitrated intermediate (Compound 6) achieves yields of approximately 61% for the protection step, 61% for nitration, and 74% for deprotection, with purities exceeding 95% after crystallization.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Honokiol Derivative Supplier
At NINGBO INNO PHARMCHEM, we recognize the transformative potential of this selective nitration technology for the development of next-generation anti-inflammatory and anti-tumor therapeutics. As a leading CDMO partner, we possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the theoretical benefits of this patent are fully realized in a commercial setting. Our state-of-the-art facilities are equipped with rigorous QC labs capable of verifying stringent purity specifications, guaranteeing that every batch of honokiol derivative meets the exacting standards required for pharmaceutical applications. We are committed to bridging the gap between innovative patent chemistry and reliable industrial supply.
We invite potential partners to engage with our technical procurement team to discuss how this advanced synthesis route can be tailored to your specific project needs. By requesting a Customized Cost-Saving Analysis, you can gain deeper insights into the economic benefits of switching to this chromatography-free process. We encourage you to contact us today to obtain specific COA data and route feasibility assessments, allowing you to make informed decisions that optimize both the quality and cost-efficiency of your supply chain for these high-value pharmaceutical intermediates.
