Advanced Synthesis of Honokiol Oxazole Derivatives for Next-Generation Antidiabetic Therapeutics
Advanced Synthesis of Honokiol Oxazole Derivatives for Next-Generation Antidiabetic Therapeutics
The pharmaceutical landscape for diabetes management is constantly evolving, driven by the need for more effective alpha-glucosidase inhibitors with improved safety profiles. Patent CN113234070A introduces a significant breakthrough in this domain by disclosing a novel series of honokiol thioether derivatives containing an oxazole ring. These compounds represent a strategic structural optimization of honokiol, a natural biphenyl neolignan derived from Magnolia officinalis, known for its broad pharmacological activities. By integrating a heterocyclic oxazole moiety via a thioether linkage, the invention achieves a synergistic enhancement in biological potency, specifically targeting alpha-glucosidase enzymes. This technical advancement offers a robust foundation for developing new candidate drug molecules, addressing the critical demand for high-efficacy antidiabetic agents in the global market.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Traditional approaches to modifying honokiol often focus on simple esterification or glycosylation of the phenolic hydroxyl groups to improve water solubility. While these methods can enhance physicochemical properties, they frequently fail to significantly boost the intrinsic inhibitory activity against metabolic enzymes like alpha-glucosidase. Furthermore, many existing synthetic routes for complex honokiol analogs involve multi-step protection and deprotection strategies, leading to reduced overall yields and increased production costs. The reliance on harsh reaction conditions or expensive transition metal catalysts in conventional cross-coupling reactions also poses challenges for environmental compliance and large-scale manufacturing efficiency. Consequently, there is a persistent need for a streamlined synthetic pathway that can introduce potent pharmacophores without compromising process simplicity.
The Novel Approach
The methodology outlined in the patent presents a divergent yet convergent strategy that overcomes these historical bottlenecks. Instead of merely modifying the hydroxyl groups, this approach constructs a rigid oxazole ring system attached via a sulfur bridge, fundamentally altering the electronic and steric environment of the molecule. This structural rigidity is crucial for locking the molecule into a bioactive conformation that fits the enzyme active site more effectively. The synthesis avoids complex catalytic cycles, relying instead on classical nucleophilic substitutions and condensations that are well-understood and easily controlled. This not only simplifies the purification process but also ensures a cleaner impurity profile, which is paramount for pharmaceutical intermediates intended for clinical development.
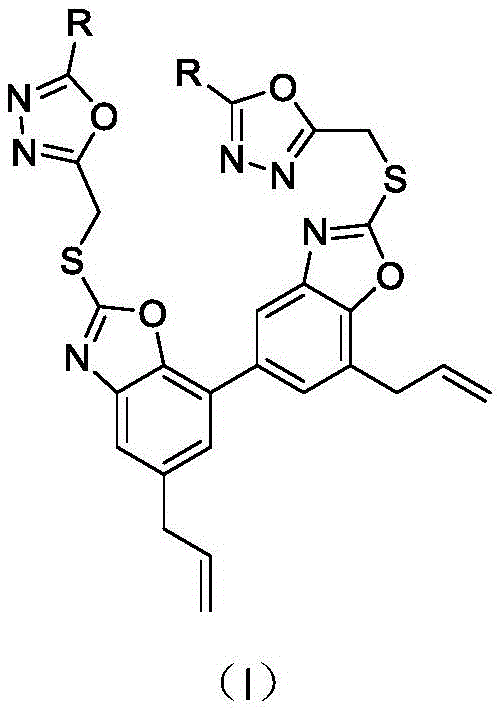
Mechanistic Insights into Heterocyclic Coupling and Cyclization
The core of this synthetic innovation lies in the construction of the 1,3,4-oxadiazole and benzoxazole-thione motifs. The formation of the oxazole ring precursor involves the cyclodehydration of acylhydrazides with chloroacetyl chloride, followed by treatment with phosphorus oxychloride. This sequence efficiently generates the reactive 2-(chloromethyl)-1,3,4-oxadiazole intermediates. On the honokiol side, the transformation of the amino group into an oxazothione ring using carbon disulfide under basic conditions creates a highly nucleophilic sulfur center. The final coupling step is a classic SN2 reaction where the thione sulfur attacks the chloromethyl group of the oxadiazole. This mechanism is highly favorable due to the soft-soft interaction between the sulfur nucleophile and the benzylic carbon electrophile, ensuring high conversion rates even at room temperature.
From an impurity control perspective, the stepwise nature of the synthesis allows for rigorous quality checks at each stage. The nitration of honokiol is performed at low temperatures (0-5°C) to prevent over-nitration or oxidation of the allyl groups, which are sensitive to oxidative degradation. The subsequent reduction using stannous chloride is selective for the nitro group, leaving the allyl double bonds intact. This chemoselectivity is vital for maintaining the structural integrity of the honokiol backbone. The final purification via column chromatography or recrystallization removes unreacted starting materials and inorganic salts, resulting in a high-purity final product suitable for biological evaluation and further drug development processes.
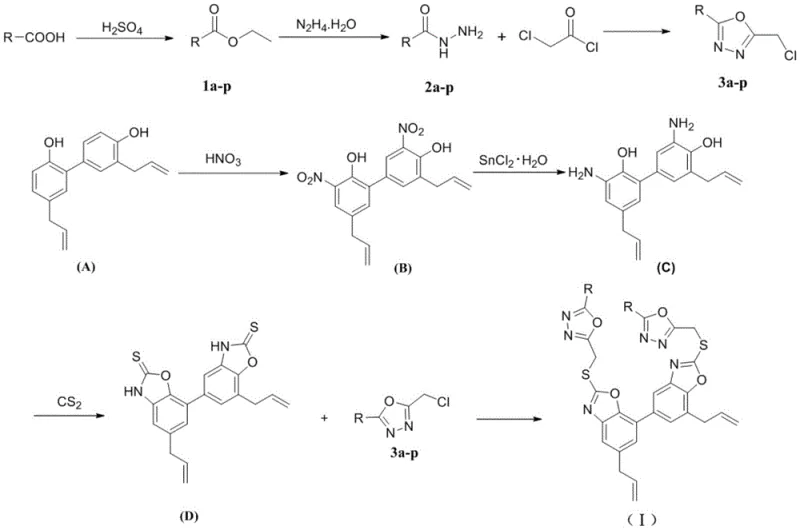
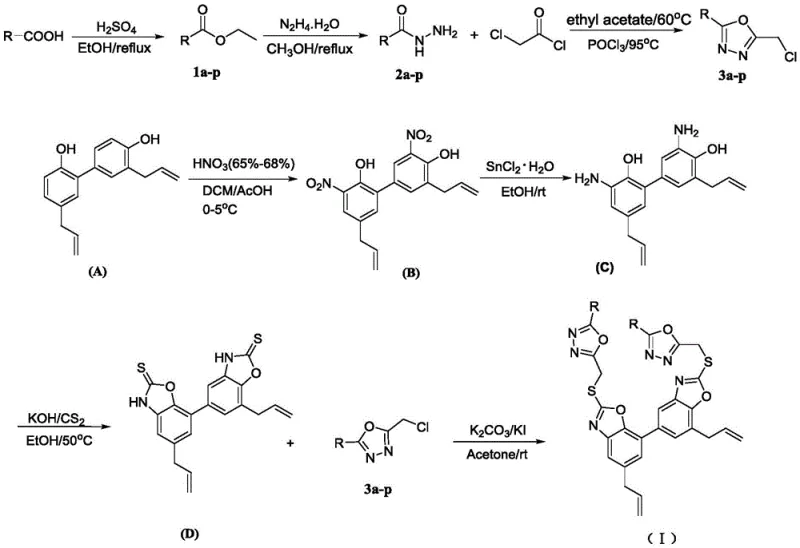
How to Synthesize Honokiol Oxazole Derivatives Efficiently
The synthesis of these high-value intermediates follows a logical progression from readily available starting materials. The process begins with the functionalization of benzoic acid derivatives and parallel modification of the natural product honokiol. The convergence of these two streams in the final step maximizes efficiency and allows for the rapid generation of a library of analogs by simply varying the substituent on the initial benzoic acid. This modularity is a key advantage for medicinal chemistry campaigns aiming to optimize structure-activity relationships. The detailed standardized synthesis steps, including specific molar ratios, solvent choices, and temperature controls, are provided in the guide below to ensure reproducibility and safety.
- Preparation of Benzyl Chloride Intermediates: React substituted benzoic acids with ethanol and sulfuric acid to form esters, convert to hydrazides using hydrazine hydrate, and cyclize with chloroacetyl chloride followed by POCl3 to yield 5-substituted-2-(chloromethyl)-1,3,4-oxadiazoles.
- Modification of Honokiol Core: Nitrate honokiol using concentrated nitric acid in DCM/AcOH at low temperature, then reduce the nitro group to an amine using stannous chloride hydrate in ethanol.
- Final Coupling Reaction: Convert the amino-honokiol to an oxazothione intermediate using carbon disulfide and KOH, then react with the benzyl chloride intermediates in acetone with potassium carbonate to form the final thioether derivatives.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement specialists and supply chain managers, the adoption of this synthetic route offers tangible strategic benefits beyond mere technical novelty. The reliance on commodity chemicals such as benzoic acids, hydrazine hydrate, and carbon disulfide ensures a stable and resilient supply chain, minimizing the risk of raw material shortages that often plague specialized pharmaceutical manufacturing. The avoidance of precious metal catalysts like palladium or platinum eliminates the need for costly metal scavenging steps and reduces the burden of heavy metal residue testing, which directly translates to lower operational expenditures and faster batch release times.
- Cost Reduction in Manufacturing: The synthetic pathway is designed with cost-efficiency in mind, utilizing inexpensive reagents and solvents like ethanol and acetone which are easily recoverable and recyclable. The high yields reported in the patent examples suggest that material throughput is optimized, reducing the cost per gram of the final active intermediate. By eliminating the need for cryogenic conditions or high-pressure equipment, the process lowers capital expenditure requirements for reactor infrastructure, making it accessible for both pilot and commercial scale production facilities.
- Enhanced Supply Chain Reliability: The robustness of the chemical transformations ensures consistent batch-to-batch quality, a critical factor for maintaining long-term supply contracts with pharmaceutical partners. The intermediates generated, such as the chloromethyl oxadiazoles, are stable enough to be stored or transported if necessary, providing flexibility in production scheduling. This stability mitigates the risks associated with just-in-time manufacturing constraints, allowing for the buildup of safety stock without significant degradation concerns.
- Scalability and Environmental Compliance: The process operates largely at atmospheric pressure and moderate temperatures, which simplifies the engineering controls required for scale-up from kilograms to tons. The waste streams generated are primarily aqueous salts and organic solvents that can be treated using standard effluent treatment protocols, aligning with increasingly stringent environmental regulations. This eco-friendly profile enhances the sustainability credentials of the supply chain, a growing priority for multinational corporations seeking green chemistry partners.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the production and application of these honokiol derivatives. These insights are derived directly from the experimental data and structural analysis provided in the patent documentation, offering clarity on the feasibility and potential of this technology for industrial adoption.
Q: What is the primary biological activity of these honokiol oxazole derivatives?
A: These derivatives function as potent alpha-glucosidase inhibitors, showing significantly higher inhibitory activity compared to the parent honokiol molecule and the standard drug acarbose in preliminary screenings.
Q: How does the substitution pattern on the phenyl ring affect efficacy?
A: Structure-activity relationship studies indicate that electron-donating groups (such as methyl or methoxy) on the phenyl ring generally enhance alpha-glucosidase inhibitory activity more effectively than electron-withdrawing groups like halogens.
Q: Is the synthesis process scalable for industrial production?
A: Yes, the patented method utilizes common reagents and standard reaction conditions such as reflux and room temperature stirring, avoiding extreme pressures or exotic catalysts, which facilitates straightforward commercial scale-up.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Honokiol Derivative Supplier
At NINGBO INNO PHARMCHEM, we recognize the transformative potential of the honokiol oxazole thioether series in the fight against diabetes. As a premier CDMO partner, we possess the technical expertise to translate this patented laboratory methodology into a robust, GMP-compliant manufacturing process. Our team has extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that your supply needs are met with precision and reliability. We employ stringent purity specifications and operate rigorous QC labs equipped with advanced analytical instrumentation to guarantee that every batch of high-purity honokiol derivatives meets the exacting standards required for clinical trials and commercial drug formulation.
We invite you to collaborate with us to optimize your supply chain for next-generation antidiabetic intermediates. Our technical procurement team is ready to provide a Customized Cost-Saving Analysis tailored to your specific volume requirements and timeline. Contact us today to request specific COA data for our catalog compounds or to discuss route feasibility assessments for your proprietary analogs. Let us help you accelerate your drug development pipeline with our superior manufacturing capabilities and commitment to quality excellence.
Engineering Bottleneck?
Can't scale up this synthesis? Upload your target structure or CAS, and our CDMO team will evaluate the industrial feasibility within 24 hours. Request Evaluation →
