Advanced Buffered Reduction Technology for Commercial Scale-up of High-Purity Tetrahydrofolic Acid
Advanced Buffered Reduction Technology for Commercial Scale-up of High-Purity Tetrahydrofolic Acid
The pharmaceutical and fine chemical industries are constantly seeking robust methodologies for synthesizing critical intermediates with exceptional purity profiles. Patent CN102329318A introduces a groundbreaking preparation method for high-purity tetrahydrofolic acid that addresses longstanding challenges in yield optimization and impurity control. This technology leverages a sophisticated buffer-controlled reduction strategy using sodium borohydride, fundamentally shifting the paradigm from traditional harsh catalytic processes to a milder, more controllable chemical environment. For R&D directors and procurement specialists, this patent represents a significant opportunity to enhance supply chain reliability while drastically simplifying the manufacturing workflow. By maintaining the reaction pH within a precise window of 6-10 through the use of buffer solutions, the process effectively suppresses the hydrolytic degradation pathways that typically plague folic acid reduction, resulting in a product suitable for high-value downstream applications such as the synthesis of levofolinic acid and methyltetrahydrofolate.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the industrial production of tetrahydrofolic acid has been hindered by the severe limitations inherent in catalytic hydrogenation and enzymatic reduction techniques. Conventional catalytic hydrogenation, as documented in earlier patents like US4665176, relies heavily on precious metal catalysts such as platinum-carbon or rhodium-carbon, which not only inflate raw material costs but also introduce significant safety hazards due to the requirement for activation and high-pressure operations. Furthermore, these traditional methods suffer from excessively long reaction cycles, often ranging from 8 to 24 hours, and in some low-temperature scenarios extending up to 74 hours, which severely bottlenecks production throughput. The resulting product from these legacy processes typically exhibits suboptimal purity levels around 85% and yields hovering near 75%, necessitating complex and costly downstream purification steps to meet the stringent quality standards required for pharmaceutical intermediates. Additionally, enzymatic methods, while specific, are often economically unviable for large-scale manufacturing due to the prohibitive cost of cofactors like NADPH and the complexity of regeneration systems.
The Novel Approach
In stark contrast to these cumbersome legacy techniques, the novel approach detailed in the patent utilizes a buffered chemical reduction system that operates under mild, ambient conditions without the need for expensive transition metals. By employing a buffer solution—such as borate, phosphate, or citrate systems—the process maintains a stable pH environment that counteracts the alkalinity introduced by the sodium borohydride reducing agent, thereby preventing the molecular fragmentation that leads to impurities like pterin and PADGA. This method employs a strategic batch-feeding protocol for the reducing agent over a period of 5-8 hours, which ensures a consistent concentration of active reductant without triggering violent exothermic spikes or incomplete conversions. The result is a streamlined synthesis route that achieves impressive yields of 84-90% with purity levels exceeding 98% in optimized examples, all while eliminating the need for high-pressure equipment or precious metal catalysts. This shift not only simplifies the operational complexity but also aligns perfectly with modern green chemistry principles by reducing waste and energy consumption.
Mechanistic Insights into Buffer-Controlled Borohydride Reduction
The core innovation of this synthesis lies in the precise management of the reaction microenvironment to stabilize the labile tetrahydrofolic acid structure during formation. Sodium borohydride is inherently alkaline, and its addition to an unbuffered aqueous solution causes a rapid rise in pH, which triggers the hydrolytic cleavage of the C9-N10 bond in the folic acid molecule. This degradation pathway generates unwanted byproducts such as pterin and p-aminobenzoyl glutamic acid, which are structurally similar and notoriously difficult to separate from the target product. The introduction of a buffer system with a capacity to maintain pH between 6 and 10 acts as a chemical shock absorber, neutralizing the hydroxide ions released during reduction and keeping the reaction medium within the stability zone of the tetrahydrofolate molecule. This mechanistic control is critical for preserving the integrity of the pteridine ring system, ensuring that the reduction proceeds selectively at the 5,6,7,8-positions without compromising the rest of the molecular architecture.
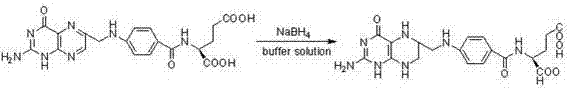
Furthermore, the kinetics of the reduction are optimized through a multi-batch feeding strategy rather than a single bolus addition. In a traditional one-time addition, the initial high concentration of borohydride can lead to localized overheating and rapid gas evolution, while the subsequent decomposition of the reagent in the aqueous phase leaves insufficient reducing power for the final stages of the reaction. By dividing the total amount of sodium borohydride into 3 to 6 batches added at regular intervals over 5-8 hours, the process maintains a steady-state concentration of the active hydride species. This approach ensures that the reduction proceeds to completion even at moderate temperatures of 10-30°C, minimizing the formation of dihydrofolate intermediates that often persist in incomplete reactions. The combination of pH buffering and kinetic control creates a highly reproducible process window that is ideal for scaling from laboratory benchtop to multi-ton commercial production.
How to Synthesize Tetrahydrofolic Acid Efficiently
The implementation of this buffered reduction protocol offers a clear pathway for manufacturing teams to achieve consistent high-quality output with minimal process variability. The procedure begins with the suspension of folic acid in a selected buffer solution under an inert nitrogen atmosphere to prevent oxidative degradation of the sensitive product. Following pH adjustment to neutrality, the reducing agent is introduced in a controlled manner, requiring careful monitoring of temperature and stirring speed to ensure homogeneity. The detailed standardized synthetic steps below outline the specific parameters for reagent ratios, timing, and workup procedures necessary to replicate the high yields reported in the patent data.
- Suspend folic acid in a specific buffer solution (e.g., borate or phosphate) under nitrogen protection and adjust pH to neutral using sodium hydroxide.
- Add sodium borohydride (NaBH4) reducing agent in multiple batches over 5-8 hours while maintaining reaction temperature between 10-30°C and pH between 6-10.
- Adjust the final reaction mixture to pH 3.0-3.5 using hydrochloric acid to precipitate the product, followed by filtration, washing, and vacuum drying.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain directors, the adoption of this buffered reduction technology translates directly into tangible operational efficiencies and risk mitigation strategies. The elimination of precious metal catalysts such as platinum oxide removes a significant variable cost component and eliminates the supply chain risks associated with volatile noble metal markets. Moreover, the ability to conduct the reaction at ambient temperature and pressure reduces the energy footprint of the manufacturing process and allows for the utilization of standard glass-lined or stainless steel reactors without the need for specialized high-pressure containment systems. This simplification of equipment requirements lowers capital expenditure barriers and facilitates faster technology transfer between production sites. The robustness of the buffer system also means that the process is less sensitive to minor fluctuations in raw material quality, enhancing overall supply chain resilience.
- Cost Reduction in Manufacturing: The most immediate financial benefit arises from the complete removal of expensive platinum or rhodium catalysts, which traditionally account for a substantial portion of the raw material budget in catalytic hydrogenation routes. By replacing these with inexpensive and readily available sodium borohydride and common buffer salts, the direct material cost is significantly reduced. Additionally, the shorter reaction time of 5-8 hours compared to the 24-74 hours required by older methods increases reactor turnover rates, allowing for higher production volumes within the same fixed asset base. The high crude purity achieved also minimizes the solvent and labor costs associated with extensive recrystallization or chromatographic purification steps.
- Enhanced Supply Chain Reliability: The reagents utilized in this process, including sodium borohydride, sodium hydroxide, and common buffer components, are commodity chemicals with stable global supply chains, unlike specialized enzymes or activated metal catalysts which may have limited suppliers. This commoditization of inputs ensures that production schedules are not disrupted by niche supply shortages. Furthermore, the mild reaction conditions reduce the wear and tear on processing equipment, leading to lower maintenance downtime and more predictable manufacturing calendars. The process stability also ensures consistent batch-to-batch quality, reducing the risk of rejected lots that can disrupt downstream customer commitments.
- Scalability and Environmental Compliance: From an environmental and safety perspective, this method offers distinct advantages by avoiding the use of high-pressure hydrogen gas, which requires rigorous safety protocols and specialized infrastructure. The aqueous nature of the reaction medium and the absence of heavy metal residues simplify wastewater treatment and effluent management, aiding in compliance with increasingly strict environmental regulations. The process generates minimal hazardous waste, and the high atom economy of the reduction step contributes to a greener manufacturing profile. These factors make the technology highly scalable, capable of being expanded from pilot plant quantities to hundreds of tons annually without encountering the engineering bottlenecks typical of high-pressure catalytic processes.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this high-purity tetrahydrofolic acid synthesis method. These answers are derived directly from the experimental data and technical specifications outlined in the patent documentation to provide clarity on process capabilities and limitations. Understanding these details is essential for technical teams evaluating the feasibility of integrating this route into their existing manufacturing portfolios.
Q: Why is a buffer solution critical in the reduction of folic acid?
A: Sodium borohydride is strongly alkaline. Without a buffer, the pH would spike during addition, causing the folic acid molecule to hydrolyze into impurities like pterin and p-aminobenzoyl glutamic acid (PADGA), drastically lowering purity.
Q: How does this method compare to catalytic hydrogenation?
A: Unlike catalytic hydrogenation which requires expensive platinum catalysts, high pressure, and long reaction times (up to 74 hours), this buffered chemical reduction operates at ambient temperature and pressure without precious metals, achieving yields of 84-90%.
Q: What is the advantage of batch feeding the reducing agent?
A: Batch feeding prevents the initial violent reaction caused by high local concentrations of reducing agent and maintains effective reducing power throughout the process, ensuring complete conversion of folic acid without excessive decomposition.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Tetrahydrofolic Acid Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical importance of high-purity intermediates in the development of next-generation pharmaceutical formulations and nutritional supplements. Our technical team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the transition from laboratory innovation to industrial reality is seamless and efficient. We are committed to delivering products that meet stringent purity specifications through our rigorous QC labs, utilizing advanced analytical methods to verify identity and potency. Our facility is equipped to handle the specific requirements of buffered reduction chemistries, providing a secure and compliant environment for the manufacture of sensitive folate derivatives.
We invite potential partners to engage with our technical procurement team to discuss how this advanced synthesis technology can be tailored to your specific volume and quality requirements. By requesting a Customized Cost-Saving Analysis, you can gain deeper insights into the economic benefits of switching to this catalyst-free process. We encourage you to contact us today to obtain specific COA data and route feasibility assessments that demonstrate our capability to serve as your long-term strategic partner in the global supply of high-value pharmaceutical intermediates.
