Revolutionizing Chiral Alcohol Production: Advanced Biocatalytic Solutions for Pharmaceutical Intermediates
The pharmaceutical industry is currently witnessing a paradigm shift towards greener, more efficient synthetic methodologies, particularly in the production of chiral intermediates which are critical for drug efficacy. A groundbreaking development in this sector is detailed in Chinese patent CN116179502A, which discloses a novel ketoreductase mutant capable of catalyzing the asymmetric reduction of carbonyl compounds with unprecedented efficiency. This technology addresses the longstanding challenges of low selectivity and poor stability associated with wild-type enzymes, offering a robust solution for the synthesis of high-value chiral alcohols. By leveraging rational protein engineering, specifically targeting key amino acid residues such as N113, S141, D149, W152, A197, and S207, this innovation transforms difficult-to-reduce substrates into optically pure products. For R&D directors and procurement specialists seeking reliable pharmaceutical intermediates supplier partnerships, this biocatalytic approach represents a significant leap forward in process chemistry, promising not only superior product quality but also a more sustainable manufacturing footprint that aligns with modern environmental regulations.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Traditional chemical synthesis of chiral alcohols often relies on stoichiometric reducing agents like borohydrides or bulky hydride reagents such as DIP-Cl, which are not only expensive but also generate substantial amounts of hazardous waste. Furthermore, achieving high enantiomeric excess (ee) through chemical means frequently necessitates complex resolution steps or the use of precious metal catalysts, which introduces the risk of heavy metal contamination in the final active pharmaceutical ingredient (API). In the specific context of reducing bulky or sterically hindered ketones, conventional chemical methods often struggle to differentiate between enantiotopic faces of the carbonyl group, leading to racemic mixtures that require costly and yield-loss-inducing separation processes. Additionally, many chemical reduction processes require harsh reaction conditions, including extreme temperatures or anhydrous environments, which increase energy consumption and operational complexity. These limitations create significant bottlenecks in the supply chain for high-purity pharmaceutical intermediates, driving up costs and extending lead times for drug manufacturers who demand stringent purity specifications.
The Novel Approach
In stark contrast, the biocatalytic method described in the patent utilizes a highly engineered ketoreductase that operates under mild, aqueous conditions, typically around 40°C and neutral pH, thereby eliminating the need for hazardous solvents and extreme parameters. The core of this innovation lies in the specific mutation profile, such as the sextuple mutant comprising N113R, S114A, D149F, W152Y, A197M, and S207A, which reshapes the enzyme's active site to perfectly accommodate specific prochiral ketones like the bicyclic substrate shown in Formula I. 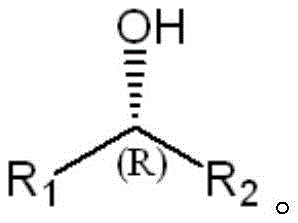 As demonstrated in the patent data, while the wild-type enzyme fails completely to induce chirality (0% ee) on this specific substrate, the engineered mutant achieves an exceptional enantiomeric excess of greater than 99.5% with conversion rates surpassing 99% within just 10 hours. This dramatic improvement in both activity and selectivity allows for a streamlined, one-step synthesis that bypasses the need for downstream purification of racemates, significantly simplifying the overall process flow and enhancing the economic viability of producing complex chiral building blocks.
As demonstrated in the patent data, while the wild-type enzyme fails completely to induce chirality (0% ee) on this specific substrate, the engineered mutant achieves an exceptional enantiomeric excess of greater than 99.5% with conversion rates surpassing 99% within just 10 hours. This dramatic improvement in both activity and selectivity allows for a streamlined, one-step synthesis that bypasses the need for downstream purification of racemates, significantly simplifying the overall process flow and enhancing the economic viability of producing complex chiral building blocks.
Mechanistic Insights into Rational Protein Engineering and Biocatalysis
The success of this ketoreductase mutant is rooted in a sophisticated rational design strategy that combines structural biology with computational modeling to optimize enzyme-substrate interactions. By obtaining the three-dimensional structure of the wild-type enzyme (PDB: 6QHE_A) and performing molecular docking simulations with the target substrate, researchers identified specific amino acid residues lining the binding pocket that were suboptimal for catalysis. Mutations such as W152Y and D149F likely alter the hydrophobicity and steric environment of the active site, facilitating tighter binding and proper orientation of the bulky carbonyl substrate for hydride transfer. The introduction of charged residues like N113R may also stabilize the transition state or improve the interaction with the cofactor NADPH. This precise tuning of the protein architecture ensures that the hydride ion is delivered exclusively to one face of the carbonyl group, resulting in the high stereoselectivity observed. 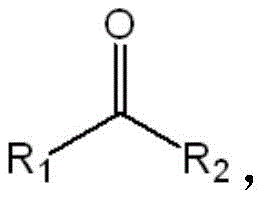 Furthermore, the stability of the mutant is enhanced, allowing it to withstand the rigors of industrial fermentation and catalytic cycles without rapid denaturation, which is a common failure point for many biocatalysts in large-scale applications.
Furthermore, the stability of the mutant is enhanced, allowing it to withstand the rigors of industrial fermentation and catalytic cycles without rapid denaturation, which is a common failure point for many biocatalysts in large-scale applications.
From an impurity control perspective, the enzymatic route offers inherent advantages due to its high chemoselectivity and regioselectivity. Unlike chemical reducers that might indiscriminately reduce other functional groups present in a complex molecule, the ketoreductase mutant is highly specific for the target ketone moiety, minimizing the formation of side products and over-reduced impurities. The use of a whole-cell biocatalyst system, where the enzyme is expressed in E. coli BL21(DE3), further simplifies the process as the cells provide a protective environment for the enzyme and contain endogenous systems that can assist in cofactor recycling. The patent highlights the use of a coupled enzyme system involving glucose dehydrogenase (GDH) to regenerate the consumed NADPH from NADP+ using glucose as a sacrificial donor. This continuous regeneration loop ensures that only catalytic amounts of the expensive cofactor are needed, driving the reaction to completion while maintaining a clean reaction profile that facilitates easier downstream processing and isolation of the high-purity chiral alcohol product.
How to Synthesize High-Purity Chiral Alcohol Efficiently
The implementation of this technology involves a well-defined workflow starting from gene synthesis to final product isolation, designed for seamless integration into existing biomanufacturing facilities. The process begins with the construction of a recombinant expression vector, such as pET-28a, carrying the optimized gene sequence encoding the mutant ketoreductase, which is then transformed into a robust host strain like E. coli BL21(DE3). Following colony selection and verification, the engineered strain is subjected to high-density fermentation to produce the biocatalyst in large quantities. The subsequent biotransformation step is conducted in a buffered aqueous medium where the whole cells act as the catalyst, converting the ketone substrate into the desired chiral alcohol with high efficiency. Detailed standardized synthesis steps are provided in the guide below to ensure reproducibility and optimal performance.
- Construct the recombinant expression vector containing the mutated ketoreductase gene (e.g., SEQ ID NO: 5) into a host like E. coli BL21(DE3).
- Perform high-density fermentation of the engineered strain at controlled temperatures (20-30°C) and dissolved oxygen levels (>20%) to maximize enzyme yield.
- Conduct the biocatalytic reduction of the carbonyl substrate (Formula I) using the whole-cell catalyst in a buffered system with cofactor regeneration (NADP+/GDH) at 40°C.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the adoption of this engineered ketoreductase technology translates into tangible strategic benefits that go beyond mere technical superiority. The shift from chemical reduction to biocatalysis fundamentally alters the cost structure of manufacturing chiral intermediates by removing the dependency on expensive stoichiometric reagents and precious metal catalysts. The elimination of heavy metals from the process not only reduces raw material costs but also significantly lowers the burden on waste treatment facilities, as there is no need for complex metal scavenging steps or the disposal of toxic chemical byproducts. This simplification of the manufacturing process leads to a drastic reduction in operational expenditures and shortens the overall production cycle time, allowing for faster response to market demands. Moreover, the high conversion rates and exceptional selectivity mean that less raw material is wasted on off-spec products, maximizing the yield per batch and improving the overall material efficiency of the supply chain.
- Cost Reduction in Manufacturing: The biocatalytic process eliminates the need for costly chiral resolving agents and stoichiometric reducing agents like DIP-Cl, which are significant cost drivers in traditional synthesis. By utilizing a renewable biocatalyst that can be produced via fermentation, the variable cost per kilogram of the product is substantially lowered. The high atom economy of the enzymatic reaction ensures that the majority of the starting material is converted into the desired product, minimizing waste disposal costs and maximizing resource utilization. Additionally, the mild reaction conditions reduce energy consumption associated with heating, cooling, and pressure control, contributing to further operational savings.
- Enhanced Supply Chain Reliability: Relying on biological fermentation for catalyst production creates a more resilient supply chain compared to sourcing specialized chemical reagents that may be subject to geopolitical or logistical disruptions. The genetically engineered strains can be stored and propagated indefinitely, ensuring a consistent and reliable source of the biocatalyst. The robustness of the mutant enzyme under process conditions reduces the risk of batch failures due to catalyst degradation, thereby guaranteeing consistent delivery schedules. This reliability is crucial for pharmaceutical manufacturers who require uninterrupted supply of critical intermediates to maintain their own production timelines and meet regulatory commitments.
- Scalability and Environmental Compliance: The technology is inherently scalable, having been demonstrated from shake flask levels to high-density fermentation tanks, making it suitable for commercial scale-up of complex pharmaceutical intermediates. The use of aqueous buffers and benign co-substrates like glucose aligns perfectly with green chemistry principles, facilitating easier compliance with increasingly stringent environmental regulations. The absence of volatile organic solvents and toxic heavy metals simplifies the permitting process for new manufacturing lines and reduces the environmental footprint of the facility. This sustainability profile enhances the brand value of the final drug product and meets the growing demand from consumers and regulators for eco-friendly manufacturing practices.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this ketoreductase technology, providing clarity for stakeholders evaluating its potential for their specific applications. These insights are derived directly from the experimental data and embodiments disclosed in the patent documentation, ensuring accuracy and relevance for decision-makers. Understanding these nuances is essential for assessing the feasibility of integrating this biocatalytic route into existing production workflows.
Q: How does the engineered ketoreductase improve upon wild-type enzymes?
A: The engineered mutant features specific amino acid substitutions (N113R, S114A, D149F, W152Y, A197M, S207A) that drastically enhance catalytic activity and stereoselectivity. While the wild-type enzyme shows 0% ee value on specific substrates like Formula I, the mutant achieves >99.5% ee with conversion rates exceeding 99%.
Q: What are the scalability advantages of this biocatalytic process?
A: The process utilizes standard E. coli fermentation technology (e.g., BL21(DE3) host), which is highly scalable from shake flasks to industrial fermenters. The enzyme operates under mild conditions (40°C, aqueous buffer), eliminating the need for cryogenic temperatures or high-pressure equipment often required in chemical reduction.
Q: Is cofactor regeneration required for this reaction?
A: Yes, the reaction requires NADPH or NADH. The patent describes an efficient in-situ regeneration system using glucose dehydrogenase (GDH) and glucose, or alternatively formate dehydrogenase, ensuring the costly cofactor is recycled continuously throughout the reaction cycle.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Chiral Alcohol Supplier
At NINGBO INNO PHARMCHEM, we recognize the transformative potential of advanced biocatalysis in the synthesis of high-value pharmaceutical intermediates. Our team of experts possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that innovative laboratory discoveries like the ketoreductase mutants described in CN116179502A can be successfully translated into robust industrial processes. We are committed to delivering products with stringent purity specifications, supported by our rigorous QC labs that employ state-of-the-art analytical techniques to verify identity and optical purity. Our capability to handle complex enzymatic transformations positions us as a strategic partner for companies seeking to optimize their supply chains for chiral alcohols and other fine chemicals.
We invite you to collaborate with us to explore how this technology can be tailored to your specific synthesis needs. Please contact our technical procurement team to request a Customized Cost-Saving Analysis that evaluates the economic impact of switching to this biocatalytic route for your target molecules. We are ready to provide specific COA data and route feasibility assessments to demonstrate how we can support your goals for cost reduction in pharmaceutical intermediates manufacturing while ensuring the highest standards of quality and supply continuity.
