Advanced Iodine-Catalyzed Synthesis of Substituted Pyrazoles for Industrial Scale-Up
Advanced Iodine-Catalyzed Synthesis of Substituted Pyrazoles for Industrial Scale-Up
The landscape of heterocyclic chemistry is constantly evolving, driven by the need for more sustainable and economically viable synthetic routes for high-value intermediates. A pivotal advancement in this domain is detailed in Chinese Patent CN1261351A, which discloses a robust method for producing substituted pyrazoles through the condensation of carbonyl compounds with hydrazine in the presence of catalytic iodine and sulfuric acid. This technology addresses critical bottlenecks in the manufacturing of pharmaceutical and agrochemical intermediates by replacing hazardous stoichiometric oxidants with a catalytic system that leverages sulfuric acid as a multifunctional reagent. For R&D directors and process chemists, this represents a significant opportunity to streamline synthesis workflows while adhering to stricter environmental compliance standards. The versatility of this approach allows for the generation of diverse pyrazole scaffolds, which are essential building blocks in modern drug discovery and crop protection formulations.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of pyrazole derivatives has relied on a variety of oxidative dehydrogenation strategies that present substantial industrial challenges. Prior art, such as that described in German patents DE-A3035395 and DE-A3029160, utilizes chlorine, hypochlorites, sulfur, or selenium to effect the oxidation of 2-pyrazolines. These conventional pathways are fraught with difficulties, including the generation of highly toxic and corrosive byproducts like hydrogen sulfide and selenides, which complicate downstream purification and waste management. Furthermore, other methods involving aqueous hydrogen peroxide or thermal dehydrogenation on precious metal catalysts like palladium or platinum often suffer from low atom economy and high operational costs due to the requirement for expensive reagents and complex multi-step sequences. The reliance on such mordant oxidants not only poses safety risks to plant personnel but also creates significant liability regarding environmental discharge, making these traditional routes increasingly untenable for modern large-scale manufacturing facilities seeking green chemistry certifications.
The Novel Approach
In stark contrast to these legacy methods, the novel approach outlined in the patent introduces a streamlined one-pot synthesis that eliminates the need for stoichiometric amounts of hazardous external oxidants. By utilizing sulfuric acid not merely as a solvent but as a condensing agent and oxygen source, coupled with a catalytic amount of iodine or iodide salts, the process achieves efficient aromatization of the pyrazoline intermediate. This methodology drastically simplifies the reaction setup, allowing for the direct conversion of readily available carbonyl precursors and hydrazine into high-purity pyrazole derivatives. The elimination of heavy metal catalysts and toxic sulfur or selenium reagents translates directly into a cleaner reaction profile, reducing the burden on effluent treatment plants and lowering the overall cost of goods sold. This shift towards a catalytic, acid-mediated process exemplifies a strategic move towards more sustainable industrial chemistry, offering a reliable pathway for the cost reduction in pharmaceutical intermediates manufacturing without compromising on yield or product quality.
Mechanistic Insights into Iodine-Catalyzed Cyclization in Sulfuric Acid
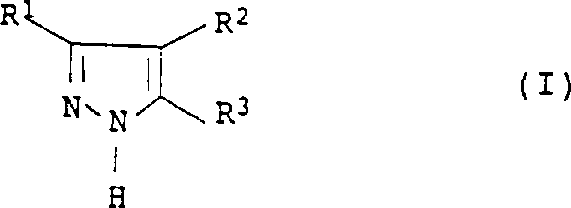
The core of this technological breakthrough lies in the unique interplay between the carbonyl substrate, hydrazine, and the iodine-sulfuric acid catalytic system. The reaction initiates with the condensation of a carbonyl compound of structural formula II with hydrazine or its hydrate to form a hydrazone intermediate, which subsequently undergoes cyclization. The presence of iodine or iodide ions facilitates the oxidative dehydrogenation step, likely through the formation of transient iodo-species that promote the removal of hydrogen atoms to establish the aromatic pyrazole ring. Crucially, the sulfuric acid medium serves a dual purpose: it provides the acidic environment necessary for imine formation and cyclization while simultaneously acting as the terminal oxidant, being reduced to sulfur dioxide in the process. This mechanistic pathway avoids the accumulation of halogenated organic byproducts often seen with chlorine-based oxidants, thereby enhancing the purity profile of the crude reaction mixture and simplifying the isolation of the final active pharmaceutical ingredient (API) intermediate.
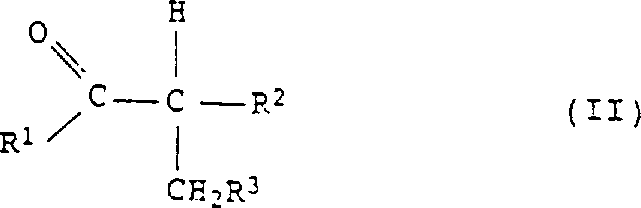
From an impurity control perspective, this mechanism offers distinct advantages over thermal dehydrogenation or metal-catalyzed routes. The use of sulfuric acid ensures that basic impurities and unreacted hydrazine are effectively protonated and retained in the aqueous phase during workup, facilitating a clean separation of the organic pyrazole product. Furthermore, the catalytic nature of the iodine species means that residual metal contamination, a critical concern for regulatory compliance in drug substance manufacturing, is virtually non-existent compared to palladium or platinum-catalyzed processes. The reaction conditions, typically maintained between 80°C and 200°C, are sufficiently vigorous to drive the equilibrium towards the aromatic product while avoiding the extreme thermal stress that can lead to polymerization or charring of sensitive organic substrates. This balance of reactivity and selectivity ensures that the resulting pyrazole derivatives, such as 3,4-dimethylpyrazole or 3-phenyl-4-methylpyrazole, are obtained with high structural integrity and minimal side-chain degradation.
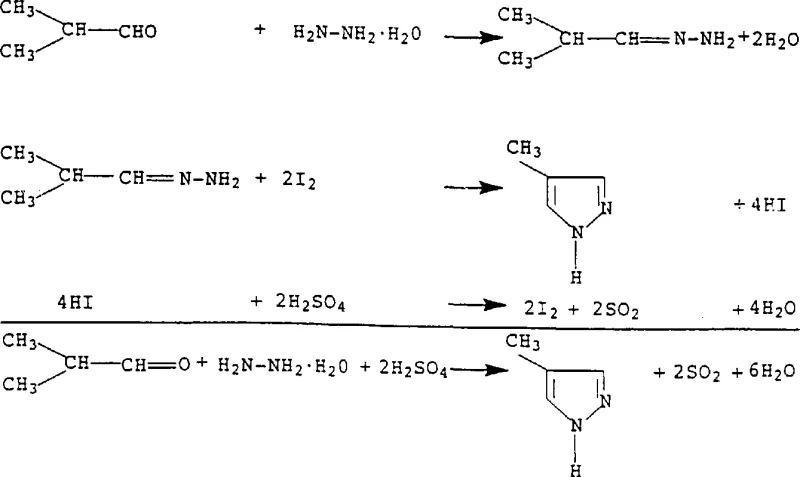
How to Synthesize 3,4-Dimethylpyrazole Efficiently
The practical implementation of this synthesis route is demonstrated through specific embodiments that highlight its operational simplicity and robustness. The process generally involves preparing a suspension of sulfuric acid, hydrazine hydrate, and a catalytic quantity of sodium iodide, followed by the controlled addition of the ketone or aldehyde precursor. As the reaction proceeds, water is distilled off to shift the equilibrium and raise the temperature, driving the cyclization to completion. The detailed standardized synthetic steps see the guide below, which outlines the precise molar ratios, temperature gradients, and workup procedures required to achieve yields exceeding 80% with purities greater than 99% after distillation. This level of efficiency makes the process highly attractive for commercial scale-up of complex pharmaceutical intermediates where consistency and throughput are paramount.
- Prepare a reaction suspension by mixing concentrated sulfuric acid (30-100% weight), hydrazine hydrate, and a catalytic amount of sodium iodide or elemental iodine in a reaction vessel.
- Add the carbonyl compound (such as methyl isopropyl ketone or isobutyraldehyde) dropwise to the suspension while maintaining a temperature between 110°C and 130°C.
- Distill off water to raise the temperature to 130-140°C, stir to complete the reaction, then neutralize with sodium hydroxide and extract the product using isopropylcarbinol.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the adoption of this iodine-catalyzed technology presents a compelling value proposition centered on cost stability and operational reliability. By shifting away from volatile and expensive oxidants like selenium or precious metals, manufacturers can insulate their production costs from the fluctuations of the specialty chemicals market. The use of commodity chemicals such as sulfuric acid and sodium iodide ensures a stable supply chain, reducing the risk of production stoppages due to raw material shortages. Furthermore, the simplified workup procedure, which often requires only neutralization and extraction followed by distillation, significantly reduces the consumption of solvents and energy compared to multi-step purification protocols associated with older technologies. These factors combine to create a manufacturing process that is not only economically superior but also more resilient to external market pressures, ensuring a steady flow of high-quality intermediates to downstream customers.
- Cost Reduction in Manufacturing: The elimination of stoichiometric amounts of expensive and hazardous oxidants such as selenium, chlorine, or hydrogen peroxide leads to a drastic reduction in raw material expenditures. Additionally, the avoidance of precious metal catalysts like palladium or platinum removes the need for costly metal scavenging and recovery steps, further lowering the operational overhead. The ability to use sulfuric acid as a recyclable medium and the recovery of iodine species from the distillate contribute to a circular economy within the plant, maximizing resource utilization and minimizing waste disposal fees associated with toxic byproducts.
- Enhanced Supply Chain Reliability: The reliance on widely available bulk chemicals like sulfuric acid, hydrazine hydrate, and common carbonyl compounds ensures that the supply chain is robust and less susceptible to geopolitical disruptions or niche supplier bottlenecks. Unlike specialized reagents that may have long lead times or single-source dependencies, the inputs for this process are commoditized, allowing for flexible sourcing strategies and better negotiation leverage with vendors. This stability is crucial for maintaining continuous production schedules and meeting the just-in-time delivery requirements of major pharmaceutical and agrochemical clients who demand unwavering supply continuity.
- Scalability and Environmental Compliance: The process is inherently scalable, capable of being run in batch, semi-batch, or continuous modes under atmospheric or slightly pressurized conditions, facilitating easy transition from pilot plant to full commercial production. From an environmental standpoint, the absence of heavy metal waste and toxic selenium or sulfur byproducts simplifies regulatory compliance and reduces the carbon footprint of the manufacturing site. The generation of sulfur dioxide as a byproduct can be managed through established scrubbing technologies to regenerate sulfuric acid, closing the loop on reagent usage and aligning with global sustainability goals for green chemical manufacturing.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this patented pyrazole synthesis technology. These insights are derived directly from the experimental data and claims presented in the patent documentation, providing a clear understanding of the process capabilities and limitations for potential adopters. Understanding these nuances is essential for evaluating the feasibility of integrating this route into existing production lines or new facility designs.
Q: What are the primary advantages of using iodine catalysts over traditional oxidants in pyrazole synthesis?
A: Traditional methods often rely on hazardous oxidants like chlorine, selenium, or hydrogen peroxide, which generate toxic byproducts such as hydrogen sulfide and selenides. The iodine-catalyzed method described in CN1261351A utilizes sulfuric acid as both solvent and oxidant, significantly reducing environmental hazards and simplifying waste treatment protocols.
Q: What represents the optimal temperature range for this cyclization reaction?
A: The reaction is typically conducted at temperatures ranging from 50°C to 250°C, with a preferred operational window between 80°C and 200°C. Specifically, examples demonstrate optimal yields when the mixture is heated to approximately 110°C to 135°C during the addition and dehydration phases.
Q: Can this process be adapted for continuous flow manufacturing?
A: Yes, the patent explicitly states that the method can be carried out continuously or intermittently under normal atmosphere, pressurization, or weak decompression. This flexibility makes it highly suitable for scaling from batch processing to continuous flow reactors for large-volume production.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Substituted Pyrazoles Supplier
At NINGBO INNO PHARMCHEM, we recognize the transformative potential of the iodine-catalyzed pyrazole synthesis route in delivering high-value intermediates to the global market. As a leading CDMO partner, we possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the theoretical benefits of this patent are fully realized in a GMP-compliant environment. Our state-of-the-art facilities are equipped with rigorous QC labs and stringent purity specifications, guaranteeing that every batch of substituted pyrazoles meets the exacting standards required for pharmaceutical and agrochemical applications. We are committed to leveraging this advanced chemistry to provide our partners with a competitive edge through superior quality and consistent supply.
We invite you to engage with our technical procurement team to discuss how this innovative synthesis method can be tailored to your specific project needs. By requesting a Customized Cost-Saving Analysis, you can gain a deeper understanding of the economic benefits specific to your volume requirements. We encourage you to reach out today to obtain specific COA data and route feasibility assessments, allowing us to demonstrate our capability as your trusted partner in bringing next-generation heterocyclic intermediates from the laboratory to the marketplace efficiently and sustainably.
