Advanced Base-Catalyzed Synthesis of Electron-Deficient Multi-Substituted Pyrazole Derivatives for Commercial Scale
The pharmaceutical and fine chemical industries are constantly seeking robust methodologies to construct nitrogen-containing heterocycles, particularly pyrazoles, which serve as privileged scaffolds in numerous bioactive molecules. Patent CN102766095A introduces a significant technological advancement in this domain by disclosing a novel preparation method for multi-substituted pyrazole derivatives containing electron-deficient groups. This innovation addresses long-standing challenges in heterocyclic synthesis by utilizing electron-deficient conjugated enynes and hydrazines as primary building blocks. The process is catalyzed by common bases in organic solvents under remarkably mild conditions, typically ranging from room temperature to 40°C. This represents a paradigm shift from traditional high-energy pathways, offering a streamlined, one-step route to complex molecular architectures that include sensitive functionalities such as aldehydes and ketones. For R&D directors and process chemists, this patent provides a critical blueprint for accessing diverse compound libraries with high atom economy and exceptional regioselectivity, thereby accelerating the timeline for lead optimization and candidate selection in drug discovery programs.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of multi-substituted pyrazole derivatives has relied heavily on the condensation of 1,3-diketones or their equivalents with hydrazines, or alternatively, through 1,3-dipolar cycloadditions involving diazo compounds or iminonitriles with unsaturated olefins or alkynes. However, these conventional strategies are fraught with significant limitations that hinder their utility in modern process chemistry. A primary concern is the issue of regioselectivity; reactions involving unsymmetrical 1,3-diketones often yield mixtures of isomers that are difficult and costly to separate, drastically reducing overall process efficiency. Furthermore, methods employing diazo compounds pose serious safety hazards due to their potential explosiveness and instability, requiring specialized handling equipment and stringent safety protocols that increase operational costs. Additionally, existing oxidative methods often necessitate high temperatures and excessive amounts of oxidants, such as three equivalents of copper acetate, which not only complicates waste treatment but also limits functional group tolerance, particularly for sensitive carbonyl moieties that may degrade under harsh oxidative conditions.
The Novel Approach
In stark contrast to these legacy methods, the technology disclosed in CN102766095A utilizes a base-catalyzed cyclization of electron-deficient conjugated enynes with hydrazines to achieve superior outcomes. This novel approach operates under mild thermal conditions, typically between 23°C and 40°C, eliminating the need for high-energy inputs or hazardous explosive reagents. The reaction demonstrates exceptional compatibility with a wide array of electron-withdrawing groups, including esters, ketones, aldehydes, amides, and cyano groups, which are often incompatible with traditional oxidative protocols. By avoiding transition metal catalysts in the cyclization step itself, the process simplifies downstream purification and reduces the risk of heavy metal contamination in the final active pharmaceutical ingredient. The ability to selectively synthesize single isomers of multi-substituted pyrazoles in one step significantly enhances the synthetic efficiency, providing a reliable pathway for generating high-purity intermediates essential for rigorous biological evaluation and commercial manufacturing.

Mechanistic Insights into Base-Catalyzed Cyclization of Conjugated Enynes
The core of this innovative synthesis lies in the unique reactivity of electron-deficient conjugated enynes, which serve as versatile electrophilic partners in the cyclization process. The mechanism likely involves the nucleophilic attack of the hydrazine nitrogen on the electron-deficient alkyne or alkene system of the enyne substrate, facilitated by the presence of a base such as triethylamine, DBU, or potassium carbonate. This initial addition triggers a cascade of intramolecular cyclization events that efficiently construct the pyrazole ring system. The presence of the electron-withdrawing group (EWG) is crucial as it activates the conjugated system towards nucleophilic attack while simultaneously stabilizing the intermediate species formed during the reaction coordinate. Unlike metal-catalyzed processes that rely on oxidative addition and reductive elimination cycles, this base-mediated pathway avoids the formation of organometallic intermediates, thereby reducing the complexity of the reaction profile and minimizing side reactions associated with metal coordination. The result is a clean transformation that preserves the integrity of sensitive functional groups attached to the pyrazole core, a feature that is paramount for the synthesis of complex drug candidates.
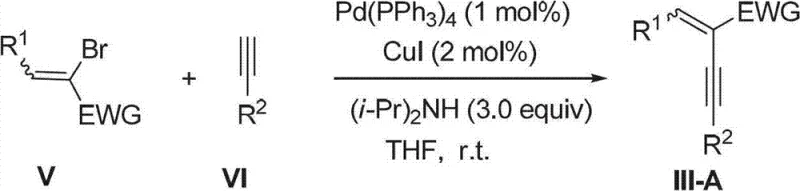
Furthermore, the versatility of this method is underscored by the broad scope of substrates that can be employed, as evidenced by the successful synthesis of various derivatives including fused ring systems. The precursors, specifically the electron-deficient conjugated enynes, can be readily prepared via established Sonogashira coupling reactions between brominated or iodinated unsaturated olefins and terminal alkynes, utilizing palladium and copper catalysts. This modular approach allows for the easy introduction of diverse aryl, heteroaryl, and alkyl substituents at multiple positions on the pyrazole ring. The ability to tune the electronic and steric properties of the starting enynes provides chemists with precise control over the final product's characteristics. This level of control is essential for optimizing the physicochemical properties of pharmaceutical intermediates, ensuring that the resulting compounds meet the stringent requirements for solubility, stability, and bioavailability needed in later stages of drug development.
How to Synthesize Multi-Substituted Pyrazole Derivatives Efficiently
The practical implementation of this synthesis route is designed for ease of operation and scalability, making it highly attractive for both laboratory research and industrial production. The general procedure involves dissolving the electron-deficient conjugated enyne, the chosen hydrazine derivative, and a catalytic amount of base in a suitable organic solvent such as N,N-dimethylformamide (DMF), N,N-dimethylacetamide (DMAc), dimethyl sulfoxide (DMSO), or acetonitrile. The reaction mixture is then stirred at temperatures ranging from room temperature to 40°C, with reaction times typically spanning from 1 to 24 hours depending on the specific substrate reactivity. Progress is conveniently monitored using thin-layer chromatography (TLC), allowing for real-time assessment of conversion. Upon completion, the solvent is removed either directly via rotary evaporation for low-boiling solvents like acetonitrile or after extraction with ether or ethyl acetate for high-boiling solvents like DMAc. The resulting crude product is subsequently purified by column chromatography using standard eluent systems, such as petroleum ether and ethyl acetate mixtures, to afford the pure pyrazole derivative in high yields.
- Dissolve electron-deficient conjugated enyne, hydrazine, and a catalytic amount of base (organic or inorganic) in an organic solvent such as DMF or acetonitrile.
- Stir the reaction mixture at a temperature ranging from room temperature to 40°C for 1 to 24 hours, monitoring progress via TLC.
- Remove the solvent via rotary evaporation or extraction, and purify the crude product using column chromatography to obtain the pure pyrazole derivative.
Commercial Advantages for Procurement and Supply Chain Teams
From a procurement and supply chain perspective, the adoption of this base-catalyzed synthesis method offers substantial strategic benefits that directly impact the bottom line and operational reliability. The reliance on readily available, commodity-grade raw materials such as simple hydrazines, common organic bases, and standard solvents ensures a stable and resilient supply chain that is less susceptible to the volatility often associated with specialized reagents or precious metal catalysts. The elimination of expensive transition metal catalysts in the key cyclization step translates to significant cost reductions in raw material expenditure and simplifies the procurement process by removing the need for sourcing high-purity palladium or copper complexes. Moreover, the mild reaction conditions reduce energy consumption associated with heating and cooling, contributing to lower utility costs and a smaller carbon footprint, which aligns with increasingly stringent environmental regulations and corporate sustainability goals.
- Cost Reduction in Manufacturing: The process achieves cost efficiency through multiple vectors, primarily by utilizing inexpensive base catalysts instead of costly transition metals and by operating at near-ambient temperatures which minimizes energy overhead. The high atom economy of the reaction ensures that a maximum proportion of the starting materials is incorporated into the final product, reducing waste disposal costs and maximizing material utilization. Additionally, the simplified workup procedure, which often avoids complex aqueous quenching or extensive washing steps required for metal removal, lowers labor and processing time costs. These factors combined create a highly economical manufacturing route that enhances profit margins for high-volume production of pharmaceutical intermediates without compromising on quality or purity standards.
- Enhanced Supply Chain Reliability: The robustness of this synthetic route significantly de-risks the supply chain by relying on chemically stable precursors that have long shelf lives and are sourced from a broad global supplier base. Unlike methods dependent on unstable diazo compounds or sensitive organometallic reagents that require cold chain logistics and special handling, the reagents used here are stable solids or liquids that can be stored and transported under standard conditions. This stability ensures consistent production schedules and reduces the likelihood of batch failures due to reagent degradation. For supply chain managers, this translates to predictable lead times and the ability to maintain optimal inventory levels, ensuring continuous availability of critical intermediates for downstream API synthesis and preventing costly production stoppages.
- Scalability and Environmental Compliance: The simplicity of the reaction setup and the absence of hazardous explosive intermediates make this process inherently safer and easier to scale from gram-scale laboratory experiments to multi-ton commercial production. The use of common organic solvents facilitates straightforward solvent recovery and recycling programs, further enhancing the environmental profile of the manufacturing process. The high selectivity of the reaction minimizes the formation of by-products, reducing the burden on waste treatment facilities and ensuring compliance with strict environmental discharge regulations. This scalability and environmental compatibility position the technology as a sustainable choice for long-term manufacturing partnerships, supporting the growing demand for green chemistry solutions in the fine chemical and pharmaceutical sectors.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this pyrazole synthesis technology. These insights are derived directly from the experimental data and scope defined within the patent documentation, providing clarity on the method's capabilities and limitations for potential adopters. Understanding these details is crucial for process engineers and project managers evaluating the feasibility of integrating this route into existing production workflows or new drug development pipelines.
Q: What are the key advantages of this pyrazole synthesis method over traditional 1,3-diketone condensation?
A: Unlike traditional methods that often suffer from poor regioselectivity and require harsh conditions, this base-catalyzed approach using conjugated enynes offers high selectivity for single isomers, operates under mild temperatures (room temp to 40°C), and tolerates sensitive functional groups like aldehydes and ketones which are difficult to preserve in other routes.
Q: Can this method be scaled for industrial production of pharmaceutical intermediates?
A: Yes, the process utilizes commercially available raw materials, simple organic solvents, and common bases without requiring expensive transition metal catalysts for the cyclization step itself. The simple workup procedure involving solvent removal and column chromatography makes it highly suitable for scale-up in fine chemical manufacturing.
Q: What types of electron-deficient groups are compatible with this reaction?
A: The method demonstrates broad compatibility with various electron-withdrawing groups (EWG), including ester groups, ketone carbonyls, aldehyde groups, amide groups, cyano groups, sulfone groups, and ketoester groups, allowing for diverse structural modifications essential for drug discovery.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Multi-Substituted Pyrazole Derivatives Supplier
As the global demand for complex heterocyclic intermediates continues to rise, partnering with an experienced CDMO like NINGBO INNO PHARMCHEM ensures access to cutting-edge synthetic technologies and reliable supply. We possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, leveraging our deep expertise in process optimization to deliver high-purity intermediates that meet rigorous quality specifications. Our state-of-the-art rigorous QC labs employ advanced analytical techniques to verify the structural integrity and purity of every batch, ensuring that the multi-substituted pyrazole derivatives we supply are perfectly suited for your critical drug discovery and development needs. We are committed to supporting our clients through every stage of the product lifecycle, from initial route scouting to full-scale commercial manufacturing.
We invite you to engage with our technical procurement team to discuss how this innovative base-catalyzed synthesis can be tailored to your specific project requirements. By requesting a Customized Cost-Saving Analysis, you can gain a detailed understanding of the economic benefits of switching to this efficient route. We encourage you to contact us today to obtain specific COA data for our available pyrazole derivatives and to receive comprehensive route feasibility assessments that will help you accelerate your development timelines and secure a competitive advantage in the marketplace.
