Advanced Manufacturing of Pitavastatin Calcium: A Technical Breakthrough for Global Supply Chains
Advanced Manufacturing of Pitavastatin Calcium: A Technical Breakthrough for Global Supply Chains
The pharmaceutical industry continuously seeks robust, scalable, and cost-effective methodologies for producing high-value active pharmaceutical ingredients (APIs) and their intermediates. Patent CN101219991A introduces a significant advancement in the synthesis of Pitavastatin Calcium, a potent third-generation HMG-CoA reductase inhibitor used for treating hypercholesterolemia. This technical disclosure outlines a streamlined four-step process that begins with an achiral keto-ester precursor and culminates in the high-purity calcium salt. By leveraging a novel combination of stereoselective reduction and chiral resolution via salt crystallization, this method addresses critical bottlenecks found in earlier synthetic routes. For R&D directors and procurement specialists, understanding this pathway offers insights into achieving superior impurity profiles and enhanced supply chain stability for this crucial cardiovascular therapeutic.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of Pitavastatin Calcium has been plagued by significant technical and safety challenges that hinder efficient commercial scale-up. Early literature, such as reports in Bull. Chem. Soc. Jpn., described routes initiating from chiral epoxides that required the use of highly toxic potassium cyanide for ring-opening reactions. This not only imposes severe safety regulations and waste disposal costs but also complicates the operational environment for manufacturing teams. Furthermore, other documented approaches, including those cited in Tetrahedron Asymmetry, relied heavily on noble metal reagents for carbon chain elongation and chiral center introduction. These methods often necessitated the installation and subsequent removal of protecting groups like phenylthio moieties to control geometric configuration, resulting in excessive step counts, cumbersome operations, and inflated raw material costs. Additionally, prior art involving non-selective reductions often yielded mixtures of four chiral isomers, creating a nightmare for downstream purification and drastically reducing overall process yield.
The Novel Approach
In stark contrast, the methodology disclosed in CN101219991A represents a paradigm shift towards simplicity and efficiency. This novel approach bypasses the need for pre-existing chiral centers in the starting material, instead utilizing a common achiral keto-ester intermediate. The core innovation lies in a highly selective reduction step followed by a sophisticated resolution strategy. Rather than attempting to prevent the formation of unwanted isomers through expensive chiral auxiliaries, the process allows for the generation of a specific pair of diastereomers which are then easily separated. By reacting the crude acid mixture with D-(+) benzylmethylamine, the process forms salt complexes that exhibit distinct solubility characteristics in specific solvent systems like methyl isobutyl ketone mixed with acetone or ethanol. This crystallization-driven purification effectively isolates the desired (3R,5S) isomer with high optical purity, eliminating the need for complex chromatography and significantly reducing the environmental footprint associated with solvent consumption and hazardous reagent usage.
Mechanistic Insights into Stereoselective Reduction and Chiral Resolution
The chemical elegance of this synthesis is anchored in the precise control of stereochemistry during the reduction phase. The process initiates with the reduction of the 3-keto group in the heptenoic acid ester precursor. Unlike standard borohydride reductions which might yield a random mixture of alcohols, this method employs a specialized reducing system comprising a boron compound, such as diethylmethoxyborane or triethylborane, in conjunction with a borohydride like sodium borohydride. This reaction is conducted under rigorous cryogenic conditions, typically maintained between -50°C and -100°C under a nitrogen atmosphere. These low temperatures are critical for kinetically controlling the hydride delivery, ensuring that the reduction predominantly yields the (3S,5R) and (3R,5S) diastereomers, which collectively account for approximately 98.2% of the product mixture, while minimizing the formation of the undesired (3S,5S) and (3R,5R) isomers to merely 1.8%.
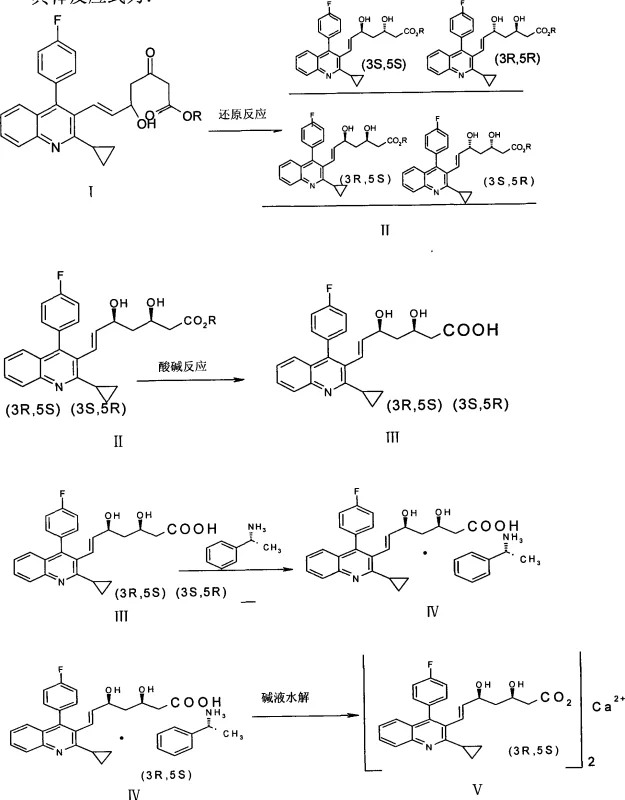
Following the reduction and subsequent hydrolysis to the free acid, the mechanism shifts to thermodynamic control during the resolution phase. The addition of D-(+) benzylmethylamine creates a diastereomeric salt mixture. The success of this separation relies on the subtle differences in lattice energy and solvation of the (3R,5S)-amine salt versus the (3S,5R)-amine salt in the chosen separation medium. The patent specifies the use of methyl isobutyl ketone (MIBK) blended with acetone or ethanol in volume ratios ranging from 1:0.5 to 1:1.5. In this environment, the target (3R,5S) isomer exhibits significantly lower solubility, prompting it to crystallize out of the solution as a white powder, while the unwanted isomer remains dissolved in the mother liquor. This physical separation technique is far more robust and scalable than chiral HPLC, providing a reliable mechanism for achieving the stringent enantiomeric excess required for pharmaceutical grade Pitavastatin Calcium.
How to Synthesize Pitavastatin Calcium Efficiently
Implementing this synthesis route requires careful attention to reaction parameters, particularly temperature control and solvent selection during the crystallization steps. The process is divided into four distinct operational stages: selective reduction, ester hydrolysis, chiral resolution via salt formation, and final calcium salt precipitation. Each stage is designed to maximize yield while maintaining high purity standards. The initial reduction sets the stereochemical foundation, while the resolution step acts as the critical purification gate. Operators must ensure that the cryogenic reduction is performed under inert gas protection to prevent moisture interference, and the crystallization solvents must be strictly proportioned to optimize the recovery of the target isomer. The detailed standardized synthesis steps for this process are outlined in the guide below.
- Perform stereoselective reduction of the keto-ester precursor using a boron compound and borohydride combination at cryogenic temperatures (-50°C to -100°C) to generate the dihydroxy ester intermediate.
- Hydrolyze the ester intermediate using alkali followed by acidification to obtain the free carboxylic acid form of the heptenoic acid derivative.
- React the acid with D-(+) benzylmethylamine to form a salt mixture, then utilize differential solubility in methyl isobutyl ketone and acetone/ethanol to crystallize and isolate the desired (3R,5S) isomer.
- Hydrolyze the isolated amine salt with aqueous sodium hydroxide and treat with calcium chloride solution to precipitate the final Pitavastatin Calcium product.
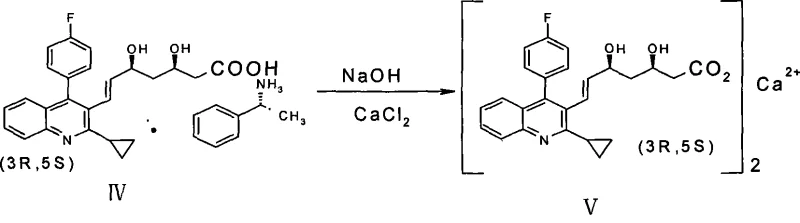
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the transition to this patented synthesis route offers substantial strategic benefits beyond mere technical feasibility. The primary advantage lies in the drastic simplification of the raw material supply chain. By eliminating the dependency on scarce, expensive, or hazardous chiral starting materials—such as the (3R)-3-alkylsilyloxy precursors mentioned in competing patents—manufacturers can source bulk chemicals from a wider array of suppliers. This diversification mitigates the risk of supply disruptions and reduces the leverage of single-source vendors, leading to more stable pricing structures. Furthermore, the removal of toxic reagents like potassium cyanide from the process flow significantly lowers the regulatory burden and insurance costs associated with hazardous material handling, translating into direct operational expenditure savings.
- Cost Reduction in Manufacturing: The economic impact of this process is driven by the elimination of costly purification steps and expensive reagents. Traditional routes often require multiple protection and de-protection cycles or the use of precious metal catalysts, both of which add significant cost per kilogram. In this novel method, the reliance on common boron reagents and the use of crystallization rather than chromatography for purification drastically reduces solvent consumption and processing time. The high conversion rate and the ability to recover the desired isomer efficiently mean that less raw material is wasted, optimizing the overall material balance and driving down the cost of goods sold (COGS) for the final API intermediate.
- Enhanced Supply Chain Reliability: Supply continuity is paramount for long-term pharmaceutical contracts. This synthesis route enhances reliability by utilizing commodity chemicals that are readily available in the global market. Solvents such as methanol, tetrahydrofuran, ethyl acetate, and methyl isobutyl ketone are produced at massive scales worldwide, ensuring that production is never halted due to niche reagent shortages. Additionally, the robustness of the crystallization step means that the process is less sensitive to minor fluctuations in reaction conditions compared to enzymatic or highly sensitive catalytic methods, ensuring consistent batch-to-batch quality and reliable delivery schedules for downstream API manufacturers.
- Scalability and Environmental Compliance: Scaling chemical processes from the laboratory to multi-ton production often reveals hidden inefficiencies, but this method is inherently designed for scale. The unit operations involved—reduction, extraction, crystallization, and filtration—are standard in the fine chemical industry and can be easily replicated in large reactors. From an environmental perspective, the avoidance of heavy metals and cyanides simplifies waste treatment protocols. The aqueous workups and standard organic solvents allow for easier recycling and disposal compliance, aligning with increasingly stringent global environmental regulations and supporting the sustainability goals of modern pharmaceutical supply chains.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this Pitavastatin Calcium synthesis method. These answers are derived directly from the technical specifications and beneficial effects described in the patent documentation, providing clarity on process capabilities and quality assurance measures. Understanding these details is essential for technical teams evaluating the feasibility of adopting this route for commercial production.
Q: How does this method improve upon conventional Pitavastatin synthesis routes?
A: Unlike conventional methods that rely on toxic reagents like potassium cyanide or expensive chiral starting materials with low yields, this patented process utilizes readily available achiral precursors. It achieves high optical purity through a robust resolution step using D-(+) benzylmethylamine, significantly simplifying the purification workflow and eliminating the need for hazardous cyanide handling.
Q: What is the key mechanism for ensuring high stereochemical purity?
A: The process employs a specific reducing agent system, typically a combination of a boron compound (such as diethylmethoxyborane) and a borohydride, at controlled low temperatures. This selectively generates a pair of isomers ((3S,5R) and (3R,5S)) accounting for over 98% of the product. Subsequent salt formation with D-(+) benzylmethylamine exploits solubility differences in mixed solvents to effectively separate the target (3R,5S) isomer from its counterpart.
Q: Is this synthesis route suitable for large-scale commercial production?
A: Yes, the method is designed for scalability. It avoids complex chromatographic separations and uses standard industrial solvents like methyl isobutyl ketone, ethanol, and acetone. The crystallization-based purification is inherently scalable, and the avoidance of precious metal catalysts or highly toxic reagents makes it safer and more cost-effective for multi-ton manufacturing campaigns.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Pitavastatin Calcium Supplier
The technical potential of this synthesis route is immense, offering a clear path to high-quality, cost-efficient production of this vital cardiovascular intermediate. At NINGBO INNO PHARMCHEM, we possess the extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production required to bring this chemistry to life. Our facility is equipped with state-of-the-art cryogenic reactors capable of maintaining the precise -50°C to -100°C temperatures necessary for the stereoselective reduction step, alongside advanced crystallization units for the resolution phase. We adhere to stringent purity specifications and operate rigorous QC labs to ensure that every batch of Pitavastatin Calcium meets the highest international pharmacopoeia standards, guaranteeing consistency and safety for your final drug product.
We invite you to collaborate with us to leverage this advanced manufacturing technology for your supply chain. Our technical procurement team is ready to provide a Customized Cost-Saving Analysis tailored to your specific volume requirements, demonstrating how this route can optimize your budget without compromising quality. Please contact us today to request specific COA data and route feasibility assessments, and let us demonstrate how our expertise in complex pharmaceutical intermediates can become a cornerstone of your sourcing strategy.
