Advanced Ruthenium-Catalyzed Synthesis of Polyaromatic Naphthalenes for Commercial Scale-Up
The pharmaceutical and optoelectronic industries are constantly seeking robust synthetic routes for complex polycyclic aromatic hydrocarbons, specifically polyaromatic substituted naphthalene derivatives, which serve as critical scaffolds for next-generation functional materials. Patent CN108033866B introduces a groundbreaking methodology that leverages ruthenium-catalyzed cyclization of dibenzyl ketone with internal alkynes to access these valuable structures efficiently. This innovation addresses the longstanding challenge of constructing multi-substituted naphthalene cores under mild conditions without the need for harsh oxidants or expensive ligands. The core structural motif, as illustrated in the general formula below, allows for significant diversification, making it a versatile platform for drug discovery and material science applications.
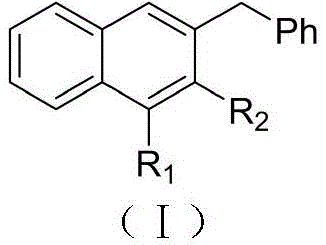
Furthermore, the strategic placement of substituents R1 and R2 enables fine-tuning of electronic properties, which is paramount for designing high-performance organic semiconductors or potent kinase inhibitors. By utilizing relatively inexpensive ruthenium precursors like [RuCl2(p-cymene)]2, this technology democratizes access to high-value intermediates that were previously cost-prohibitive due to complex multi-step syntheses. For R&D directors and procurement managers alike, understanding this patent is key to securing a reliable polyaromatic substituted naphthalene supplier who can deliver high-purity pharmaceutical intermediates with a reduced environmental footprint.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of polyaromatic substituted naphthalene derivatives has relied heavily on transition-metal-catalyzed C-H activation strategies that often demand stoichiometric amounts of oxidants to close the catalytic cycle. Traditional protocols frequently employ copper or silver salts as terminal oxidants, which not only escalate the raw material costs significantly but also generate substantial quantities of heavy metal waste that require complex and expensive disposal procedures. Moreover, many existing methods necessitate the use of specialized ligands or pre-functionalized substrates such as aryl halides or aryl acids, which adds multiple synthetic steps and reduces the overall atom economy of the process. These harsh conditions, often involving high temperatures or strong acids, can lead to poor functional group tolerance, limiting the scope of substrates that can be successfully converted into the desired naphthalene cores. Consequently, the conventional approaches suffer from low scalability and high environmental impact, creating a bottleneck for the commercial scale-up of complex polymer additives or electronic chemicals.
The Novel Approach
In stark contrast, the novel approach detailed in CN108033866B utilizes a ruthenium-catalyzed system that operates effectively without any external oxidants or additives, relying solely on simple inorganic bases to facilitate the transformation. This method activates the beta-hydrogen of dibenzyl ketone to drive the cyclization with internal alkynes, thereby eliminating the need for stoichiometric metal oxidants and drastically simplifying the reaction workup. The process is conducted in common non-polar solvents like toluene at moderate temperatures around 100°C, which enhances safety and energy efficiency compared to high-temperature pyrolysis or cryogenic conditions. By removing the dependency on expensive ligands and oxidants, this technology offers a direct pathway to cost reduction in optoelectronic material manufacturing while maintaining high selectivity for the target naphthalene derivatives. This green chemistry approach aligns perfectly with modern sustainability goals, offering a scientifically reasonable and economically practical solution for large-scale production.
Mechanistic Insights into Ruthenium-Catalyzed Cyclization
The mechanistic elegance of this transformation lies in the ability of the ruthenium catalyst to orchestrate a sequential C-H activation and annulation process without external redox agents. The catalytic cycle initiates with the coordination of the ruthenium species to the carbonyl oxygen of the dibenzyl ketone, followed by the activation of the benzylic C-H bond to form a ruthenacycle intermediate. This key intermediate then undergoes insertion of the internal alkyne, followed by reductive elimination or protonolysis to release the polyaromatic naphthalene product and regenerate the active catalyst species. The absence of external oxidants suggests that the hydrogen atoms removed during the aromatization process are likely scavenged by the base or released as hydrogen gas, a feature that distinguishes this method from oxidative coupling reactions. Understanding this mechanism is crucial for process chemists aiming to optimize reaction parameters for specific substrate classes, ensuring maximum conversion and minimal byproduct formation.
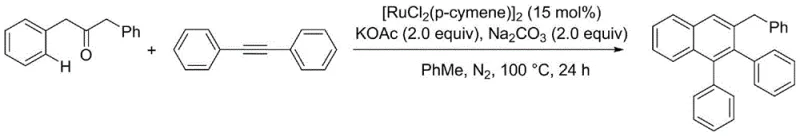
Regarding impurity control, the mild reaction conditions and the specific selectivity of the ruthenium catalyst minimize the formation of oligomeric byproducts or over-oxidized species that often plague traditional Friedel-Crafts type cyclizations. The use of a mixed base system comprising KOAc and Na2CO3 appears to play a dual role in neutralizing acidic byproducts and facilitating the deprotonation steps essential for catalyst turnover. This precise control over the reaction environment ensures that the resulting polyaromatic substituted naphthalenes possess high purity levels, which is a critical requirement for their application in sensitive fields such as organic light-emitting diodes (OLEDs) or as active pharmaceutical ingredients. For instance, the synthesis of 3-benzyl-1,2-diphenylnaphthalene proceeds with a commendable yield of 70%, demonstrating the robustness of the protocol even for sterically demanding substrates.
How to Synthesize 3-Benzyl-1,2-diphenylnaphthalene Efficiently
To implement this synthesis in a laboratory or pilot plant setting, operators must adhere to strict anhydrous conditions and inert atmosphere protocols to prevent catalyst deactivation. The procedure involves charging a sealed reaction vessel with dibenzyl ketone and the chosen internal alkyne in a 2:1 molar ratio, followed by the addition of the ruthenium catalyst and the binary base system in toluene. Detailed standardized synthesis steps are provided in the guide below to ensure reproducibility and safety during the heating and purification phases.
- Charge a sealed tube with dibenzyl ketone (0.2 mmol), internal alkyne (0.1 mmol), and [RuCl2(p-cymene)]2 catalyst (15 mol%).
- Add toluene (0.5 mL) as solvent, followed by dry sodium carbonate (0.2 mmol) and potassium acetate (0.2 mmol) as bases.
- Purge with nitrogen three times, heat to 100°C for 24 hours, and purify the crude mixture via column chromatography using petroleum ether.
Commercial Advantages for Procurement and Supply Chain Teams
From a supply chain perspective, this patented technology offers transformative benefits by simplifying the sourcing of raw materials and reducing the complexity of waste management logistics. The elimination of stoichiometric heavy metal oxidants means that procurement teams no longer need to secure volatile supplies of silver or copper salts, which are subject to market price fluctuations and regulatory scrutiny regarding hazardous material transport. Furthermore, the use of toluene as a solvent and simple inorganic bases ensures that the process relies on commodity chemicals that are readily available globally, enhancing supply chain reliability and reducing the risk of production delays due to material shortages. This robustness is essential for maintaining continuous manufacturing operations, especially when producing high-purity pharmaceutical intermediates that require consistent quality batches.
- Cost Reduction in Manufacturing: The primary driver for cost optimization in this process is the removal of expensive oxidants and ligands, which traditionally account for a significant portion of the bill of materials in C-H activation chemistry. By utilizing a catalytic amount of inexpensive ruthenium and simple bases, the direct material costs are substantially lowered, allowing for more competitive pricing of the final naphthalene derivatives. Additionally, the simplified workup procedure, which avoids complex metal scavenging steps, reduces labor costs and processing time, leading to overall operational efficiency gains that improve the profit margin for manufacturers.
- Enhanced Supply Chain Reliability: The reliance on stable, commercially available starting materials like dibenzyl ketone and various internal alkynes ensures a resilient supply chain that is less susceptible to disruptions caused by specialty chemical shortages. Since the reaction does not require moisture-sensitive or air-sensitive reagents beyond the standard inert atmosphere setup, the logistical burden of storage and transportation is minimized, facilitating smoother inventory management. This stability allows suppliers to offer shorter lead times for high-purity polyaromatic substituted naphthalenes, meeting the urgent demands of downstream clients in the electronics and pharma sectors.
- Scalability and Environmental Compliance: The green nature of this synthesis, characterized by the absence of heavy metal waste and the use of recyclable solvents, aligns perfectly with stringent environmental regulations, reducing the compliance costs associated with waste disposal. The mild reaction temperature of 100°C is easily achievable in standard industrial reactors, ensuring that the process can be scaled from gram to ton quantities without requiring specialized high-pressure or cryogenic equipment. This ease of scale-up supports the commercial production of complex naphthalene derivatives needed for mass-market applications like display technologies and generic drug formulations.
Frequently Asked Questions (FAQ)
The following questions address common technical inquiries regarding the implementation and scope of this ruthenium-catalyzed methodology, derived directly from the experimental data and beneficial effects described in the patent documentation. These insights are intended to clarify the operational parameters and potential applications for technical teams evaluating this route for adoption.
Q: What are the advantages of this Ru-catalyzed method over traditional cyclometallation?
A: Unlike traditional methods requiring stoichiometric heavy metal oxidants like copper or silver salts, this patent utilizes a catalytic amount of inexpensive ruthenium without external oxidants, significantly reducing heavy metal waste and production costs.
Q: What are the optimal reaction conditions for maximizing yield?
A: The patent data indicates that using toluene as the solvent with a mixed base system of KOAc and Na2CO3 at 100°C for 24 hours provides the highest yields, reaching up to 70% for unsubstituted diphenylacetylene substrates.
Q: Can this synthesis be scaled for industrial optoelectronic material production?
A: Yes, the process uses mild conditions (100°C), common solvents (toluene), and avoids sensitive additives, making it highly suitable for commercial scale-up of complex naphthalene derivatives used in OLEDs and semiconductor applications.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Polyaromatic Substituted Naphthalene Derivatives Supplier
At NINGBO INNO PHARMCHEM, we recognize the immense potential of this ruthenium-catalyzed technology to revolutionize the production of high-value intermediates for the global market. As a premier CDMO partner, we possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that your transition from lab-scale discovery to full-scale manufacturing is seamless and efficient. Our state-of-the-art facilities are equipped with rigorous QC labs capable of meeting stringent purity specifications, guaranteeing that every batch of polyaromatic naphthalene derivatives meets the exacting standards required for pharmaceutical and electronic applications.
We invite you to collaborate with our technical procurement team to explore how this innovative synthesis can be tailored to your specific project needs. By requesting a Customized Cost-Saving Analysis, you can gain a clear understanding of the economic benefits of switching to this greener, more efficient route. Contact us today to obtain specific COA data and route feasibility assessments, and let us help you secure a sustainable supply of high-quality chemical intermediates for your future projects.
