Advanced Asymmetric Synthesis of Bicalutamide Enantiomers for Commercial Scale API Production
The pharmaceutical landscape for oncology treatments has long recognized the critical importance of optical purity in drug efficacy, particularly for non-steroidal antiandrogens like Bicalutamide, commercially known as Casodex. Patent CN1409702A presents a groundbreaking methodology for the asymmetric synthesis of Casodex enantiomers and their derivatives, addressing the longstanding economic and technical challenges associated with producing pure (R)-enantiomers. Traditionally, the industry has relied on resolving racemic mixtures or utilizing expensive chiral pool starting materials, which inherently limits scalability and inflates production costs. This patent introduces a novel pathway leveraging (S)-citramalic acid, a readily accessible chiral building block, to construct the critical propionamide backbone with high stereochemical fidelity. By shifting the synthetic paradigm from resolution-based methods to direct asymmetric synthesis, this technology offers a robust framework for manufacturing high-purity pharmaceutical intermediates.
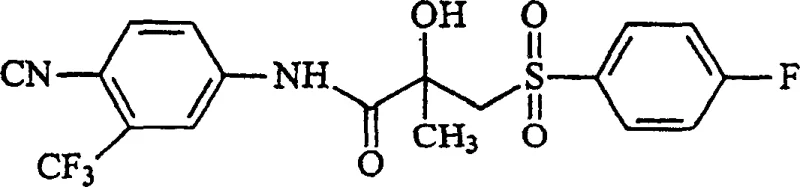
The structural complexity of Bicalutamide, specifically the chiral beta-carbon atom within the propionamide moiety, necessitates precise control during synthesis to ensure therapeutic potency. The patent elucidates that while racemic mixtures are commercially available, the (R)-enantiomer possesses significantly higher antiandrogenic activity, making the exclusion of the less active (S)-enantiomer a priority for modern drug development. The disclosed method achieves this by employing a ring-opening strategy of specific lactone structures, which serves as a versatile platform for introducing various substituents. This approach not only streamlines the synthetic route but also enhances the impurity profile of the final active pharmaceutical ingredient (API), a crucial factor for regulatory approval and patient safety in global markets.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of optically active Bicalutamide has been plagued by reliance on inefficient resolution techniques or prohibitively expensive starting materials. Prior art, such as the methods described by Tucker and later refined by Miller, often utilized (R)-proline as a chiral auxiliary or starting material. While chemically feasible, (R)-proline is not only difficult to procure in bulk quantities but also commands a premium price that drastically impacts the cost of goods sold (COGS). Furthermore, resolution processes inherently suffer from a maximum theoretical yield of 50% for the desired enantiomer, requiring the disposal or recycling of the unwanted isomer, which adds significant waste management burdens and operational complexity. These conventional pathways often involve multiple protection and deprotection steps that lower overall atom economy and increase the risk of racemization during harsh reaction conditions.
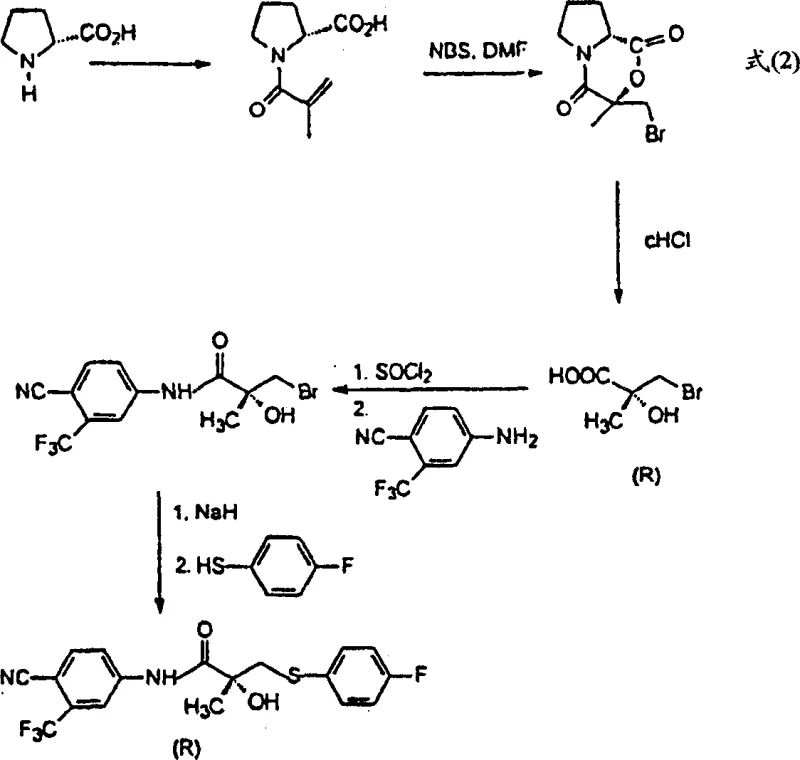
The Novel Approach
In stark contrast, the methodology outlined in CN1409702A circumvents these bottlenecks by utilizing (S)-citramalic acid as the primary chiral source. This abundant and cost-effective raw material allows for the construction of the requisite carbon skeleton without the need for scarce amino acid derivatives. The innovation lies in the conversion of citramalic acid into specific bromolactone intermediates, which then undergo nucleophilic substitution with thiols such as 4-fluorobenzenethiol. This sequence preserves the stereochemical integrity established in the starting material, effectively transferring the chirality from the citramalic acid to the final drug molecule. By avoiding the 50% yield ceiling of resolution and eliminating the dependency on expensive proline, this novel approach provides a direct, linear, and economically superior route to the target enantiomer, fundamentally altering the cost structure of Bicalutamide manufacturing.
Mechanistic Insights into Citramalic Acid Derived Lactone Synthesis
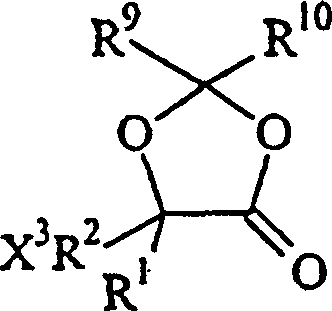
The core mechanistic advantage of this patent lies in the strategic formation and manipulation of the lactone ring system derived from citramalic acid. The process begins with the protection of the hydroxy acid functionality through the formation of a dioxolanone ring using tribromoacetaldehyde. This protection step is not merely defensive; it increases the molecular weight and lipophilicity of the intermediate, facilitating easier separation from polar byproducts in subsequent stages. Following protection, a decarboxylative bromination is performed, typically utilizing reagents like DCC and 2-mercaptopyridine N-oxide, to install the leaving group necessary for nucleophilic attack. This transformation converts the carboxylic acid handle into a reactive bromomethyl group while maintaining the cyclic structure, creating a highly electrophilic center ready for functionalization.
Subsequent nucleophilic substitution by 4-fluorobenzenethiol opens the door to introducing the aryl-thio moiety essential for Bicalutamide's biological activity. The patent highlights that performing this substitution on the lactone prior to hydrolysis offers distinct purification advantages over hydrolyzing first. The resulting sulfide intermediate can then be oxidized to the corresponding sulfone using standard oxidants like mCPBA, completing the assembly of the side chain. Throughout this cascade, the chiral center remains undisturbed, ensuring that the final product retains the high enantiomeric excess (ee) inherent to the starting (S)-citramalic acid. This mechanistic elegance minimizes the formation of diastereomeric impurities, simplifying the downstream purification process and ensuring a consistent, high-quality output suitable for sensitive pharmaceutical applications.
How to Synthesize Bicalutamide Intermediates Efficiently
The practical execution of this synthesis involves a series of well-defined chemical transformations that balance reactivity with selectivity. Operators must carefully control reaction temperatures and stoichiometry, particularly during the decarboxylation and oxidation steps, to prevent side reactions that could compromise optical purity. The protocol emphasizes the use of standard laboratory equipment and reagents, making it highly adaptable for both pilot-scale validation and full commercial production. The detailed procedural steps involve specific workup procedures, such as pH adjustments and solvent extractions, designed to maximize recovery at each stage. For a comprehensive understanding of the operational parameters and safety considerations, please refer to the standardized synthesis guide below.
- Protect (S)-citramalic acid using tribromoacetaldehyde and sulfuric acid to form a dioxolanone derivative.
- Perform decarboxylative bromination using DCC and 2-mercaptopyridine N-oxide to generate the bromo-lactone intermediate.
- React the bromo-lactone with 4-fluorobenzenethiol followed by hydrolysis and oxidation to yield the final sulfone intermediate.
Commercial Advantages for Procurement and Supply Chain Teams
From a strategic procurement perspective, the adoption of this citramalic acid-based route offers profound advantages in terms of cost stability and supply security. The shift away from niche chiral auxiliaries like (R)-proline to commodity chemicals like citramalic acid decouples the production cost from the volatility of the specialty amino acid market. This transition ensures a more predictable pricing model for the final intermediate, allowing procurement managers to negotiate longer-term contracts with greater confidence. Furthermore, the simplified synthetic sequence reduces the number of unit operations required, which directly correlates to lower utility consumption, reduced labor hours, and decreased capital expenditure on specialized processing equipment. These factors collectively contribute to a leaner, more resilient supply chain capable of withstanding market fluctuations.
- Cost Reduction in Manufacturing: The elimination of expensive chiral starting materials and the avoidance of resolution steps significantly lowers the raw material input costs. By utilizing a linear synthesis that theoretically approaches 100% yield of the desired enantiomer rather than being capped at 50%, the process maximizes the value extracted from every kilogram of input material. Additionally, the improved separation characteristics of the protected lactone intermediates reduce the need for extensive chromatographic purification, which is often a major cost driver in fine chemical manufacturing. This efficiency translates into substantial savings in solvent usage and waste disposal fees, further enhancing the overall economic viability of the project.
- Enhanced Supply Chain Reliability: Sourcing (S)-citramalic acid is far less risky than sourcing (R)-proline, as it is produced by multiple global suppliers as a bulk chemical. This diversification of the supply base mitigates the risk of single-source bottlenecks that can halt production lines. The robustness of the chemistry also means that the process is less sensitive to minor variations in reagent quality, ensuring consistent batch-to-batch performance. For supply chain heads, this reliability is paramount in maintaining continuous API production schedules and meeting the rigorous delivery commitments required by downstream pharmaceutical partners.
- Scalability and Environmental Compliance: The synthetic route is designed with scalability in mind, avoiding hazardous reagents or extreme conditions that are difficult to manage on a large scale. The use of standard oxidation and substitution chemistries allows for straightforward scale-up from kilogram to multi-ton batches without significant re-engineering. Moreover, the reduction in waste generation, particularly the avoidance of discarding half the product mass as seen in resolution processes, aligns with modern green chemistry principles. This environmental efficiency not only reduces the carbon footprint of the manufacturing process but also simplifies regulatory compliance regarding effluent treatment and hazardous waste management.
Frequently Asked Questions (FAQ)
Understanding the technical nuances of this synthesis is vital for stakeholders evaluating its implementation. The following questions address common inquiries regarding the chemical feasibility, regulatory implications, and operational requirements of the citramalic acid route. These answers are derived directly from the technical disclosures within the patent documentation, providing a factual basis for decision-making. For further clarification on specific process parameters or customization options, our technical team is available to provide detailed assessments.
Q: Why is (S)-citramalic acid preferred over (R)-proline for Bicalutamide synthesis?
A: (S)-citramalic acid is a readily available and inexpensive commodity chemical, whereas (R)-proline is difficult to source and significantly more expensive, impacting the overall cost of goods.
Q: What is the key advantage of the lactone intermediate strategy described in CN1409702A?
A: The lactone intermediate allows for easier separation and purification of the reaction products compared to open-chain acid derivatives, leading to higher overall purity and yield.
Q: Can this process be scaled for commercial API manufacturing?
A: Yes, the process utilizes standard reagents and avoids complex chiral catalysts or resolution steps that limit scale, making it highly suitable for multi-kilogram to ton-scale production.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Bicalutamide Intermediate Supplier
At NINGBO INNO PHARMCHEM, we recognize the transformative potential of the asymmetric synthesis technologies described in CN1409702A for the global oncology market. As a premier CDMO partner, we possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that innovative laboratory methods are successfully translated into robust industrial processes. Our facility is equipped with stringent purity specifications and rigorous QC labs capable of verifying the enantiomeric excess and chemical purity required for FDA and EMA submissions. We are committed to delivering high-purity Bicalutamide intermediates that meet the exacting standards of the pharmaceutical industry, leveraging our expertise to optimize yield and minimize impurities.
We invite forward-thinking pharmaceutical companies to collaborate with us to leverage this cost-effective synthesis route for their next-generation antiandrogen therapies. By partnering with our technical procurement team, you can request a Customized Cost-Saving Analysis tailored to your specific volume requirements and quality targets. We encourage you to reach out today to obtain specific COA data and route feasibility assessments, ensuring that your supply chain is built on a foundation of scientific excellence and commercial reliability. Let us help you secure a competitive edge in the market through superior chemical manufacturing solutions.
