Advanced Manufacturing of Clopidogrel Intermediates via Novel Strecker Reaction Pathways
The pharmaceutical industry continuously seeks robust and scalable synthetic routes for critical cardiovascular medications, and the present analysis focuses on the innovative methodology disclosed in patent CN1487943A. This intellectual property outlines a superior process for the preparation of thieno[3,2-c]pyridine derivatives, specifically targeting the synthesis of Clopidogrel and its pharmaceutically acceptable salts. Unlike traditional methods that rely on hazardous alpha-halophenylacetic acid derivatives, this novel approach leverages a Strecker reaction mechanism to construct the core carbon-nitrogen framework with exceptional efficiency. The patent details a versatile pathway capable of producing racemic or optically active forms of the drug substance, addressing the critical market demand for high-purity anti-thrombotic agents. By shifting the synthetic strategy to utilize 2-chlorobenzaldehyde and tetrahydrothienopyridine amine, the process mitigates significant safety risks associated with lachrymatory reagents while simultaneously improving overall yield profiles. For global supply chain stakeholders, understanding this technological pivot is essential for securing reliable sources of high-quality pharmaceutical intermediates.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the industrial synthesis of Clopidogrel has been plagued by reliance on alpha-halophenylacetic acid derivatives, which present severe operational challenges in a manufacturing environment. These precursors are inherently lachrymatory and irritating, posing significant health risks to personnel and requiring specialized containment infrastructure that drives up capital expenditure. Furthermore, conventional routes often involve multi-step sequences with mediocre overall yields, leading to substantial material loss and increased waste generation. The use of such hazardous chemicals complicates regulatory compliance and environmental management, making the scale-up of these traditional processes economically and logistically burdensome. Additionally, the purification of intermediates in older methods frequently requires complex chromatographic separations, which are difficult to translate from laboratory benchtop to multi-ton commercial production. These factors collectively contribute to higher production costs and longer lead times, creating vulnerabilities in the supply chain for this essential medication.
The Novel Approach
In stark contrast, the methodology described in CN1487943A introduces a streamlined synthetic route that fundamentally alters the construction of the chiral center. This novel approach utilizes a Strecker-type condensation between 4,5,6,7-tetrahydrothieno[3,2-c]pyridine, a cyanide source, and 2-chlorobenzaldehyde to generate a nitrile intermediate with remarkable efficiency. As illustrated in the overall process flow, this pathway allows for the direct formation of the key carbon-carbon bond without the need for toxic alpha-halo acids. 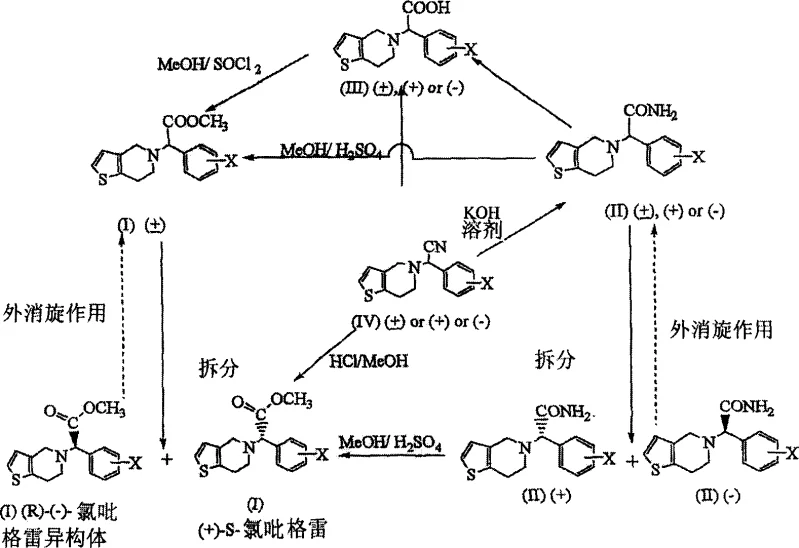 The process demonstrates exceptional versatility, enabling the conversion of the nitrile intermediate into either the acetamide or directly into the methyl ester, providing manufacturers with flexible options for process optimization. Experimental data within the patent indicates that the initial Strecker reaction can achieve yields as high as 97%, representing a drastic improvement over legacy methods. This high-yielding transformation not only reduces raw material consumption but also simplifies downstream processing, thereby facilitating cost reduction in pharmaceutical intermediates manufacturing. The ability to operate under milder conditions further enhances the safety profile, making this route highly attractive for commercial scale-up of complex pharmaceutical substances.
The process demonstrates exceptional versatility, enabling the conversion of the nitrile intermediate into either the acetamide or directly into the methyl ester, providing manufacturers with flexible options for process optimization. Experimental data within the patent indicates that the initial Strecker reaction can achieve yields as high as 97%, representing a drastic improvement over legacy methods. This high-yielding transformation not only reduces raw material consumption but also simplifies downstream processing, thereby facilitating cost reduction in pharmaceutical intermediates manufacturing. The ability to operate under milder conditions further enhances the safety profile, making this route highly attractive for commercial scale-up of complex pharmaceutical substances.
Mechanistic Insights into Strecker Reaction and Chiral Resolution
The core chemical innovation lies in the execution of the Strecker reaction, which serves as the foundation for building the molecular architecture of the drug. In this mechanism, the secondary amine, 4,5,6,7-tetrahydrothieno[3,2-c]pyridine, reacts with 2-chlorobenzaldehyde in the presence of a cyanide source such as sodium cyanide or potassium cyanide. 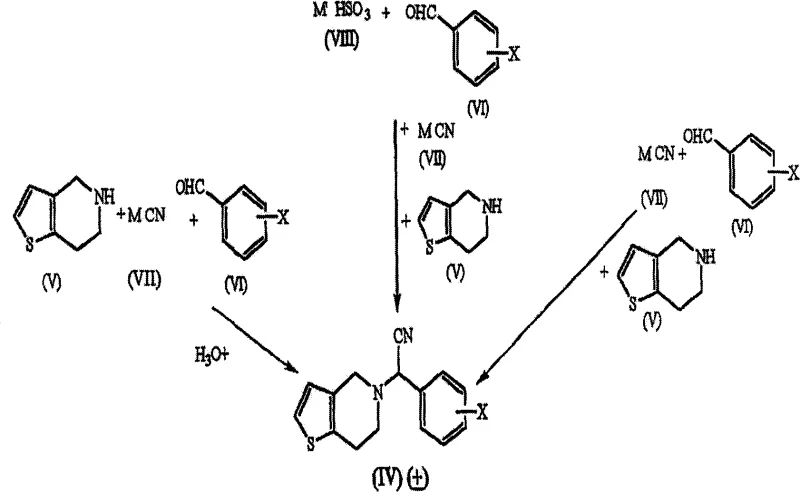 The reaction proceeds through the formation of an iminium ion intermediate, which is subsequently trapped by the cyanide nucleophile to form the alpha-amino nitrile derivative. The patent specifies that this transformation can be catalyzed by various acids, including acetic acid or hydrochloric acid, and can be conducted in aqueous or alcoholic solvents at temperatures ranging from 40°C to 80°C. This mechanistic pathway is particularly advantageous because it avoids the generation of stoichiometric amounts of halogenated byproducts. Following the formation of the nitrile, the process allows for hydrolysis to the corresponding acetamide using alkaline conditions, such as potassium hydroxide in tert-butanol, or acidic conditions. This step is crucial as the acetamide intermediate serves as an ideal candidate for chiral resolution, possessing physical properties that facilitate efficient separation of enantiomers.
The reaction proceeds through the formation of an iminium ion intermediate, which is subsequently trapped by the cyanide nucleophile to form the alpha-amino nitrile derivative. The patent specifies that this transformation can be catalyzed by various acids, including acetic acid or hydrochloric acid, and can be conducted in aqueous or alcoholic solvents at temperatures ranging from 40°C to 80°C. This mechanistic pathway is particularly advantageous because it avoids the generation of stoichiometric amounts of halogenated byproducts. Following the formation of the nitrile, the process allows for hydrolysis to the corresponding acetamide using alkaline conditions, such as potassium hydroxide in tert-butanol, or acidic conditions. This step is crucial as the acetamide intermediate serves as an ideal candidate for chiral resolution, possessing physical properties that facilitate efficient separation of enantiomers.
Control over stereochemistry is paramount in the production of Clopidogrel, as biological activity resides primarily in the (S)-(+)-enantiomer. The patent discloses a robust resolution strategy wherein the racemic acetamide is treated with a chiral resolving agent, typically (1S)-(+)-camphor-10-sulfonic acid, to form diastereomeric salts. These salts exhibit different solubility profiles, allowing for the selective crystallization of the desired (S)-isomer salt. The mother liquor containing the unwanted (R)-isomer can be subjected to racemization conditions using bases like potassium tert-butoxide, effectively recycling the material back into the process stream. This dynamic kinetic resolution capability significantly enhances atom economy and reduces waste. Furthermore, the process ensures high optical purity, with examples demonstrating enantiomeric excess values exceeding 99% after recrystallization. Such rigorous control over impurity profiles is critical for meeting stringent regulatory standards for active pharmaceutical ingredients.
How to Synthesize Clopidogrel Intermediates Efficiently
The synthesis of these critical thienopyridine derivatives involves a sequence of well-defined chemical transformations that prioritize yield and safety. The process begins with the condensation of the amine and aldehyde components to form the nitrile, followed by hydrolysis to the amide and subsequent resolution. Detailed standard operating procedures for each reaction step, including specific solvent ratios, temperature gradients, and workup protocols, are essential for reproducibility. The following guide outlines the standardized synthesis steps derived from the patent examples to ensure consistent quality.
- Perform Strecker reaction between 4,5,6,7-tetrahydrothieno[3,2-c]pyridine, 2-chlorobenzaldehyde, and a cyanide source to form the nitrile intermediate.
- Convert the nitrile intermediate to the corresponding acetamide derivative using acid or base hydrolysis conditions.
- Resolve the racemic amide using a chiral acid such as camphor sulfonic acid to isolate the optically active (S)-(+)-isomer.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain directors, the adoption of this novel synthetic route offers tangible strategic benefits that extend beyond simple chemistry. The elimination of hazardous alpha-halo reagents removes a significant bottleneck in raw material sourcing and handling, thereby enhancing supply chain reliability. Since the starting materials, such as 2-chlorobenzaldehyde and the thienopyridine amine, are commodity chemicals available from multiple global suppliers, the risk of single-source dependency is drastically minimized. This diversification of the supply base ensures continuity of supply even during market fluctuations or geopolitical disruptions. Moreover, the simplified workup procedures, which often involve straightforward extraction and crystallization rather than complex chromatography, reduce the operational complexity at the manufacturing site. This simplicity translates directly into faster batch cycle times and increased production throughput, allowing manufacturers to respond more agilely to market demand.
- Cost Reduction in Manufacturing: The economic impact of this process is driven by the substantial increase in reaction yields and the reduction in waste disposal costs. By achieving yields near quantitative levels in the initial Strecker step, the consumption of expensive starting materials is optimized, leading to significant cost savings per kilogram of product. Additionally, the ability to racemize and recycle the unwanted stereoisomer means that nearly all input material is eventually converted into the desired product, maximizing resource utilization. The avoidance of specialized containment equipment for lachrymatory agents further lowers capital and operational expenditures. These factors combine to create a highly cost-effective manufacturing model that improves margin potential for downstream partners.
- Enhanced Supply Chain Reliability: The robustness of the chemical process contributes directly to supply security. The reaction conditions are mild and tolerant of minor variations, reducing the likelihood of batch failures due to process deviations. The use of common solvents like methanol, ethanol, and acetone ensures that solvent supply chains remain stable and cost-efficient. Furthermore, the solid-state properties of the intermediates, such as the acetamide salt, facilitate easy handling, storage, and transportation without degradation. This stability allows for the strategic stocking of key intermediates, providing a buffer against unexpected demand spikes. Consequently, partners can rely on a steady and predictable flow of high-purity materials to support their own formulation and distribution schedules.
- Scalability and Environmental Compliance: From an environmental and regulatory perspective, this route represents a greener alternative to legacy technologies. The reduction in hazardous waste generation aligns with increasingly strict global environmental regulations, minimizing the risk of compliance violations. The process is inherently scalable, as demonstrated by the patent examples which cover ranges from gram to multi-gram scales without loss of efficiency. The simplicity of the unit operations involved, such as filtration and distillation, makes the technology easily transferable to large-scale reactors. This scalability ensures that the manufacturing capacity can be expanded rapidly to meet growing global demand for cardiovascular therapies without requiring extensive process re-engineering.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this synthesis technology. These answers are derived directly from the experimental data and claims presented in the patent documentation, providing clarity on process capabilities and product specifications. Understanding these details is vital for technical teams evaluating the feasibility of integrating this route into their existing manufacturing portfolios.
Q: What are the advantages of the Strecker reaction route for Clopidogrel?
A: The Strecker reaction route avoids the use of lachrymatory and irritating alpha-halophenylacetic acid derivatives found in conventional methods. It utilizes readily available starting materials like 2-chlorobenzaldehyde and offers significantly higher yields, up to 97% in initial steps, under milder reaction conditions.
Q: How is chiral purity achieved in this process?
A: Chiral purity is achieved through resolution of racemic intermediates, specifically the acetamide or nitrile forms, using chiral resolving agents like (1S)-(+)-camphor-10-sulfonic acid. The unwanted isomers can be racemized and recycled back into the process, enhancing overall efficiency.
Q: Can this process produce specific polymorphs of Clopidogrel Bisulfate?
A: Yes, the process allows for the preparation of Clopidogrel bisulfate Polymorph I, characterized by a melting point of approximately 184°C. Specific crystallization conditions involving acetone and sulfuric acid at controlled temperatures ensure the formation of the desired crystal lattice structure.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Clopidogrel Supplier
The technological advancements detailed in CN1487943A represent a significant leap forward in the manufacturing of anti-thrombotic agents, offering a pathway to higher purity and greater efficiency. NINGBO INNO PHARMCHEM stands at the forefront of adopting such innovative chemistries to serve the global pharmaceutical market. As a dedicated CDMO partner, we possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that our clients receive a consistent and reliable supply of critical intermediates. Our facilities are equipped with rigorous QC labs and adhere to stringent purity specifications, guaranteeing that every batch meets the highest international standards for safety and efficacy. We understand the complexities of chiral synthesis and have the expertise to manage resolution and racemization cycles effectively.
We invite potential partners to engage with our technical procurement team to discuss how this advanced synthesis route can benefit your specific supply chain requirements. By collaborating with us, you gain access to a Customized Cost-Saving Analysis tailored to your volume needs, helping you optimize your overall procurement strategy. We encourage you to request specific COA data and route feasibility assessments to verify the compatibility of our processes with your quality systems. Together, we can ensure the uninterrupted availability of high-quality Clopidogrel intermediates, supporting the health and well-being of patients worldwide.
