Industrial Scale Synthesis Of Tolterodine Tartrate Via Stereochemical Inversion Technology
The pharmaceutical industry continuously seeks robust synthetic pathways that balance high purity with economic viability, particularly for high-volume therapeutic agents like Tolterodine. Patent CN1245377C introduces a transformative methodology for the preparation of Tolterodine and its tartrate salt, addressing critical bottlenecks found in legacy manufacturing processes. This innovation centers on a novel stereochemical inversion technique that converts the less desirable S-enantiomer by-product back into the therapeutically active R-enantiomer, thereby maximizing atomic economy and reducing raw material waste. By circumventing the need for hazardous reducing agents such as Lithium Aluminium Hydride and eliminating expensive methylation steps, this protocol offers a safer, more sustainable route for the production of this vital urinary incontinence medication. For global procurement teams and R&D directors, understanding the mechanistic advantages of this patent is essential for securing a stable supply of high-purity pharmaceutical intermediates.
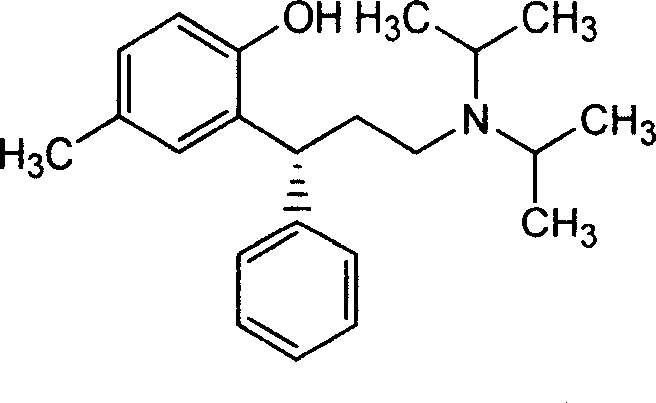
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the industrial synthesis of Tolterodine has been plagued by significant technical and economic inefficiencies that hinder scalable production. Traditional routes, such as those disclosed in earlier literature, often rely on the reduction of ester or amide groups using highly reactive and dangerous hydride reagents like DIBAL-H or Lithium Aluminium Hydride. These reagents necessitate strictly anhydrous environments and inert gas protection, creating substantial operational hazards and increasing the complexity of reactor management. Furthermore, conventional asymmetric synthesis routes can involve upwards of sixteen steps, leading to cumulative yield losses and excessive solvent consumption. The reliance on expensive chiral reagents and the inability to effectively utilize the unwanted enantiomer by-products result in inflated production costs and a larger environmental footprint, making these methods suboptimal for modern, cost-sensitive supply chains.
The Novel Approach
In stark contrast, the methodology outlined in CN1245377C streamlines the synthesis through a clever integration of condensation and stereochemical recycling. The process initiates with the acid-catalyzed condensation of p-methyl phenol and trans-cinnamyl alcohol esters bearing strong electron-withdrawing groups, avoiding the need for toxic methyl iodide. Following coupling with diisopropylamine, the resulting racemic mixture is resolved using L-(+)-tartaric acid. The true breakthrough lies in the treatment of the separated S-tolterodine by-product; rather than being discarded as waste, it undergoes a stereospecific conversion using Grignard reagents such as n-Butyl Lithium or isopropyl magnesium bromide. This inversion transforms the inactive isomer back into the active R-tolterodine, which is then re-salted to form the final tartrate product. This closed-loop approach drastically simplifies the workflow and enhances the overall mass balance of the manufacturing campaign.
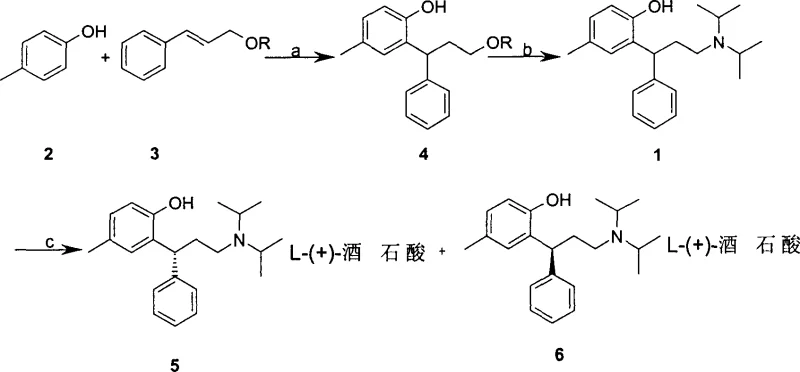
Mechanistic Insights into Acid-Catalyzed Condensation and Stereoinversion
The core chemical innovation of this patent relies on the precise control of electrophilic aromatic substitution and subsequent base-mediated configurational inversion. In the initial condensation step, the use of trans-cinnamyl alcohol esters with electron-withdrawing groups activates the allylic position for nucleophilic attack by p-methyl phenol under acidic conditions. This generates the key intermediate condensate with high regioselectivity, avoiding the formation of complex impurity profiles often seen in Friedel-Crafts alkylations. The subsequent resolution step leverages the differential solubility of diastereomeric salts formed between racemic tolterodine and L-(+)-tartaric acid. However, the most sophisticated aspect is the stereoinversion mechanism. By treating the S-enantiomer with strong organometallic bases like n-Butyl Lithium at controlled low temperatures ranging from -40°C to 25°C, the alpha-proton adjacent to the chiral center is abstracted. The resulting carbanion intermediate loses its stereochemical information and, upon reprotonation during the quench phase, equilibrates to favor the thermodynamically more stable or kinetically accessible R-configuration, effectively recycling the chiral material.
From an impurity control perspective, this mechanism offers distinct advantages over reduction-based routes. The avoidance of powerful hydride reducers eliminates the risk of over-reduction side reactions and the formation of difficult-to-remove aluminum or boron residues, which are common pain points in API purification. The purification strategy employs standard solvent extraction and recrystallization techniques using common solvents like ethyl acetate, toluene, and alcohols, ensuring that the final API meets stringent purity specifications without requiring exotic chromatography. The ability to convert the S-isomer by-product not only boosts yield but also reduces the burden on waste treatment facilities, as the volume of chiral waste requiring disposal is significantly minimized. This level of process control is critical for R&D directors aiming to validate a synthesis route that is both chemically elegant and regulatorily compliant for global markets.
How to Synthesize Tolterodine Efficiently
The implementation of this synthesis route requires careful attention to reaction parameters, particularly temperature control during the stereoinversion phase and the selection of appropriate electron-withdrawing esters for the initial condensation. The process is designed to be telescoped where possible, minimizing isolation steps to enhance throughput. While the general workflow involves condensation, amination, resolution, and inversion, the specific operational details regarding stoichiometry, solvent volumes, and quenching protocols are critical for reproducibility. For technical teams looking to adopt this methodology, adherence to the standardized operating procedures derived from the patent examples is essential to achieve the reported yields and purity levels. The detailed standardized synthesis steps see the guide below.
- Condense p-methyl phenol with trans-cinnamyl alcohol ester under acidic conditions to form the intermediate condensate.
- React the condensate with diisopropylamine to generate racemic tolterodine base.
- Resolve the racemate using L-(+)-tartaric acid, then convert the unwanted S-isomer by-product back to the active R-isomer using Grignard reagents.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the adoption of the technology described in CN1245377C translates directly into enhanced supply security and optimized cost structures. The primary economic driver is the stereochemical recycling of the S-enantiomer. In traditional resolution processes, the theoretical maximum yield is capped at 50% unless a dynamic kinetic resolution is employed; however, this patent achieves a similar effect through a discrete conversion step, effectively doubling the utility of the chiral starting materials. This leads to substantial cost savings in raw material procurement, as the expensive chiral resolving agents and precursors are utilized with near-total efficiency. Furthermore, the elimination of hazardous reagents like Lithium Aluminium Hydride reduces the costs associated with specialized handling, storage, and hazardous waste disposal, contributing to a leaner operational expenditure model without compromising on output quality.
- Cost Reduction in Manufacturing: The process achieves significant cost optimization by removing the dependency on expensive methylating agents and specialized reducing catalysts that drive up the bill of materials in conventional routes. By utilizing readily available Grignard reagents and common organic solvents for the stereoinversion step, the manufacturing overhead is drastically simplified. The ability to recycle the by-product means that the effective cost per kilogram of the active pharmaceutical ingredient is lowered, as the input material is converted into saleable product rather than waste. This logical deduction of cost efficiency makes the process highly attractive for high-volume generic drug manufacturing where margin compression is a constant challenge.
- Enhanced Supply Chain Reliability: Supply continuity is bolstered by the use of commodity chemicals rather than bespoke or scarce reagents. Conventional routes relying on specific chiral catalysts or sensitive hydrides are vulnerable to supply disruptions; in contrast, reagents like p-methyl phenol, diisopropylamine, and tartaric acid are produced on a massive global scale with stable pricing and availability. The robustness of the reaction conditions, which do not require extreme cryogenic temperatures or ultra-dry environments for every step, further ensures that production can be maintained across multiple manufacturing sites without significant technology transfer friction, guaranteeing consistent delivery timelines for downstream partners.
- Scalability and Environmental Compliance: The environmental profile of this synthesis is markedly improved, aligning with modern green chemistry principles and regulatory expectations. The avoidance of heavy metal catalysts and the reduction of solvent intensity through efficient crystallization steps simplify the wastewater treatment process. Scalability is inherently supported by the use of standard unit operations such as extraction, distillation, and filtration, which are easily replicated in large-scale reactors ranging from pilot plants to commercial tonnage facilities. This ease of scale-up ensures that supply can be rapidly ramped to meet market demand surges without the need for complex process re-engineering or new capital equipment investment.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this Tolterodine synthesis technology. These insights are derived directly from the comparative data and experimental examples provided in the patent documentation, offering clarity on yield improvements, safety profiles, and industrial feasibility. Understanding these nuances is vital for stakeholders evaluating the transition from legacy manufacturing methods to this optimized protocol.
Q: How does this patent improve the overall yield of Tolterodine production?
A: The process significantly improves yield by implementing a stereochemical conversion step. Instead of discarding the unwanted S-tolterodine by-product generated during resolution, the method utilizes Grignard reagents to invert its configuration back to the pharmacologically active R-tolterodine, effectively recycling waste into valuable product.
Q: What are the safety advantages of this synthesis route compared to conventional methods?
A: Conventional routes often rely on hazardous reducing agents like Lithium Aluminium Hydride (LiAlH4) or DIBAL-H which require strict anhydrous conditions and pose significant safety risks. This patented method avoids these dangerous reagents, utilizing milder reaction conditions and more manageable solvents, thereby enhancing operational safety and reducing regulatory compliance burdens.
Q: Is this process suitable for large-scale commercial manufacturing?
A: Yes, the patent explicitly highlights suitability for large-scale industrial production. The elimination of expensive methylating agents and the simplification of purification steps through effective crystallization and extraction protocols make the process economically viable and technically robust for multi-tonnage manufacturing campaigns.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Tolterodine Tartrate Supplier
At NINGBO INNO PHARMCHEM, we recognize that the transition to advanced synthetic methodologies requires a partner with deep technical expertise and proven manufacturing capabilities. As a leading CDMO and supplier, we possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the theoretical benefits of patent CN1245377C are fully realized in practice. Our facilities are equipped with rigorous QC labs and adhere to stringent purity specifications, guaranteeing that every batch of Tolterodine Tartrate meets the exacting standards required by global regulatory bodies. We understand the critical nature of urinary incontinence medications in the healthcare sector and are committed to maintaining uninterrupted supply chains through robust process validation and inventory management.
We invite procurement leaders and R&D directors to collaborate with us to leverage this cost-effective synthesis route for your next project. By engaging with our technical procurement team, you can request a Customized Cost-Saving Analysis tailored to your specific volume requirements. We encourage you to reach out today to obtain specific COA data and route feasibility assessments, allowing you to make informed decisions that drive value and efficiency in your pharmaceutical manufacturing operations.
