Scalable Production of High-Purity Tetramethylbiphenyl Diamine for Advanced Polyimide Applications
The rapid evolution of the flexible electronics sector demands insulating materials that possess exceptional thermal stability and mechanical strength, driving the need for advanced polyimide precursors like 4,4'-bis(4-aminophenoxy)-3,3',5,5'-tetramethylbiphenyl. Patent CN101186579B introduces a groundbreaking preparation method that addresses the critical bottlenecks of traditional synthesis, offering a pathway to high-purity intermediates essential for next-generation flexible copper-clad laminates (FCCL). This technology leverages a unique two-step process involving nucleophilic substitution followed by an innovative iron-catalyzed reduction, effectively bypassing the reliance on costly noble metals. For R&D directors and procurement specialists, this represents a significant opportunity to optimize the cost reduction in electronic chemical manufacturing while ensuring a robust supply of high-performance materials. The method's ability to operate under normal pressure with recyclable solvents aligns perfectly with modern sustainability goals, making it a superior choice for large-scale industrial adoption.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of aromatic diamines for polyimide production has relied heavily on hydrogenation processes using palladium on carbon (Pd/C) catalysts, which present substantial economic and safety challenges for commercial operations. These conventional routes often require high-pressure equipment to facilitate hydrogen gas reactions, introducing significant security risks and necessitating heavy capital investment in specialized infrastructure. Furthermore, the use of noble metal catalysts drastically inflates production costs, and the subsequent removal of trace metal residues to meet electronic-grade purity standards adds complex purification steps that generate additional hazardous waste. Traditional solvent systems also pose environmental liabilities, as many protocols struggle with efficient solvent recovery, leading to high processing costs and unfavorable environmental impacts due to wastewater generation. The cumulative effect of low yields in multi-step sequences, often hovering around 53% in older literature, further exacerbates the inefficiency, making these methods less viable for competitive mass production in the global market.
The Novel Approach
The methodology disclosed in the patent data revolutionizes this landscape by substituting the expensive hydrogenation step with a ferric chloride and hydrazine hydrate reduction system that operates safely at atmospheric pressure. This novel approach not only eliminates the safety hazards associated with high-pressure hydrogen but also removes the dependency on scarce and expensive palladium resources, thereby fundamentally altering the cost structure of the final product. By utilizing a mixed solvent system comprising strong polar aprotic solvents and water-insoluble organic solvents, the process enables efficient water separation during the reflux stage, which drives the equilibrium forward and significantly boosts reaction yields to over 90% in the first step. The integration of activated carbon in the reduction phase further enhances product quality by adsorbing impurities, resulting in a final crystalline product with purity levels exceeding 99% without the need for energy-intensive recrystallization. This streamlined workflow ensures that the commercial scale-up of complex polymer additives becomes more feasible, offering a distinct competitive advantage in terms of both operational simplicity and environmental compliance.
Mechanistic Insights into FeCl3-Catalyzed Reduction
The core chemical transformation in this synthesis involves a precise nucleophilic aromatic substitution followed by a selective reduction, both of which are optimized to minimize byproduct formation and maximize atom economy. In the initial stage, 3,3',5,5'-tetramethyl-4,4'-biphenol reacts with 4-halonitrobenzene in the presence of a salt-forming agent, where the phenoxide ion attacks the electron-deficient aromatic ring of the nitrobenzene derivative. The use of a mixed solvent system is critical here, as it facilitates the azeotropic removal of water generated during the reaction, shifting the equilibrium towards the formation of the nitro-ether intermediate according to Le Chatelier's principle. This careful control of reaction conditions prevents the hydrolysis of reactants and ensures a high conversion rate, laying the foundation for a high-yield process.
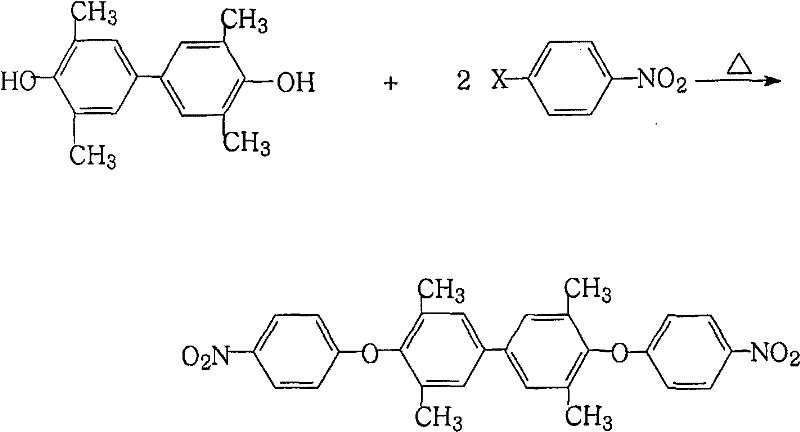
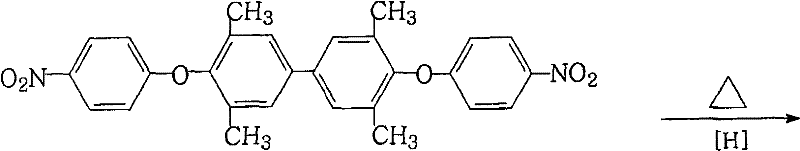
Following the isolation of the nitro intermediate, the second stage employs a sophisticated reduction mechanism where ferric chloride acts as a catalyst in conjunction with hydrazine hydrate. Unlike catalytic hydrogenation which relies on surface adsorption of hydrogen gas, this chemical reduction proceeds through a transfer hydrogenation mechanism where hydrazine serves as the hydrogen donor. The iron species facilitate the electron transfer necessary to reduce the nitro groups to amino groups while the activated carbon serves a dual purpose as both a support and an adsorbent for colored impurities. This mechanism is particularly advantageous because it avoids the over-reduction or hydrogenolysis of the ether linkage, a common side reaction in harsh hydrogenation conditions that can compromise the structural integrity of the molecule. The result is a highly selective transformation that preserves the methyl substituents and the ether bridge, which are crucial for the thermal and mechanical properties of the resulting polyimide.
How to Synthesize 4,4'-Bis(4-aminophenoxy)-3,3',5,5'-tetramethylbiphenyl Efficiently
Implementing this synthesis route requires strict adherence to the specified molar ratios and temperature profiles to ensure consistent quality and yield across batches. The process begins with the precise mixing of the biphenol derivative and the halonitrobenzene in a reactor equipped with a water separator, followed by the controlled addition of the base and solvent mixture to initiate the etherification. Once the intermediate is isolated and dried, it is subjected to the reduction conditions where temperature control between 50°C and 90°C is vital to manage the exothermic nature of the hydrazine reaction. Detailed standardized synthesis steps are provided below to guide technical teams in replicating these results reliably.
- Perform nucleophilic substitution of 3,3',5,5'-tetramethyl-4,4'-biphenol with 4-halonitrobenzene using a salt-forming agent in a mixed solvent system with water separation.
- Isolate the intermediate 4,4'-bis(4-nitrophenoxy)-3,3',5,5'-tetramethylbiphenyl via filtration and drying after concentration.
- Reduce the nitro intermediate to the final diamine using ferric chloride and hydrazine hydrate in an alcoholic solvent with activated carbon.
Commercial Advantages for Procurement and Supply Chain Teams
From a strategic sourcing perspective, this patented method offers profound benefits that directly impact the bottom line and supply chain resilience for manufacturers of high-purity OLED material and polyimide precursors. By eliminating the need for noble metal catalysts, the process removes a major variable cost driver that is subject to volatile global market prices, ensuring more stable and predictable manufacturing expenses over time. The ability to recycle solvents repeatedly not only reduces the consumption of raw materials but also simplifies waste management protocols, leading to substantial cost savings in environmental compliance and disposal fees. Furthermore, the operation under normal pressure reduces the maintenance and insurance costs associated with high-pressure vessels, contributing to a lower total cost of ownership for the production facility.
- Cost Reduction in Manufacturing: The substitution of palladium catalysts with inexpensive iron salts drastically lowers the direct material costs associated with the reduction step, which is traditionally one of the most expensive phases in diamine synthesis. This change also eliminates the need for specialized equipment to recover precious metals, simplifying the downstream processing and reducing labor hours spent on catalyst handling and regeneration. Consequently, the overall production cost is significantly reduced, allowing for more competitive pricing strategies in the global market for specialty chemicals without compromising on profit margins.
- Enhanced Supply Chain Reliability: Relying on abundant and readily available reagents like ferric chloride and hydrazine hydrate mitigates the risk of supply disruptions that are common with imported noble metals or specialized high-pressure gases. The robustness of the reaction conditions means that production can be maintained consistently even if there are fluctuations in the availability of specific high-tech equipment, as the process does not demand exotic infrastructure. This reliability ensures reducing lead time for high-purity polymer additives, enabling manufacturers to respond more quickly to market demands and secure long-term contracts with confidence.
- Scalability and Environmental Compliance: The process is inherently designed for scalability, with solvent systems that are easy to recover and reuse, minimizing the volume of hazardous waste generated per kilogram of product. This aligns with increasingly stringent global environmental regulations, reducing the risk of regulatory fines and enhancing the corporate sustainability profile of the manufacturer. The simplified three-waste treatment requirements mean that scaling up from pilot to commercial production involves fewer engineering hurdles, facilitating a smoother transition to full-scale manufacturing capacity.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this synthesis method, derived directly from the patent's background and beneficial effects sections. Understanding these details is crucial for technical teams evaluating the feasibility of adopting this route for their specific production lines. The answers provided reflect the objective data and logical deductions found within the intellectual property documentation.
Q: Why is the FeCl3/Hydrazine system preferred over Pd/C for this reduction?
A: The FeCl3/Hydrazine system eliminates the need for expensive noble metal catalysts like Palladium on Carbon, significantly reducing raw material costs and removing the complex step of heavy metal residue clearance, which is critical for electronic grade purity.
Q: How does the solvent system improve environmental compliance?
A: The process utilizes a mixed solvent system where organic solvents can be efficiently recovered and recycled repeatedly. This minimizes waste discharge and reduces the overall environmental footprint compared to traditional methods that generate significant wastewater.
Q: What purity levels can be achieved with this synthesis method?
A: The optimized protocol consistently yields products with purity exceeding 99%, meeting the stringent requirements for high-performance polyimide applications in flexible copper-clad laminates and microelectronics without requiring extensive recrystallization.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 4,4'-Bis(4-aminophenoxy)-3,3',5,5'-tetramethylbiphenyl Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical role that high-quality intermediates play in the performance of final electronic materials, which is why we have invested heavily in mastering complex synthetic pathways like the one described in CN101186579B. Our team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that we can meet the rigorous demands of the global supply chain with consistency and precision. We maintain stringent purity specifications through our rigorous QC labs, guaranteeing that every batch of 4,4'-bis(4-aminophenoxy)-3,3',5,5'-tetramethylbiphenyl meets the exacting standards required for advanced polyimide applications.
We invite you to collaborate with us to leverage these technological advancements for your own product lines, offering a partnership that goes beyond simple transaction to true technical cooperation. Please contact our technical procurement team to request a Customized Cost-Saving Analysis tailored to your specific volume requirements and to obtain specific COA data and route feasibility assessments. Together, we can drive innovation in the electronic materials sector while optimizing your operational efficiency and cost structure.
